Chapter: Biotechnology Applying the Genetic Revolution: Nanobiotechnology
Quantum Size Effect and Nanocrystal Colors
QUANTUM
SIZE EFFECT AND NANOCRYSTAL COLORS
When materials are subdivided
into sufficiently small fragments, quantum effects begin to influence their
physical properties. The fluorescent nanoparticles discussed earlier are in
fact semiconductors that are small
enough to show such quantum effects.
Semiconductors are substances
that conduct electricity under some conditions but not others. In N-type semiconductors (as in normal
electric wires) the current consists of negatively charged electrons. In P-type semiconductors the current
consists of holes.
A hole is the absence of an
electron from an atom. Although not physical particles, holes can move from
atom to atom. Electrons and holes may combine and cancel out, a process that
releases energy. Conversely, energy absorbed by certain semiconductors may
generate an electron-hole pair whose two components may then move off in
different directions.
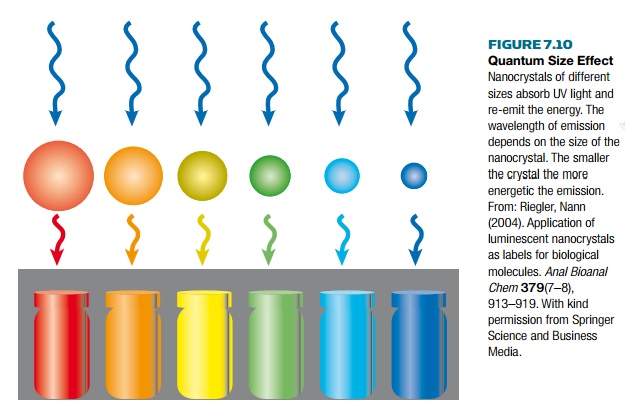
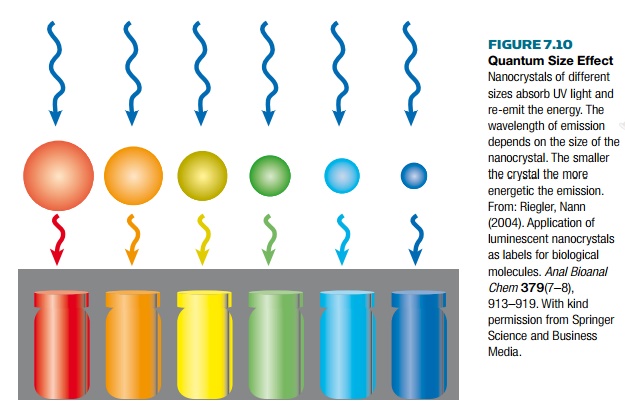
Nanoparticle labels can be
made with different emission wavelengths, covering the UV, visible spectrum,
and near infrared. Emission wavelengths obviously vary depending on the
semiconductor material. However, in addition, the quantum size effect (Fig.
7.10) allows the same semiconductor to emit at different wavelengths, depending
on the size of the nanoparticle. The smaller the nanoparticle the shorter the
wavelength (i.e., the higher the energy).
Fluorescent nanoparticles may
be regarded as miniaturized light-emitting diodes (LEDs). These are
semiconductors that work by absorbing energy (either electrical or light) and
creating electron-hole pairs. When the electrons and holes recombine, light is
emitted. For bulk material, the energy, and hence the wavelength, of the
emitted light depends on the chemical composition of the semiconductor.
However, at nanoscale dimensions, quantum effects become significant.
If the physical size of the
semiconductor is smaller than the natural radius (the Bohr radius) of the electron-hole pair, extra energy is needed to
confine the electron-hole pair. This is referred to as quantum confinement and occurs with nanocrystals of around 20 nm or less.
The smaller the semiconductor
crystal, the more energy is needed and the more energetic (shorter in
wavelength) is the light released.
Related Topics