Chapter: Biotechnology Applying the Genetic Revolution: Nanobiotechnology
Ion Channel Nanosensors
ION
CHANNEL NANOSENSORS
Somewhat more complex than
nanotubes and nanowires are nanoscale
ion channels that are assembled into membranes. These channels are designed so that they can be controlled to
permit the movement of ions under only certain conditions. The ion flow
generates an electrical current that is detected, amplified, and displayed by
appropriate electronic apparatus.
Ion channels can be used as
biosensors by attaching a binding site for the target molecule at the entry to
the channel. Attached antibodies are often used for the binding sites as
described (see Fig. 23.16). The simplest arrangement results in the channel
being open in the absence of the target molecule and shut when it is detected.
A drop in ion flow therefore signals detection of the target molecule.
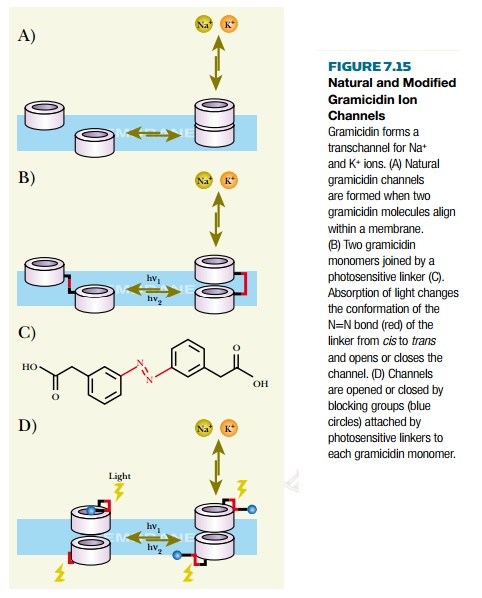
The channels are made
responsive by attaching an appropriate ligand molecule to the front end of the
gramicidin, so that it projects outward from the membrane surface. This ligand
is chosen to bind the target molecule and may be an antigen or other small molecule
that is recognized by an antibody or protein receptor. It is also possible to
attach a single-stranded segment of DNA that will recognize and bind the
complementary sequence. Thus biosensors may be designed to respond to the
presence of a variety of biological molecules.
The membrane itself may be a lipid bilayer made using natural membrane lipids. Typical phospholipids span half a membrane (i.e., one monolayer), and the two monolayers can therefore slide relative to one another. The membrane may be stabilized by including lipids that span the whole membrane. These may be found naturally in certain archaebacteria or they may be synthesized artificially. Lipid bilayers are relatively fragile and in practice must be assembled on some solid support. Building a long-lasting and stable membrane structure has so far proven difficult, and practical ion channel sensors are still under development.
Related Topics