Chapter: Basic Concept of Biotechnology : Animal Biotechnology
Protocol for Microbial Decontamination
Protocol for
Microbial Decontamination
1.
Collect the contaminated medium
carefully. If possible, the organism should be tested for sensitivity to a
range of individual antibiotics. If not, autoclave the medium or add
hypochlorite.
2.
Washthe cells in DBSS (Hanks BSS without
bicarbonate, with Penicillin, Streptomycin, Amphotericin B and Kanamycin or
Gentamycin). For monolayers, rinse the culture 3 times with DBSS, trypsinize
and then wash the cells twice more in DBSS by centrifugation and re-suspension.
For suspension cultures, wash the culture five times (in DBSS) by
centrifugation and re-suspension.
3.
Reseed a fresh flask at the lowest
reasonable seeding density, depending on cell type.
4.
Add high antibiotic medium and change
the culture every 2 days.
5.
Subculture in a high antibiotic medium.
Repeat Steps 1 to 4for three subcultures.
6.
Remove the antibiotics, and culture the
cells without them for a further three subcultures.
7.
Recheck the cultures (phase contrast
microscopy, Hoechst staining).
8.
Culture the cells for a further two
months without antibiotics, and check to make sure that all contamination has
been eliminated.
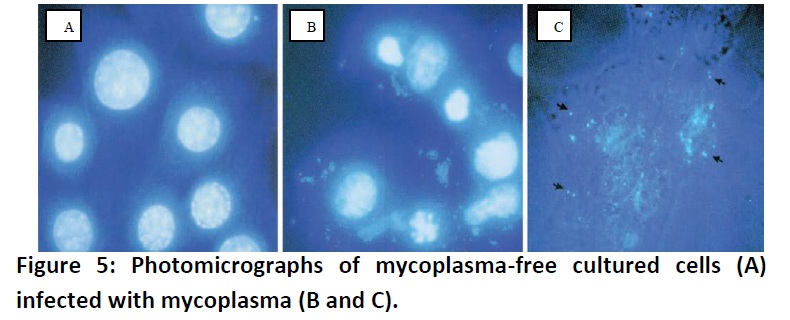
Mycoplasmas
are simple bacteria that lack a cell wall, and they are considered the smallest
self-replicating organism. Because of their extremely small size (typically
less than one micrometer), mycoplasma are very difficult to detect until they
achieve extremely high densities and cause the cell culture to deteriorate;
until then, there are often no visible signs of infection. Chronic mycoplasma
infections might manifest themselves with decreased rate of cell proliferation,
reduced saturation density, and agglutination in suspension cultures; (Fig. 5)
however, the only assured way of detecting mycoplasma contamination is by
testing the cultures periodically using fluorescent staining (e.g., Hoechst
33258), ELISA, PCR, immunostaining, autoradiography, or microbiological assays
Related Topics