Chapter: Basic Concept of Biotechnology : Animal Biotechnology
Embryonic stem cells grown in the laboratory
Embryonic stem cells grown in the laboratory
Growing cells in the laboratory is known as cell culture. Human embryonic stem cells (hESCs) are generated by transferring cells from a preimplantationstage embryo into a plastic laboratory culture dish that contains a nutrient broth known as culture medium. The cells divide and spread over the surface of the dish. The inner surface of the culture dish is typically coated with mouse embryonic skin cells that have been treated so they will not divide. This coating layer of cells is called afeeder layer. The mouse cells in the bottom of the culture dish provide the cells a sticky surface to which they can attach. Also, the feeder cells release nutrients into the culture medium. Researchers have devised ways to grow embryonic stem cells without mouse feeder cells. This is a significant scientific advance because of the risk that viruses or other macromolecules in the mouse cells may be transmitted to the human cells.
The process of generating an embryonic stem cell line is somewhat inefficient, so lines are not produced each time cells from the preimplantation stage embryo are placed into a culture dish. However, if the plated cells survive, divide, and multiply enough to crowd the dish, they are removed gently and plated into several fresh culture dishes. The process of re-plating or subculturingthe cells is repeated many times and for many months. Each cycle of subculturing the cells is referred to as a passage. Once the cell line is established, the original cells yield millions of embryonic stem cells. Embryonic stem cells that have proliferated in cell culture for six or more months without differentiating, are pluripotent, and appear genetically normal are referred to as an embryonic stem cell line. At any stage in the process, batches of cells can be frozen and shipped to other laboratories for further culture and experimentation.
Embryonic stem cells stimulated to differentiate
As long as the embryonic stem cells in culture are grown under appropriate conditions, they can remain undifferentiated (unspecialized). But if cells are allowed to clump together to form embryoid bodies, they begin to differentiate spontaneously. They can form muscle cells, nerve cells, and many other cell types. Although spontaneous differentiation is a good indicator that a culture of embryonic stem cells is healthy, the process is uncontrolled andtherefore an inefficient strategy to produce cultures of specific cell types. So, to generate cultures of specific types of differentiated cells heart muscle cells, blood cells, or nerve cells, for example: scientists try to control the differentiation of embryonic stem cells. They change the chemical composition of the culture medium, alter the surface of the culture dish, or modify the cells by inserting specific genes. Through years of experimentation, scientists have established some basic protocols or “recipes” for the directed differentiationof embryonic stem cells into some specific cell types (Fig. 8).
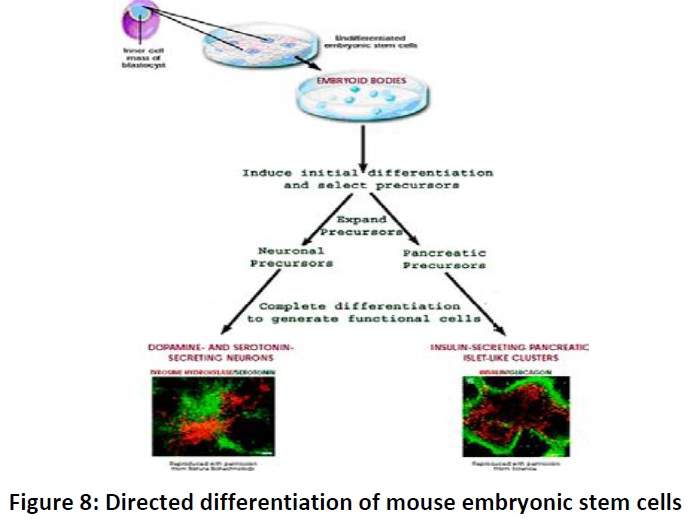
If scientists can reliably direct the differentiation of embryonic stem cells into specific cell types, they may be able to use the resulting, differentiated cells to treat certain diseases in the future. Diseases that might be treated by transplanting cells generated from human embryonic stem cells include diabetes, traumatic spinal cord injury,Duchenne’s muscular dystrophy, heart disease, and vision and hearing loss.
· Adult stem cells:
An adult stem cell is thought to be an undifferentiated cell, found among differentiated cells in a tissue or organ that can renew itself and can differentiate to yield some or all of the major specialized cell types of the tissue or organ. The primary roles of adult stem cells in a living organism are to maintain and repair the tissue in which they are found. Scientists also use the term somatic stem cell instead of adult stem cell, where somatic refers to cells of the body (not the germ cells, sperm or eggs). Unlike embryonic stem cells, which are defined by their origin (cells from the preimplantation-stage embryo), the origin of adult stem cells in some mature tissues is still under investigation. Research on adult stem cells has generated a great deal of excitement. Scientists have found adult stem cells in many more tissues than they once thought possible. This finding has led researchers and clinicians to ask whether adult stem cells could be used for transplants. In fact, adult hematopoietic, or blood-forming, stem cells from bone marrow have been used in transplants for 40 years. Scientists now have evidence that stem cells exist in the brain and the heart. If the differentiation of adult stem cells can be controlled in the laboratory, these cells may become the basis of transplantation-based therapies
The history of research on adult stem cells began about 50 years ago. In the 1950s, researchers discovered that the bone marrow contains at least two kinds of stem cells. One population, called hematopoietic stem cells, forms all the types of blood cells in the body. A second population, called bone marrow stromal stem cells (also called mesenchymal stem cells, or skeletal stem cells by some) was discovered a few years later. These non-hematopoietic stem cells make up a small proportion of the stromal cell population in the bone marrow and cangenerate bone, cartilage, and fat cells that support the formation of blood and fibrous connective tissue. In the 1960s, scientists who were studying rats discovered two regions of the brain that contained dividing cells that ultimately become nerve cells. Despite these reports, most scientists believed that the adult brain could not generate new nerve cells. It was not until the 1990s that scientists agreed that the adult brain does contain stem cells that are able to generate the brain’s three major cell types - astrocytes and oligodendrocytes, which are non-neuronal cells, and neurons, or nerve cells.
· Adult stem cell differentiation:
As indicated above, scientists have reported that adult stem cells occur in many tissues and that they enter normal differentiation pathways to form the specialized cell types of the tissue in which they reside. Normal differentiation pathways of adult stem cells. In a living animal, adult stem cells are available to divide for a long period, when needed, and can give rise to mature cell types that have characteristic shapes and specialized structures and functions of a particular tissue. The following are examples of differentiation pathways of adult stem cells (Fig. 9) that have been demonstrated in vitroor in vivo.
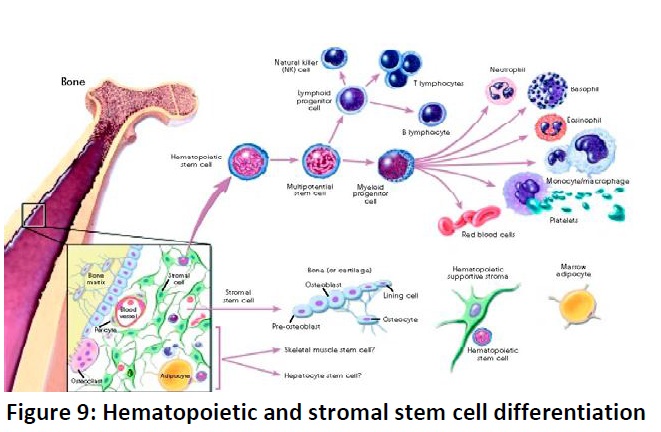
· Hematopoietic stem cells give rise to all the types of blood cells:red blood cells, B lymphocytes, T lymphocytes, natural killer cells, neutrophils, basophils, eosinophils, monocytes, and macrophages.
· Mesenchymal stem cells have been reported to be present inmany tissues. Those from bone marrow (bone marrow stromal stem cells, skeletal stem cells) give rise to a variety of cell types: bone cells (osteoblasts and osteocytes), cartilage cells (chondrocytes), fat cells (adipocytes), and stromal cells that support blood formation. However, it is not yet clear how similar or dissimilar mesenchymal cells derived from non-bone marrow sources are to those from bone marrow stroma.
· Neural stem cells in the brain give rise to its three major celltypes: nerve cells (neurons) and two categories of non-neuronal cells - astrocytes andoligodendrocytes.
· Epithelial stem cells in the lining of the digestive tract occur indeep crypts and give rise to several cell types: absorptive cells, goblet cells, Paneth cells, and enteroendocrine cells.
· Skin stem cells occur in the basal layer of the epidermis and atthe base of hair follicles. The epidermal stem cells give rise to keratinocytes, which migrate to the surface of the skin and form a protective layer. The follicular stem cells can give rise to both the hair follicle and to the epidermis.
Transdifferentiation:
A number of experiments have reported that certain adult stem cell types can differentiate into cell types seen in organs or tissues other than those expected from the cells’ predicted lineage (i.e., brain stem cells that differentiate into blood cells or blood-forming cells that differentiate into cardiac muscle cells, and so forth). Although isolated instances of transdifferentiation have been observed in some vertebrate species, whether this phenomenon actually occurs in humans is under debate by the scientific community. Instead of transdifferentiation, the observed instances may involve fusion of a donor cell with a recipient cell. Another possibility is that transplanted stem cells are secreting factors that encourage the recipient’s own stem cells to begin the repair process. Even when transdifferentiation has been detected, only a very small percentage of cells undergo the process.
In a variation of transdifferentiation experiments, scientists have recently demon strated that certain adult cell types can be “reprogrammed” into other cell types in vivo using a well-controlled process of genetic modification. This strategy may offer a way to reprogram available cells into other cell types that have been lost or damaged due to disease. For example, one recent experiment shows how pancreatic beta cells, the insulin-producing cells that are lost ordamaged in diabetes, could possibly be created by reprogramming other pancreatic cells. By “re-starting” expression of three critical beta cell genes in differentiated adult pancreatic exocrine cells, researchers were able to create beta cell-like cells that can secrete insulin. The reprogrammed cells were similar to beta cells in appearance, size, and shape; expressed genes characteristic of beta cells; and were able to partially restore blood sugar regulation in mice whose own beta cells had been chemically destroyed. While not transdifferentiation by definition, this method for reprogramming adult cells may be used as a model for directly reprogramming other adult cell types.
In addition to reprogramming cells to become a specific cell type, it is now possible to reprogram adult somatic cells to become like embryonic stem cells (induced pluripotent stem cells, iPSCs) through the introduction of embryonic genes. Thus, a source of cells can be generated that are specific to the donor, thereby avoiding issues of histocompatibility, if such cells were to be used for tissue regeneration. However, like embryonic stem cells, determination of the methods by which iPSCs can be completely and reproducibly committed to appropriate cell lineages is still under investigation.
Related Topics