Chapter: Introduction to Human Nutrition: Digestion and Metabolism of Carbohydrates
Nonglycemic carbohydrates
Nonglycemic carbohydrates
Carbohydrates that are not absorbed in the small intestine enter the large bowel, where they are par-tially or completely broken down by bacteria in the colon by a process called fermentation. McCance and Lawrence in 1929 were the first to classify carbohy-drates as “available” and “unavailable.”
They realized that not all carbohydrates provide “carbohydrates for metabolism” to the body. They called these carbohy-drates “unavailable.” This was a very useful concept because it drew attention to the fact that some carbo-hydrate is not digested and absorbed in the small intestine, but rather reaches the large bowel where it is fermented. However, it is now realized that it is misleading to talk of carbohydrate as unavailable because some indigestible carbohydrate can provide the body with energy through fermentation in the colon. Thus, “unavailable carbohydrates” are not really unavailable. For this reason, it has been sug-gested by the Food and Agriculture Organization (FAO 1998) of the United Nations and World Health Organization that the term “nonglycemic carbohy-drates” is more appropriate.
Nature of carbohydrates that enter the colon
Carbohydrates that enter the colon can be classified either physiologically or chemically. Neither of these classifications is entirely satisfactory because it is dif-ficult to measure the physiologically indigestible car-bohydrate and this varies in different people. Further, the chemical structure of carbohydrates does not always predict their physiological behavior.
Physiological classification of carbohydrates entering the colon
Carbohydrates enter the colon because (1) monosac-charide transporters do not exist in the intestinal mucosa or do not function at a high enough rate; (2) the enzymes needed to digest the carbohydrates are not present in the small intestine; (3) the enzymes are present but cannot gain access to the carbohydrates; or (4) the enzymes do not digest the carbohydrates rapidly enough for them to be completely absorbed. In addition, a small amount of carbohydrate entering the colon consists of carbohydrate residues occurring on mucopolysaccharides (mucus) secreted by the small and large intestinal mucosal cells.
Some carbohydrates are always nonglycemic because the human species lacks the enzymes neces-sary for their digestion. However, a significant pro-portion (perhaps up to half ) of all carbohydrates that escape digestion in the small intestine have a chemical structure which is such that they could potentially be digested or absorbed in the small intestine, but they are variably absorbed for various reasons, examples of which are given below.
First, some monosaccharides and sugar alcohols are only partially absorbed because of low affinity for intestinal transporters. Xylose is taken up by the glucose transporter, but is only partly absorbed because of a low affinity. Fructose is poorly absorbed on its own, but readily absorbed in the presence of glucose. The surface area of the small intestine avail-able for absorption is reduced by diseases that cause atrophy of the intestinal mucosa, such as tropical sprue or celiac disease, or surgical resection of a portion of the intestine (e.g., for Crohn’s disease). An increased rate of intestinal transit (e.g., high osmotic load in the small intestinal lumen from undigested sugars) reduces the time available for absorption to occur.
Third, although starch (amylopectin or amylose) is potentially digested in the small intestine, if it is trapped inside intact cell walls or other plant cell structures, intestinal enzymes may not be able to gain access to it, and therefore it remains undigested. The digestibility of the carbohydrates in banana depends on the degree of ripeness. The starch in green banana is very indigestible, but, as the banana ripens, the starch is converted to digestible sugars.
Finally, there are many reasons why carbohydrates may not be digested rapidly enough to be completely absorbed. Some forms of retrograded or resistant starch, or foods with a large particle size, are digested so slowly that the time spent in the small intestine is not long enough for their complete digestion. Diges-tion of these carbohydrates can be altered by factors that affect transit time. The presence of osmotically active and unabsorbed molecules (such as unabsorbed sugars) will draw water into the intestine and speed the rate of transit. Substances that increase bulk, such as wheat bran, will have similar effects. Transit rate is slowed in old age and in the presence of viscous fibers. Drugs may increase or decrease the rate of transit. Certain disorders can also affect transit time, such as gastroparesis, a complication of type I diabetes.
Chemical classification of carbohydrates entering the colon
The chemical classification of carbohydrates entering the colon is as follows:
●Monosaccharides: all except for glucose, fructose, and galactose are partly or completely unabsorbed. Fructose in the absence of a source of glucose (mono-, di-, or polysaccharide) is partly unabsorbed.
●Sugar alcohols: all are partly or completely unabsorbed.
●Disaccharides: all except for maltose, sucrose, and lactose are unabsorbed. Lactose is completely or partly unabsorbed in individuals with low intestinal lactase activity.
●Oligosaccharides: all are unabsorbed except for maltodextins.
●Polysaccharides: all nonstarch polysaccharides are unabsorbed.
●Resistant starch.
Amount of carbohydrate entering the colon
It is difficult to measure the amount of carbohydrate entering the human colon. However, it has been esti-mated that at least 30 g of carbohydrate is required to support the growth of the bacterial population in the colon of an individual on a typical Western diet pro-ducing about 100 g stool per day. About half of that amount will come from nonstarch polysaccharide (NSP, also known as dietary fiber), 1–2 g from indi- gestible oligosaccharides, and probably about 1–2 g from intestinal mucopolysaccharides. These compo-nents add up to only 18–20 g. Where does the other 10–12 g come from? It is believed to come from starch, because experiments in humans show that about 5– 15% of the starch in foods enters the colon. Typical Western diets contain about 120–150 g starch per day, and, if 8% of this enters the colon, this will provide the additional 10–12 g carbohydrate. The amount of carbohydrate entering the colon, however, can be increased several-fold, up to 100 g/day or more, by changes in diet such as increased intake of NSP, non-digestible or partially digestible carbohydrates (ingredients in functional foods), total starch, resis-tant starch, or slowly digested, low-GI foods.
Resistant starch
Resistant starch is starch that escapes digestion in the small intestine and enters the colon. However, there is controversy over the amounts of resistant starch in foods because there is no universally accepted method for measuring it (different methods yield different results). The amount of resistant starch measured chemically is generally less than that observed to enter the colon (or leave the small intestine) in experiments in human volunteers.
The main forms of resistant starch (RS) are physi-cally enclosed starch, for example within intact cell structures (known at RS1); raw starch granules (RS2); and retrograded amylose (RS3). These kinds of starch can be identified chemically using methods developed by Englyst and colleagues (Englyst et al. 1996).
Dietary fiber
Major interest in dietary fiber began in the early 1970s with the proposal by Burkitt and Trowell (1975) that many Western diseases were due to a lack of fiber in the diet. However, the definition of dietary fiber has been, and continues to be, a source of scientific con-troversy. Indeed, two consecutive reports from the FAO (1997 and 1998) recommended that the term “dietary fiber” be phased out. Nevertheless, the term appears to be here to stay because it is accepted by consumers, the food industry, and governments.
A definition and method of measuring fiber is important for scientific studies and for food-labeling purposes. The student must be aware that the defini-tions and methods of measuring fiber have changed over time, and differ in different parts of the world. Knowledge of what is meant by the term “fiber” and what is included in the measurement is essential for proper interpretation of the scientific literature (but often is not given in the methods section of papers and reports).
The two main methods used to determine dietary fiber are chemical and gravimetric. In the chemical method (used in the UK), the residue is subjected to acid hydrolysis and the resultant sugars are measured colorimetrically, by gas chromatography or by high-performance liquid chromatography. The sum of all these sugars constitutes the NSP. The chemical method includes only carbohydrates in the NSP. In the gravi-metric method (used in the USA and elsewhere), the residue is dried and weighed, and the amounts of protein and mineral materials present are subtracted (after separate analyses). The gravimetric method includes the NSP, plus other noncarbohydrate com-ponents such as lignin and waxes. Recently, all coun-tries in Europe have recognized the gravimetric method as an approved method for measuring fiber in foods.
fiber. There are many other indigestible carbohydrate and noncarbohydrate compounds, both natural and artificial, that could be classified as “fiber” (e.g., poly-dextrose, sucrose polyester, styrofoam). Should these be included in dietary fiber? In favor of this is the argument that some of these materials have physio-logical properties associated with fiber, such as stool bulking, or effects on satiety or blood glucose and cholesterol. Against this is the feeling that dietary fiber should include only plant materials that are normally present in the diet. These are not easy issues and they have not been resolved.
Intakes of dietary fiber, oligosaccharides, and other indigestible sugars
Vegetarians tend to have higher fiber intakes than omnivores. The typical intake of dietary fiber in North America and northern and central Europe is about 15 g/day. In Scandinavia and Italy, fiber consumption is 20–30 g/day, whereas in African countries such as Uganda, Kenya, Malawi, and Nigeria intakes may be as high as 50 g/day or more. Naturally occurring oligosaccharides are consumed in legumes, onions, fennel, chicory, and similar foods. Intakes in Western countries are probably up to 2–4 g/day. Fructo- and galactooligosaccharides are now being added to certain “functional foods” in a number of countries, and intakes from such sources may increase substan-tially (up to 10–20 g/day). Many kinds of indigestible or partially digested carbohydrates are entering the food supply in dietetic, diabetic, or functional foods, including sugar alcohols (polyols, e.g., sorbitol, man-nitol, lactitol), polydextrose, resistant starch, hydroge-nated starch, and other chemically modified starches and carbohydrates. Thus, the total amount of carbo-hydrate entering the colon could become very sub-stantial for people using these foods. Individually, these ingredients are generally recognized as safe, and evidence from populations consuming 50 g and more NSP per day suggests that the colon has the capacity to adapt to large increases in the load of carbohydrate. However, safe upper limits of intake are unknown and the health implications of an increased supply of a wide range of carbohydrates to the colon are currently based on inference rather than scientific data.
Fermentation in the colon
The colon contains a complex ecosystem consisting of over 400 known species of bacteria that exist in a sym-biotic relationship with the host. The bacteria obtain the substrates that they require for growth from the host, and return to the host the by-products of their metabolism. The major substrate that the bacteria receive from the host is carbohydrate, mostly in the form of polysaccharides. They obtain nitrogen from urea (which diffuses into the colon from the blood) andundigestedaminoacidsandproteins.Fermentation is the process by which microorganisms break down monosaccharides and amino acids to derive energy for their own metabolism. Fermentation reactions do not involve respiratory chains that use molecular oxygen or nitrate as terminal electron acceptors. Most of the fermentation in the human colon is anaerobic, i.e., it proceeds in the absence of a source of oxygen. Different bacteria use different substrates via different types of chemical reaction. However, as a summary of the overall process, fermentation converts carbohydrates to energy, plus various end-products, which include the gases carbon dioxide, hydrogen, and methane, and the SCFAs acetic (C2), propionic (C3), and butyric (C4) acids. Acetate, propionate, and butyrate appear in colonic contents in approximate molar ratios of 60:20:20, respectively. Most of the SCFAs produced are absorbed and provide energy for the body (Figure 5.3).
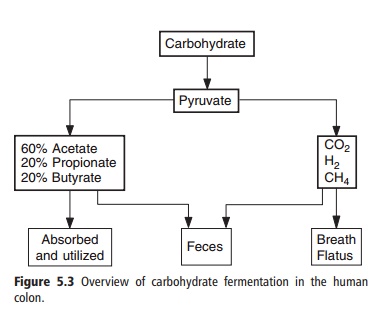
The roles of SCFAs in metabolism are discussed later. Formic acid (C1) and minor amounts of longer chain SCFAs and branched-chain SCFAs may also be produced. In addition, lactic and succinic acids and ethanol or methanol may be intermediate or end-products depending on the conditions of the fermentation. For example, rapid fermentation in an environment with a low pH results in the accumulation of lactic and succinic acids.
The first step in fermentation is the breakdown of polysaccharides, oligosaccharides, and disaccharides to their monosaccharide subunits. This is achieved either by the secretion of hydrolytic enzymes by bac-teria into the colonic lumen or, more commonly, by expression of such enzymes on the bacterial surface so that the products of hydrolysis are taken up directly by the organism producing the enzyme. To degrade the NSP of dietary fiber, the bacteria may need to attach themselves to the surface of the remnants of the plant cell walls or other particulate material.
Once the monosaccharide is internalized, the majority of carbohydrate-fermenting species in the colon use the glycolytic pathway to metabolize carbo-hydrate to pyruvate. This pathway results in the reduction of NAD+ to NADH. Fermentation reactions are controlled by the need to maintain redox balance between reduced and oxidized forms of pyridine nucleotides. The regeneration of NAD+ may be achieved in a number of different ways (Figure 5.4).
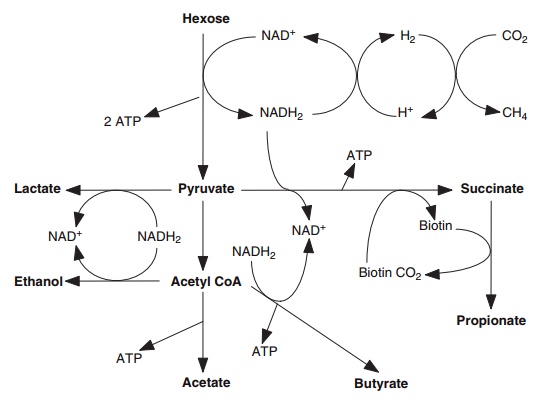
Figure 5.4 Summary of biochemical pathways used by the anaerobic bacteria in the colon. Acetyl CoA, acetyl coenzyme A.
Electron sink products such as ethanol, lactate, hydrogen, and succinate are produced by some bacteria to regenerate oxidized pyridine nucleotides. These fermentation intermediates are subsequently fermented to SCFAs by other gut bacteria, and are important factors in maintaining species diversity in the ecosystem.
Fate of short-chain fatty acids
Colonic fermentation can be viewed as a way in which the human host can recover part of the energy of malabsorbed carbohydrates. The amount of energy recovered from fermentation depends on the fer-mentability of the carbohydrate (which can range from 0% to 100%) and the nature of the products of fermentation. On a typical Western diet, about 40– 50% of the energy in carbohydrate that enters the colon is available to the human host as SCFAs. The rest of the energy is unavailable to the host, being lost as heat or unfermented carbohydrate or used to produce gases or for bacterial growth (Figure 5.5).
SCFAs are almost completely absorbed and, while some butyrate is oxidized by colonocytes (the epithe-lial cells lining the colon), most arrives at the liver via the portal vein. Propionate and butyrate are removed in first pass through the liver, but increased concen-trations of acetate can be observed in peripheral blood several hours after consumption of indigestible but fermentable carbohydrates. These absorbed
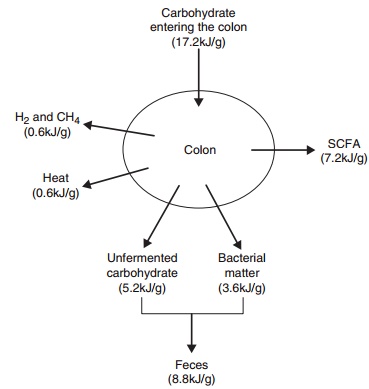
Figure 5.5 Quantitative fate of carbohydrate in the colon. SCFA, short-chain fatty acid.
There is considerable interest in the possible effects of individual SCFAs on the health of the colon and the whole body. The strongest evidence to date is for an anticancer effect of butyrate, which may be due to the ability of butyrate to induce differentiation and apoptosis (programmed cell death) of colon cancer cells. There is some support for the hypothesis that propionate may help to reduce the risk of cardiovas-cular disease by lowering blood cholesterol concen-tration and/or by an effect on hemostasis, but the evidence so far is not conclusive.
Related Topics