Chapter: Introduction to Human Nutrition: Digestion and Metabolism of Carbohydrates
Dietary management of blood glucose concentration
Dietary management of blood glucose concentration
Glycemic index
As an aid to the dietary management of blood glucose concentrations in diabetics, Jenkins and colleagues (1981) introduced the concept of the glycemic index (GI), which provides a means of comparing quanti-tatively the blood glucose responses (determined directly by in vivo experiments) following ingestion of equivalent amounts of digestible carbohydrate from different foods. When a range of carbohydrate-containing foods was ranked according to their GI values, there was a strong linear relationship with the rapidly available glucose (RAG) from similar foods determined in vitro as the sum of free glucose, glucose from sucrose, and glucose released from starches over a 20 minute period of hydrolysis with a battery of enzymes under strictly controlled conditions (Englyst method; Englyst et al. 1999). This offers the possibil-ity of assaying foods in vitro for their RAG content, which will be quicker and cheaper than the current approach based on GI measurements in vivo.
Studies with glucose and starches enriched with the stable isotope carbon-13 have demonstrated that glucose absorption from the gut following a meal con-tinues for several hours after blood concentrations have returned to fasting levels. In this later postpran-dial period, insulin secretion is sufficient to ensure that the rate of glucose absorption is matched by the rate of glucose removal from the circulation. 13C-Labeled substrates are being used increasingly to investigate the kinetics of digestion, absorption, and metabolic disposal of glucose and other sugars from a range of foods. When continued over several years, high rates of glucose absorption and the subsequent challenge to the capacity of the pancreatic β-cells to secrete insulin may be the primary determinants of insulin resistance and eventual pancreatic failure that contribute strongly to the etiology of diabetes and cardiovascular disease. Such kinetic studies are likely to be helpful in identifying foods with slower rates of intestinal hydrolysis – information that can be used in public health advice or in counseling of individuals.
Fructose
When glucose and fructose are available simultane-ously after a meal containing sucrose, how does the body select which fuel to use first for oxidative purposes? This question has been resolved by experi-ments in which volunteers consumed, on two sepa-rate occasions, high-sucrose test meals which were identical except that one or other of the constituent monomeric sugars was 13C-labeled in each meal. The volunteers blew into tubes at intervals after the meals to provide breath samples for measurement of enrich-ment of expired carbon dioxide with 13C. The results showed that, after the high sucrose meal, fructose was oxidized much more rapidly and extensively than was glucose (Figure 5.2).
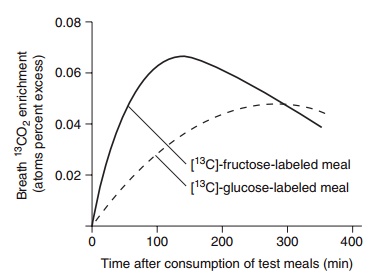
Figure 5.2 Enrichment of breath 13CO2 following ingestion of high-sucrose test meals labeled with [13C]-fructose and [13C]-glucose. (Redrawn from Daly et al., 2000, with permission of the American Society for Nutrition.)
This rapid oxidation of fructose may be explained by the fact that, because it is phosphorylated in hepa-tocytes, it bypasses 6-phosphofructokinase, one of the key regulatory enzymes in glycolysis.
Related Topics