Chapter: Biotechnology Applying the Genetic Revolution: Bacterial Infections
Iron Acquisition by Pathogenic Bacteria
IRON
ACQUISITION BY PATHOGENIC BACTERIA
Almost all bacteria need iron because it
is a cofactor for many enzymes, especially those of the respiratory chain.
However, the concentration of free iron in the body, including the bloodstream,
is kept low by a variety of specialized proteins that bind iron very tightly. Surplus
iron is bound by transferrin and lactoferrin , which act as iron transporters,
or by ferritin , which is an iron storage protein.
Bacteria use iron chelators, known as
siderophores , to bind iron and, if necessary, extract it from host proteins. Siderophores
are excreted by the bacteria, bind iron, and are then taken back into the
bacterial cell by specialized transport systems. Most bacteria have a variety
of suchiron transport systems for use under different conditions. Enterochelin
(or enterobactin) is a cyclic trimer of 2,3-dihydroxybenzoylserine (DHBS) and
is perhaps the best known siderophore. It is made by E. coli and many enteric
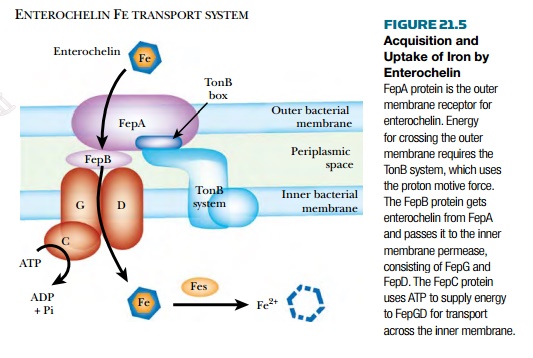
bacteria. The Fep transport system ( Fig. 21.5 ) conveys the enterochelin/Fe complex
across both outer and inner membranes. Enterochelin binds iron so tightlythat
once inside the bacterial cell it must be destroyed in order to release the
iron. Fes protein is an esterasethat hydrolyzes Fe 3+ -enterochelin into DHBS
monomers, releasing the iron. Despite this, enterochelin does not bind iron
well enough to extract it from transferrin, the major iron binding protein of
blood.
Pathogenic bacteria often possess more
potent siderophores (plus their receptor systems) that are capable of
extracting iron from transferrin. Two examples are mycobactin, made by Mycobacterium
tuberculosis , and yersiniabactin, named after Yersinia pestis , the
plague bacterium, in which it was first discovered. Yersiniabactin is
widespread among pathogenic bacteria of the enteric family, and in Yersinia itself
the genes for synthesis and uptake are clustered on a mobile pathogenicity
island. Other virulent enteric bacteria carry plasmidborne genes for hemolysin
, a toxin that lyses red blood cells, thus allowing access to the hemoglobin.
Related Topics