Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Communities, ecosystems, and the functional role of fishes
Influence of physical factors and disturbance - Diversity of Fishes
Influence of physical factors and disturbance
Ecosystem ecology is concerned with biological interactions but also with the effects of physical and climatic factors on ecosystem components. Often these effects are most obvious when extreme climatic or other disturbances occur. The structure of a community, broadly defined to include species composition, abundance, distribution, and ecological interactions, changes in response to variation in climate and other forms of disturbance. Disturbance can lead to shortterm changes in the physiology, behavior, or ecology of individuals (e.g., acclimatization, movement, trophic and reproductive adjustments), which result in alterations in community structure, which in turn affect the pathways and rates at which energy and nutrients flow through an ecosystem. Abiotic factors that function as disturbances to fishes and cause alterations in community structure and ecosystem function include but are not limited to reductions in dissolved oxygen, often in concert with increased water temperature; changes in stream and river discharge as the result of storms, floods, dams, and drought; and cyclonic storms in coral and kelpbed habitats. Biological disturbances that have ecosystem-wide repercussions include outbreaks of disease or population explosions of destructive species that affect the food and habitat resources of a system (Karr & Freemark 1985; Karr et al. 1992).
Temperature, oxygen, and water flow
Water holds relatively little oxygen, seldom more than 8 ppm, and levels below 1 ppm are generally fatal to fishes (see Water as a respiratory environment). Hence periods of deoxygenation – due to excessive decomposition of plants and animals, a concentration of fishes trapped in pools during drought or following floods, high summer temperatures leading to thermal and oxygen stratifi cation in lakes, or ice cover – are natural events that strongly affect the distribution and survival of fishes. Species with narrow ranges of tolerance for temperature and oxygen variation will obviously be most strongly affected by extreme conditions. Adult Striped Bass, Morone saxatilis, prefer temperatures between 18 and 25°C and oxygen concentrations above 3 ppm. In southern US rivers such as the St. John’s in Florida and Flint in Georgia, Striped Bass in summer avoid high water temperatures in the main river and aggregate near inflowing springs that provide cooler, preferred temperatures. By the end of summer, these narrow thermal tolerances result in emaciated fish because of limited feeding opportunities in the springs. Thermal stratifi cation in large reservoirs during the summer can also squeeze stripers into an increasingly small region near the thermocline to the point where mortality occurs if the fish are forced into the relatively deoxygenated but cooler waters of the hypolimnion (Coutant 1985; Van Den Avyle & Evans 1990; Thermal preference).
Extreme water flow can constrain fishes in streams and rivers. Small fishes and small species typically cannot hold position in swift water as readily as can larger individuals. Eggs and larvae may be washed out of a system or covered with silt if high flow conditions occur during the breeding season. Juveniles of quiet water species (e.g., many minnows, sunfishes) are frequently flushed out of headwaters during flood conditions (Schlosser 1985; Harvey 1987). Adult stream fishes can also be adversely affected if floods fill pools or riffl es with debris (Minckley & Meffe 1987). Upland streams are, however, characterized by recurrent and dramatic fluctuations in flow regime and the fish faunal composition of such areas can return to its original state, probably via recolonization from downstream, within 8 months following even catastrophic floods (Matthews 1986b). In contrast, flooding further downstream is often an important signal inducing spawning in many river fishes because this is the time when riparian zones and gallery forests become inundated and create nursery habitats for juveniles (Welcomme 1985; Lowe-McConnell 1987; Reproductive seasonality).
Many species, including relatively small fishes, are of course well adapted to high flow conditions and have little difficulty maintaining position in surf zones (clingfishes, gobies) or swift flowing water (e.g., homalopterid hillstream loaches, Colorado River minnows and suckers, many darters, torrent fishes of New Zealand). Marked differences in adaptation to intermittent and extreme flow can occur between species within a family. The Sonoran Topminnow (Poeciliopsis occidentalis) and the Mosquitofish (Gambusia affinis) are both small, morphologically similar members of the livebearer family, Poeciliidae. The topminnow has evolved in desert streams of the American southwest that periodically experience flash floods. Mosquitofish are native to southeastern US lowlands where rapidly flowing water seldom occurs. Mosquitofish have been widely introduced into southwestern regions and prey on the young of topminnows, leading to population declines of the desert species. But the speed with which Mosquitofish eliminate the desert native is largely dependent on the extent of flash flooding. In locales where floods occur regularly, Mosquitofish are less successful because they lack the behavioral avoidance and orientation capabilities that topminnows display when flow rate increases. The introduced species gets flushed out of the system because of its inappropriate response to floods (Meffe 1984). The importance of maintaining natural flow regimes in preventing invasions of alien species has been shown repeatedly in studies of the fish fauna of California (e.g., Marchetti & Moyle 2001).
The opposite conditions of low flow lead to isolated habitats, desiccation, and deoxygenation. Upland fishes fre quently move downstream and floodplain fishes move into the main river as water levels decline. Isolated pools lead to an increased rate of ecological interactions as fishes crowd together. Competition may be reduced if species diverge in their resource use in response to dwindling resource availability. Seasonal shifts of just this type have been observed among Panamanian stream fishes, where the least diet overlap among species occurs during the dry season when resources are least abundant (Zaret & Rand 1971).
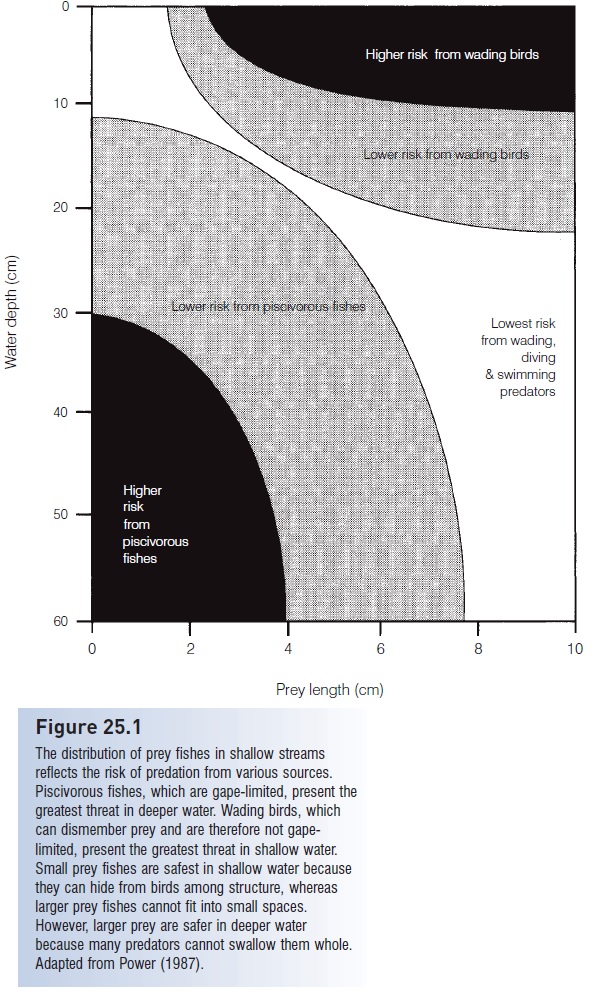
Seasonal cycles of changing water levels can lead to complex interactions among community components. The Everglades region of southern Florida experiences seasonally fluctuating water levels that usually include a dry season in the spring. This dry period concentrates fishes in relatively small pools (alligator holes) where they are preyed upon heavily by herons, ibises, and storks. The birds are dependent on the fishes for successful reproduction and eat 76% of the fishes in the pools. Although population sizes of the fishes are reduced, species diversity is highest during low water, with small species of omnivores and herbivores (mostly livebearers and topminnows) dominating. If water levels are high, the fish can escape bird predation (see Fig. 25.1) and bird populations decline. Relaxed bird predation leads to overcrowding if water levels then drop, resulting in 96% fish mortality from deoxygenation and also from predation by piscivorous fishes (bullhead catfish, Largemouth Bass, sunfishes). Overall fish diversity also declines as the predators eat the omnivores and herbivores. Hence drought conditions in the presence of predatory birds are benefi cial to small fish species, but in the absence of wading birds are benefi cial to larger, predatory fishes (Kushlan 1976, 1979; Karr & Freemark 1985).
Extreme weather
Extremes of climate act as disturbances in the marine environment also, although the effects appear to differ between sites and storms. Major tropical (cyclonic) storms can generate winds in excess of 200 km/h, creating waves more than 12 m high that break on relatively shallow coral reef environments. The influence of such waves is felt far below the surface, as massive corals are broken off and tossed around at depths exceeding 15 m and tremendous amounts of sand shift, are suspended in the water column, and scour most structure in the area. After a major cyclonic storm (such storms are generally referred to as hurricanes in the Atlantic and eastern Pacific, as typhoons in the northwestern Pacific, and as cyclones in the southwestern Pacific), few live corals remain in shallow water and major destruction can be found down to depths of 30–50 m depending on the nature and direction of the storm, tide stage when it struck, and bottom topography. When Hurricane Allen struck Jamaica’s north coast in 1980, much of the shallow water coral and other structure was destroyed or damaged.
Damselfish algal lawns were eliminated and the damselfish wandered around for over a week without displaying their usual territoriality. The territories they eventually set up were in deeper water and were associated with different coral types than were used before the hurricane. Parrotfishes formed smaller schools and stopped reproducing for 2 weeks. Normally cryptic and nocturnal species (moray eels, squirrelfishes, hawkfish, blennies) swam out in the open by day, perhaps because their refuges were destroyed. Planktivorous fishes (damselfishes, wrasses, bogas) hugged the reef rather than foraging high in the water column. Large predators (snappers, groupers, grunts) that were previously rare increased in number and swam conspicuously in the open, perhaps capitalizing on displaced and confused prey species. One year after the storm, species distributions and densities remained different, having shifted in favor of fishes associated with low relief habitats (e.g., rubble vs. upright coral). Analysis of coral recovery 6 years after the storm indicated that damselfish caused a decrease in the numbers and sizes of colonies of the dominant coral species (Woodley et al. 1981; Kaufman 1983; Knowlton et al. 1990).
Even moderate storms can have strong effects on reef fishes. A series of three relatively mild (sustained winds of 60 km/h) cyclones struck the northern Great Barrier Reef over a 2-year period. These storms caused little structural damage to corals but had major behavioral and community effects on the fishes (Lassig 1983). Suspended sand, moved by strong surge and currents, forced many otherwise benthic fishes up into the water column and caused visible wounds, apparently from collisions with corals. More importantly, juveniles suffered substantial population losses and subadults were redistributed; 60% of the species surveyed suffered density losses of juveniles following one storm. Poor recruitment in several species was attributed to injury or, more likely, settling juveniles being flushed from the system by strong currents. Hence periodic storms can play a decisive role in the structure of some reef communities.
Analogous events follow storms in temperate marine habitats. A series of severe winter storms occurred off California in 1980, destroying much of the canopy of giant kelp in coastal kelp beds and scouring the bottom. Removal of
the kelp eliminated the major food of sea urchins, which switched to a diet of benthic algae and denuded the understory regions of reefs, “transforming the reef from a richly forested site to a barren area” (Stouder 1987, p. 74). The understory turf harbored invertebrates that were the major food types of the abundant, resident surfperches. Differences in microhabitat use and feeding patterns among surfperch species decreased as fish converged on the few areas where prey remained. Although adult surfperches remained in reef areas over the next 15 months, overall fish abundance decreased by 50% as nonresident and subadult fishes abandoned the reefs, probably because of loss of food and refuge sites and unsuccessful competition with competitively superior, resident surfperches (Ebeling et al. 1985; Stouder 1987).
Not all storms, even major storms, have such clearcut effects on diversity and density of reef fishes. A severe 3-day storm struck the Kona coast of Hawaii in 1980, destroying most of the shallow water coral. Few direct fish mortalities or injuries occurred. Many shallow water species initially fled to deeper water, but returned to former areas after a few weeks or months. Sixteen months after the storm, diversity and density had returned to or surpassed prestorm levels, with the exception of a few distributional shifts involving species that remained in deeper water (Walsh 1983).
Reduction in diversity and density is reversible if the causative agents do not recur frequently. The Florida Keys were exposed to record cold temperatures in the winter of 1977 that caused an extensive fish kill. Water temperatures fell to 11°C in areas that normally do not drop much below 19°C. Dead and dying reef fishes were found throughout the area; underwater censuses showed significant decreases in both species diversity and individual densities the following summer. However, by the next year, both overall diversity and density were not different from their pre-cold snap levels; in fact, diversity on some reefs was higher than before. These increases were largely the result of successful recruitment of many new individuals, perhaps because potential competitors and predators were eliminated by the cold weather (Bohnsack 1983; see also Thomson & Lehner 1976).
Biological analogs of extreme weather
Violent storms and sudden water temperature shifts create obvious and rapid changes in the physical environment of a habitat. Fishes can experience analogous disruptive effects as a result of the action of biological processes. Major disturbance events include population explosions of animals that affect the physical structure of a habitat. One such example is the crown-of-thorns starfish, Acanthaster planci, a predator on live corals through much of the Indo-West Pacific Ocean. Normally, the starfish occurs at low densities (2–3 animals/km2), but periodically and for reasons that remain mysterious, the starfish undergoes population explosions producing densities of several starfish per square meter, with thousands of individuals swarming over a reef. The starfish consumes the live tissue on the surface of the corals; the underlying limestone skeleton first becomes covered with algae but then collapses due to biological and physical erosion. In this manner, 95% of the coral in a large area may be killed and will require 10–20 years to recover (Endean 1973; Wilkinson & Macintyre 1992). Fishes that are directly or indirectly dependent on corals, either for food or shelter, suffer as a result. Coral-feeding fishes, including butterfl yfishes, parrotfishes, gobies, wrasses, and triggerfishes, disappear from affected areas, leading to a 15–35% decline in species diversity on affected reefs. Densities of other coral-dependent fishes (many cardinalfishes, damselfishes, wrasses, gobies, blennies) also decline, leading to an overall reduction in fish density of 55–65% in an area (Sano et al. 1984).
Climate change and fishes
All of the external forces acting on fishes – temperature extremes, oxygen availability, floods, droughts, cyclonic storms, habitat loss – are influenced by climate, and there is little doubt that we have entered a period of humaninduced climate disruption. The authoritative Intergovernmental Panel on Climate Change (IPCC), in its Fourth Assessment, concluded that climate change has happened and is happening, that it is largely influenced by human activity, that unprecedented warming and sea level rise are occurring (“warming of the climate system is unequivocal”), and that the consequences for humanity and the rest of earth’s biota are serious (see IPCC 2007a, 2007b, 2007c, 2007d).
The effects that such unnatural change will have on fishes is in part speculative, but the speculation is scientifically based, and evidence of verifiable impacts grows as we learn where to look. Better studied, more accessible taxonomic groups – birds, mammals, insects, mollusks, plants, phytoplankton, zooplankton – are showing changes in distribution, abundance, physiological performance, and reproductive and migrational timing that are directly linked to documented climatic shifts (Walther et al. 2002; Ahas et al. 2006). “More than 80% of the species that show changes are shifting in the direction expected on the basis of known physiological constraints of species” (Root et al. 2003). Fishes can be expected to respond in similar ways, and the responses will affect all levels of ecological organization, from the genetics of populations, the interactions between fishes and other community components, as well as the roles that fishes play within ecosystems.
Related Topics