Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Communities, ecosystems, and the functional role of fishes
Community-level interactions between fishes and other taxonomic groups
Community-level interactions between fishes and other taxonomic groups
Fishes do not restrict their ecological interactions – chiefly competition, predation, parasitism, and symbiosis – to other fishes. Evolved, fitness enhancing or reducing interactions are also common between fishes and other taxonomic groups, ranging from plants and invertebrates to other vertebrates. Often these interactions and their influences are relatively direct, e.g., through eating and being eaten. But some dramatic effects – such as changes in habitat use, food types, life history traits – take place through fairly indirect means, either incidental to or several steps removed from the activities of the fishes involved.
Competition
Fishes compete with a variety of organisms for food and space. Bluefish (Pomatomus saltatrix) and common terns (Sterna hirundo) have a complex feeding relationship that involves both commensalism and competition and that may be applicable to many other fish–bird interactions (Safina & Burger 1985). Both species feed on anchovies and Sand Lances. The seabirds are particularly dependent on prey fish during the early summer breeding season when they must meet their own energetic demands as well as those of their growing chicks. Off Long Island, New York, Bluefish arrive in large numbers around mid-July each year as part of their annual migration (see Oceanodromy). A commensal relationship exists between the Bluefish and terns in that the feeding activities of Bluefish drive prey up in the water column, concentrate them in space, and indicate the whereabouts of prey to the seabirds, all of which facilitate prey capture by the terns. However, newly arrived Bluefish consume large numbers of prey fish, which rapidly depresses the prey resource. Any birds that initiate breeding after the arrival of the Bluefish tend to be unsuccessful. Hence Bluefish may be significant competitors with terns and may have been a strong selective force in determining the timing of reproduction by the terns.
Predation
A wide variety of non-piscine predators are dependent on fishes as a major component of their diets. Numerous invertebrate predators capture fishes. Predaceous waterbugs (Belostomatidae) and dragonfly (Odonata) larvae prey on small freshwater fishes. Marine invertebrate predators are common, including jellyfish, anemones, siphonophores, squid, dwarf triton and cone snails, and crabs (e.g., Laughlin 1982; Bouchet & Perrine 1996). Among vertebrates, reptilian predators include turtles, crocodilians, varanid monitor lizards, a few iguanid lizards, and sea and water snakes; mammals include mink, raccoons, otters, seals, sea lions, bears, dolphins, whales, bats, and, of course, humans. Amphibian predation on fishes is poorly documented, although sirens, bullfrogs, and a few other large frogs (Pipa, Xenopus) are known to be fish predators, including the impacts of introduced species on endangered fishes (e.g., Lafferty). A host of seabirds concentrate on fishes, including terns, petrels, albatrosses, gannets, auks, murres, cormorants, skimmers, spoonbills, pelicans, penguins, and gulls. In fresh waters, osprey, eagles, loons, mergansers, goldeneye ducks, kingfishers, herons, egrets, and storks are a few of the birds that eat fish, often to the birds’ detriment when the fish are in aquaculture facilities and “predator control” is put into practice. All of these non-fish groups have served as selective agents on the behavior and ecology of fishes, causing evolutionary adjustments in growth and reproductive traits that have allowed fish to thrive despite sometimes astronomical mortality rates. Many fishery-related problems arise from “unnatural” predation by humans and the evolutionary responses of fishes to such predation (see Fishing as an evolutionary force).
The impact of non-fish predators on fish populations and behavior can be substantial. In the Au Sable River, Michigan, mortality of adult Brook and Brown trout averaged 70–90% annually, most of which was from predation. Non-fish and non-human predators (mergansers, heron, kingfisher, mink, otter) accounted for 28–35% of this mortality (Alexander 1979). When the potential threat of predatory birds is combined with that from piscivorous fishes, it is not surprising that the distribution of many prey species reflects the foraging locales of their predators (Fig. 25.1). This combined threat can include a third dimension during the breeding season. Male Dollar Sunfishes (Lepomis marginatus) construct nests in shallow water to avoid predatory fishes that are typically in deeper water. The males, however, must repeatedly abandon their nests to avoid being captured by herons and kingfishers. Each time the male flees, eggs and young in the nest are subject to predation by small fishes, forcing the male into a trade-off between the conflicting demands of protecting himself and protecting his offspring (Winkelman 1996).
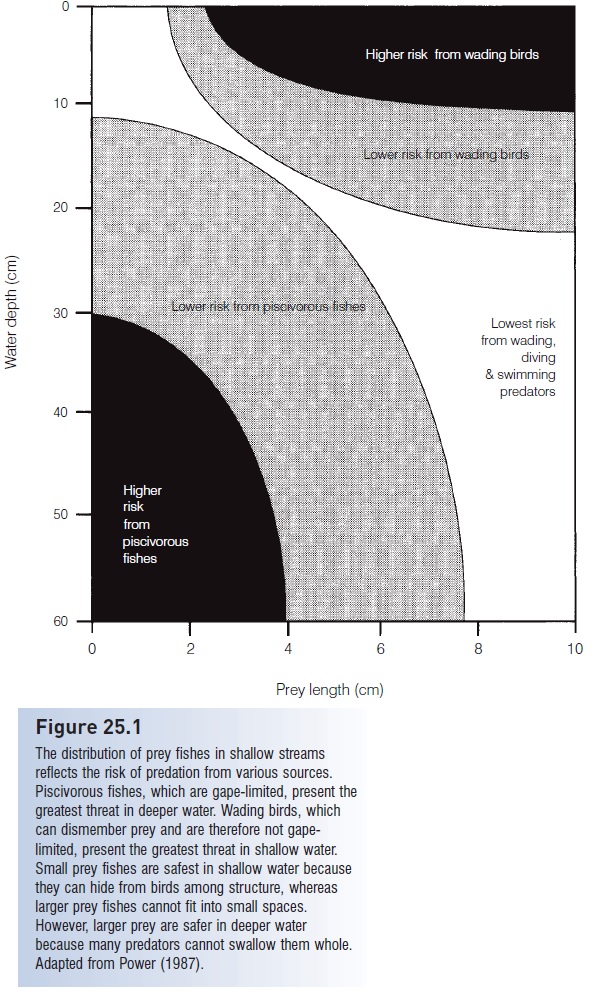
Figure 25.1
The distribution of prey fishes in shallow streams reflects the risk of predation from various sources. Piscivorous fishes, which are gape-limited, present the greatest threat in deeper water. Wading birds, which can dismember prey and are therefore not gapelimited, present the greatest threat in shallow water. Small prey fishes are safest in shallow water because they can hide from birds among structure, whereas larger prey fishes cannot fit into small spaces. However, larger prey are safer in deeper water because many predators cannot swallow them whole. Adapted from Power (1987).
Fishes also fall prey to less obvious but more insidious predators. Massive fish kills have long been attributed to harmful algal blooms (HABs) and especially dinofl agellate blooms (“red tides”) in many nearshore marine areas, but fish death has usually been considered an incidental byproduct of a bloom, via such effects as insuffi cient oxygen concentrations or the toxicity of secondary plant chemicals.
More recent events implicate an evolved, predatory response involving a dinofl agellate, Pfi esteria piscicida, and closely related forms, that has a life cycle of 20 or more flagellated, amoeboid, and cyst-like stages depending on environmental conditions, including the presence or absence of fish and fish byproducts in the water (Barker 1997; Burkholder & Glasgow 2001; Burkholder 2002). Resting cysts of the dinofl agellate are stimulated to break open in the presence of chemicals exuded by fish. The vegetative cells released from the cysts produce a neurotoxin and other substances that can kill fish in a matter of hours. The toxin induces immobility and death in fish; later-produced amoeboid cells and zoospores feed on the moribund and dead fish. Many dead and dying fish associated with Pfi esteria have ulcerated lesions (Fig. 25.2, inset). Skin sloughs off the dying fish and is attacked by the dinofl agellates, which then reproduce rapidly, leading to a massive fish kill; 1 billion Menhaden died during an episode in North Carolina’s Neuse River estuary in 1991.
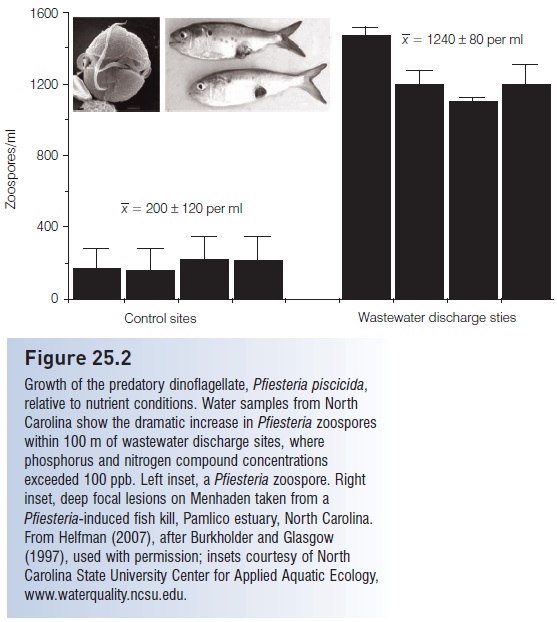
Figure 25.2
Growth of the predatory dinoflagellate, Pfiesteria piscicida, relative to nutrient conditions. Water samples from North Carolina show the dramatic increase in Pfiesteria zoospores within 100 m of wastewater discharge sites, where phosphorus and nitrogen compound concentrations exceeded 100 ppb.
When a fish population declines to some level where the cyst-breaking chemical trigger is no longer suffi ciently concentrated, the dinofl agellates return to the encysted form. Hence density-dependent population regulation of fishes could occur through the population responses of a microscopic predator. Increasing frequencies of Pfi esteria-caused fish kills correspond to increasing concentrations of human and agricultural waste-water (Fig. 25.2). One fish kill in the Neuse River estuary, North Carolina, in 1995 followed shortly on the heels of a major swine effl uent spill, during which approximately 1 x108 L of raw hog sewage was discharged from a ruptured, upstream, sewage lagoon (Burkholder & Glasgow 2001). Not only fish can be affected by P. piscicida; lesions and other pathological symptoms – including blurred vision, erratic heartbeat, and memory loss – can also occur in humans.
Parasitism
The previous example points out the difficulty of distinguishing predators, which consume most of their prey, from parasites, which consume only a small portion of their prey. All fishes have parasites, external and internal, with a great degree of host specificity (e.g., Bush et al. 2001; Combes 2001). Life cycles are complex and attachment sites are diverse, as in the 6 cm long copepod Ommatokoita elongata that anchors itself in the corneas of 6 m long Greenland Sleeper Sharks, Somniosus microcephalus, commonly resulting in blindness (Borucinska et al. 1998). Parasite–host relationships often involve co-evolved responses (see Interspecific relations: symbioses). One particularly bizarre relationship exists between fishes and parasitic isopods in the family Cymothoidae. These isopods are frequently observed on the heads and in the gills of numerous reef fishes. However, under some circumstances, the isopod attaches to the tongue of its host, causing the tongue to degenerate to a small stub (either through direct consumption or constriction of blood supply). Fish tongues lack skeletal musculature and are not protrusible, unlike in other vertebrates. Instead they fill the lower part of the mouth, covering the basibranchial and basihyal bones and serve the mechanical function of holding food against the vomerine and palatine teeth during processing. When the isopod Cymothoa exigua attaches to and destroys the tongue of the Spotted Rose Snapper (Lutjanus guttatus), it resembles the shape and size of the fish’s tongue and occludes with the vomerine teeth when the fish feeds. Snappers with the isopod in place for extended periods are typically in good condition, with full stomachs and accumulated fat, which is often not the case for fish that have cymothoid isopods attached to their gills. In essence, the
parasite functions as a prosthetic replacement tongue, allowing its host to survive normally and hence continue to provide the parasite the nourishment it needs for its own successful reproduction (Brusca & Gilligan 1983).
In the context of community relationships, parasitism can have profound effects on relationships among species,
In the lab, Prussian Carp, Carassius auratus, infected with metacercariae of the digeneanPosthodiplostomum cuticola, were eaten more often by Perch, Perca fluviatilis, compared to nonparasitized carp, probably because parasitized individuals were in poorer conditionand were visually conspicuous due to the presence of black spots associated with digenean infections (Ondrackova et al. 2006). Parasitism can also interact with availability of and competition for refuges to cause higher mortality in infected fish. When Bridled Goby, Coryphopterus glaucofraenum, were experimentally infected in the field with a copepod gill parasite, goby survival declined as a combined function of goby density, parasite occurrence, and refuge availability (Forrester & Finley 2006). The decline was steeper for parasitized gobies in reef habitats with fewer refuges than for unparasitized gobies in similar circumstances. Parasitism and a shortage of refuges jointly influenced the strength of density-dependent mortality in this reef fish, an interesting finding given that nocturnal refuges may be important in minimizing infection by other, nocturnally attacking fish parasites (e.g., Sikkel et al. 2006).
Parasites that infect fishes alter behavior (Barber et al. 2000). Some behavioral changes can facilitate transmission of the parasite to its final host. Killifish, Fundulus parvipinnis, are an intermediate host for trematodes. Killifish infected by larval trematodes alter their swimming behavior in ways that make them more conspicuous to bird predators, their definitive host. Parasitized fish in the field were 10–30 times more likely to be eaten than unparasitized fish (Lafferty & Morris 1996). Humans may be the ultimate host. The human diseases of schistosomiasis and opisthorchiasis are transmitted via fishes as intermediate hosts. Snails are an initial intermediate host for schistosomes. The snails are eaten by fishes, such as molluskivorous cichlids in Africa’s great lakes, which are then eaten by humans (Evers 2006). Numerous cyprinids in Thai waters feed on snails that harbor the trematode metacercariae larvae of the liver fluke that causes opisthorchiasis, the most prevalent food-borne parasitic disease in Thailand (Kumchoo et al. 2005).
Parasites have gone to great lengths to find intermediate hosts, including capitalizing on symbiotic relationships among fish species. Cleaning behavior is a highly evolved mutualism on coral reefs, performed by juveniles of some species and best known among adult wrasses, particularly in the cleaning-specialist genus Labroides (see Mutualism and commensalism). Digenean bucephalid trematodes have a complex life cycle usually involving at least a snail and a snailating fish as intermediate hosts, with a piscivorous fish as the definitive host. Adult bucephalids generally only occur in piscivores, the larvae having been ingested incidentally as a consequence of feeding on fish prey. However, recent surveys of labrids have shown that cleaner wrasses, especially Labroides spp., have incidences of bucephalid infection of 50–100%. The likely route of transmission is direct, the cleaners having ingested bucephalid larvae in the process of cleaning parasites from host fishes (Jones et al. 2004). The parasite has evolved a new route for transmission, exploiting the host–cleaner relationship in cleaning symbiosis.
Related Topics