Chapter: 10th Science : Chapter 9 : Solutions
Hydrated salts and Water of Crystallization
Hydrated
salts and Water of Crystallization
When ionic substances
are dissolved in water to make their saturated aqueous solution, their ions
attract water molecules which then attached chemically in certain ratio. This
process is called hydration. These ionic substances crystallize out from their
saturated aqueous solution with a definite number of molecules of water. The
number of water molecules found in the crystalline substance is called water
of crystallization. Such salts are called hydrated salts.
On heating these
hydrated crystalline salts, they lose their water of crystallization and become
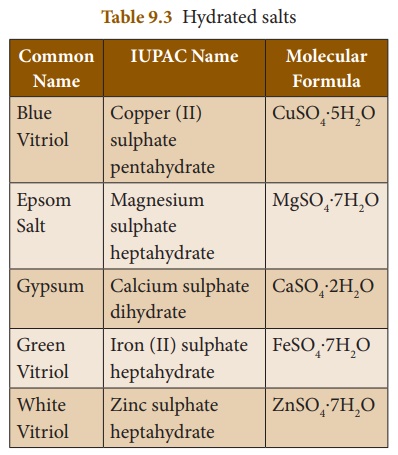
amorphous or lose their colour (if they are coloured). Table 9.3 shows some
common hydrated salts:
1. Copper sulphate pentahydrate CuSO4.5H2O (Blue vitriol)
The number of water
molecules in blue vitriol is five. So its water of crystallization is 5. When
blue coloured copper sulphate crystals are gently heated, it loses its five
water molecules and becomes colourless anhydrous copper sulphate.

If you add few drops of
water or allow it to cool, the colourless anhydrous salt again turns back into
blue coloured hydrated salt.

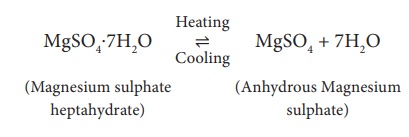
2. Magnesium sulphate heptahydrate MgSO4.7H2O (Epsom salt)
Its water of
crystallization is 7. When magnesium sulphate heptahydrate crystals are gently
heated, it loses seven water molecules, and becomes anhydrous magnesium
sulphate.
If you add few drops of
water or allow it to cool, the colourless anhydrous salt again turns back into
hydrated salt.

Related Topics