Chapter: 10th Science : Chapter 9 : Solutions
Factors affecting solubility
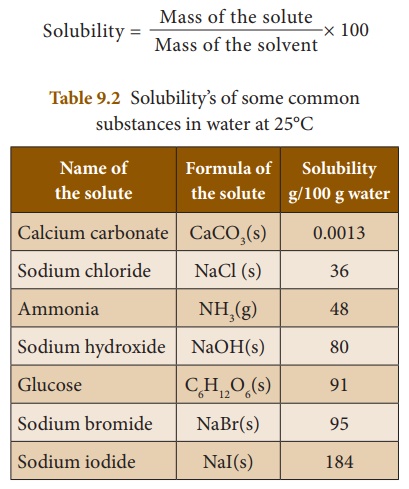
Solubility
Usually, there is a
limit to the amount of solute that can be dissolved in a given amount of
solvent at a given temperature. When this limit is reached, we have a saturated
solution and any excess solute that is added, simply resides at the bottom of
the solution. The extent of dissolution of a solute in a solvent can be better
explained by its solubility. Solubility is measure of how much of a solute can
be dissolved in a specified amount of a solvent.
Solubility is defined as the number
of grams of a solute that can be dissolved in 100 g of a solvent to form
its saturated solution at a given temperature and pressure. For example, 36 g
of sodium chloride need to be dissolved in 100 g of water to form its saturated
solution at 25°C. Thus the solubility of NaCl in water is 36 g at 25°C. The
solubility is mathematically expressed as
Factors
affecting solubility
There are three main
factors which govern the solubility of a solute. They are:
(i) Nature of the solute and solvent
(ii) Temperature
(iii) Pressure
(i) Nature of the solute and solvent
The nature of the solute
and solvent plays an important role in solubility. Although water dissolves an
enormous variety of substances, both ionic and covalent, it does not dissolve
everything. The phrase that scientists often use when predicting solubility is
“like dissolves like.” This expression means that dissolving occurs when
similarities exist between the solvent and the solute. For example: Common salt
is a polar compound and dissolves readily in polar solvent like water.
Non-polar compounds are
soluble in non-polar solvents. For example, Fat dissolved in ether. But
non-polar compounds, do not dissolve in polar solvents; polar compounds do not
dissolve in non-polar solvents.
(ii) Effect of Temperature
Solubility of Solids in
Liquid:
Generally, solubility of
a solid solute in a liquid solvent increases with increase in temperature. For
example, a greater amount of sugar will dissolve in warm water than in cold
water.
In endothermic process,
solubility increases with increase in temperature.
In exothermic process,
solubility decreases with increase in temperature.
Solubility of Gases in
liquid
Do you know why is it
bubbling when water is boiled? Solubility of gases in liquid decrease with
increase in temperature. Generally, water contains dissolved oxygen. When water
is boiled, the solubility of oxygen in water decreases, so oxygen escapes in
the form of bubbles.
Aquatic animals live
more in cold regions because, more amount of dissolved oxygen is present in the
water of cold regions. This shows that the solubility of oxygen in water is
more at low temperatures.
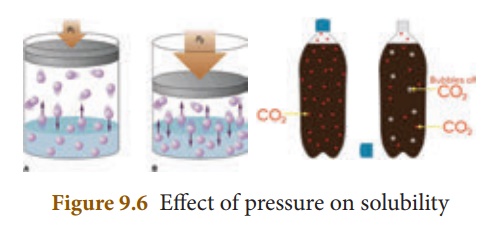
(iii) Effect of Pressure
Effect of pressure is
observed only in the case of solubility of a gas in a liquid. When the pressure
is increased, the solubility of a gas in liquid increases.
The common examples for
solubility of gases in liquids are carbonated beverages, i.e. soft drinks,
household cleaners containing aqueous solution of ammonia, formalin- aqueous
solution of formaldehyde, etc.
Related Topics