Chapter: Medical Physiology: Pregnancy and Lactation
Hormonal Factors in Pregnancy
Hormonal Factors in Pregnancy
In pregnancy, the placenta forms especially large quantities of human chorionic gonadotropin, estrogens,progesterone, and human chorionic somatomam-motropin, the first three of which, and probablythe fourth as well, are all essential to a normal pregnancy.
Human Chorionic Gonadotropin and Its Effect to Cause Persistence of the Corpus Luteum and to Prevent Menstruation
Menstruation normally occurs in a nonpregnant woman about 14 days after ovulation, at which time most of the endometrium of the uterus sloughs away from the uterine wall and is expelled to the exterior. If this should happen after an ovum has implanted, the pregnancy would terminate. However, this is prevented by the secretion of human chorionic gonadotropin by the newly developing embryonic tissues in the following manner.
Coincidental with the development of the tro-phoblast cells from the early fertilized ovum, the hormone human chorionic gonadotropin is secreted by the syncytial trophoblast cells into the fluids of the mother, as shown in Figure 82–7. The secretion of this hormone can first be measured in the blood 8 to 9 days after ovulation, shortly after the blastocyst implants in the endometrium. Then the rate of secretion rises rapidly to reach a maximum at about 10 to 12 weeks of pregnancy and decreases back to a lower value by 16 to 20 weeks. It continues at this level for the remain-der of pregnancy.
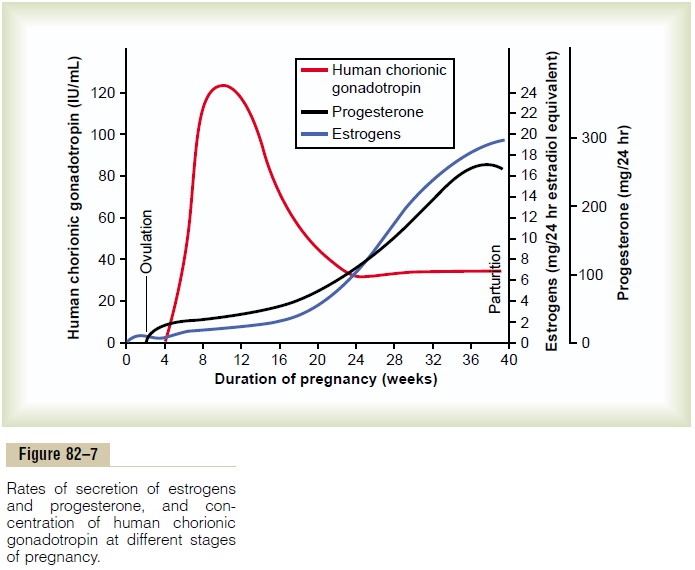
Function of Human Chorionic Gonadotropin. Humanchorionic gonadotropin is a glycoprotein having a molecular weight of about 39,000 and much the same molecular structure and function as luteinizing hormone secreted by the pituitary gland. By far, its most important function is to prevent involution of the corpus luteum at the end of the monthly female sexual cycle. Instead, it causes the corpus luteum to secrete even larger quantities of its sex hormones—proges-terone and estrogens—for the next few months. These sex hormones prevent menstruation and cause the endometrium to continue to grow and store large amounts of nutrients rather than being shed in the menstruum. As a result, the decidua-like cells that develop in the endometrium during the normal female sexual cycle become actual decidual cells—greatly swollen and nutritious—at about the time that the blastocyst implants.
Under the influence of human chorionic gonadotropin, the corpus luteum in the mother’s ovary grows to about twice its initial size by a month or so after pregnancy begins, and its continued secretion of estrogens and progesterone maintains the decidual nature of the uterine endometrium, which is necessary for the early development of the fetus.
If the corpus luteum is removed before approxi-mately the 7th week of pregnancy, spontaneous abortion almost always occurs, sometimes even up to the 12th week. After that time, the placenta secretes sufficient quantities of progesterone and estrogens to maintain pregnancy for the remainder of the gestation period. The corpus luteum involutes slowly after the 13th to 17th week of gestation.
Effect of Human Chorionic Gonadotropin on the Fetal Testes. Human chorionic gonadotropin also exerts aninterstitial cell–stimulating effect on the testes of themale fetus, resulting in the production of testosterone in male fetuses until the time of birth. This small secre-tion of testosterone during gestation is what causes the fetus to grow male sex organs instead of female organs. Near the end of pregnancy, the testosterone secreted by the fetal testes also causes the testes to descend into the scrotum.
Secretion of Estrogens by the Placenta
The placenta, like the corpus luteum, secretes both estrogens and progesterone. Histochemical and physiological studies show that these two hormones, like most other placental hormones, are secreted by the syncytial trophoblast cells of the placenta.
Figure 82–7 shows that toward the end of pregnancy, the daily production of placental estrogens in-creases to about 30 times the mother’s normal level of production. However, the secretion of estrogens by the placenta is quite different from secretion by the ovaries. Most important, the estrogens secreted by the placenta are not synthesized de novo from basic substrates in the placenta. Instead, they are formed almost entirely from androgenic steroid compounds, dehydroepiandrosterone and 16-hydroxydehydroepiandrosterone, which are formedboth in the mother’s adrenal glands and in the adrenal glands of the fetus. These weak androgens are trans-ported by the blood to the placenta and converted by the trophoblast cells into estradiol, estrone, and estriol. (The cortices of the fetal adrenal glands are extremely large, and about 80 per cent consists of a so-called fetal zone, the primary function of which seems tobe to secrete dehydroepiandrosterone during pregnancy.)
Function of Estrogen in Pregnancy. In the discussions of estrogens, we pointed out that these hor-mones exert mainly a proliferative function on most reproductive and associated organs of the mother. During pregnancy, the extreme quantities of estrogens cause (1) enlargement of the mother’s uterus, (2) enlargement of the mother’s breasts and growth of the breast ductal structure, and (3) enlargement of the mother’s female external genitalia.
The estrogens also relax the pelvic ligaments of the mother, so that the sacroiliac joints become relatively limber and the symphysis pubis becomes elastic. These changes allow easier passage of the fetus through the birth canal. There is much reason to believe that estrogens also affect many general aspects of fetal development during pregnancy, for example, by affecting the rate of cell reproduction in the early embryo.
Secretion of Progesterone by the Placenta
Progesterone is also essential for a successful pregnancy—in fact, it is just as important as estrogen. In addition to being secreted in moderate quantities by the corpus luteum at the beginning of pregnancy, it is secreted later in tremendous quantities by the placenta, averaging about a 10-fold increase during the course of pregnancy, as shown in Figure 82–7.
The special effects of progesterone that are essen-tial for the normal progression of pregnancy are as follows:
a) Progesterone causes decidual cells to develop in the uterine endometrium, and these cells play an important role in the nutrition of the early embryo.
b) Progesterone decreases the contractility of the pregnant uterus, thus preventing uterine contractions from causing spontaneous abortion.
c) Progesterone contributes to the development of the conceptus even before implantation, because it specifically increases the secretions of the mother’s fallopian tubes and uterus to provide appropriate nutritive matter for the developing morula and blastocyst. There is also reason tobelieve that progesterone affects cell cleavage in the early developing embryo.
d) The progesterone secreted during pregnancy helps the estrogen prepare the mother’s breasts for lactation.
Human Chorionic Somatomammotropin
A more recently discovered placental hormone is called human chorionic somatomammotropin. It is a protein with a molecular weight of about 38,000, and it begins to be secreted by the placenta at about the fifth week of pregnancy. Secretion of this hormone increases progressively throughout the remainder of pregnancy in direct proportion to the weight of the placenta. Although the functions of chorionic somatomammotropin are uncertain, it is secreted in quantities several times greater than all the other preg-nancy hormones combined. It has several possible important effects.
First, when administered to several types of lower animals, human chorionic somatomammotropin causes at least partial development of the animal’s breasts and in some instances causes lactation. Because this was the first function of the hormone dis-covered, it was first named human placental lactogen and was believed to have functions similar to those of prolactin. However, attempts to promote lactation in humans with its use have not been successful.
Second, this hormone has weak actions similar to those of growth hormone, causing the formation of protein tissues in the same way that growth hormone does. It also has a chemical structure similar to that of growth hormone, but 100 times as much human chorionic somatomammotropin as growth hormone is required to promote growth.
Third, human chorionic somatomammotropin causes decreased insulin sensitivity and decreased uti-lization of glucose in the mother, thereby making larger quantities of glucose available to the fetus. Because glucose is the major substrate used by the fetus to energize its growth, the possible importance of such a hormonal effect is obvious. Further, the hormone promotes the release of free fatty acids from the fat stores of the mother, thus providing this alter-native source of energy for the mother’s metabolism during pregnancy. Therefore, it appears that human chorionic somatomammotropin is a general metabolic hormone that has specific nutritional implications for both the mother and the fetus.
Other Hormonal Factors in Pregnancy
Almost all the nonsexual endocrine glands of the mother also react markedly to pregnancy. This results mainly from the increased metabolic load on the mother but also, to some extent, from the effects of placental hormones on the pituitary and other glands. Some of the most notable effects are the following.
Pituitary Secretion. The anterior pituitary gland of themother enlarges at least 50 per cent during pregnancy and increases its production ofcorticotropin, thy-rotropin, and prolactin. Conversely, pituitary secretionof follicle-stimulating hormone and luteinizing hormone is almost totally suppressed as a result of the inhibitory effects of estrogens and progesterone from the placenta.
Corticosteroid Secretion. The rate of adrenocortical secre-tion of the glucocorticoids is moderately increased throughout pregnancy. It is possible that these gluco-corticoids help mobilize amino acids from the mother’s tissues so that these can be used for synthesis of tissues in the fetus.
Pregnant women usually have about a twofold increase in the secretion of aldosterone, reaching a peak at the end of gestation. This, along with the actions of estrogens, causes a tendency for even a normal pregnant woman to reabsorb excess sodium from her renal tubules and, therefore, to retain fluid, occasionally leading to pregnancy-induced hypertension.
Secretion by the Thyroid Gland. The mother’s thyroid glandordinarily enlarges up to 50 per cent during pregnancy and increases its production of thyroxine a correspon-ding amount. The increased thyroxine production is caused at least partly by a thyrotropic effect of humanchorionic gonadotropin secreted by the placenta andby small quantities of a specific thyroid-stimulating hormone, human chorionic thyrotropin, also secreted by the placenta.
Secretion by the Parathyroid Glands. The mother’s parathy-roid glands usually enlarge during pregnancy; this is especially true if the mother is on a calcium-deficient diet. Enlargement of these glands causes calcium absorption from the mother’s bones, thereby maintain-ing normal calcium ion concentration in the mother’s extracellular fluid even while the fetus removes calcium to ossify its own bones. This secretion of parathyroid hormone is even more intensified during lactation after the baby’s birth, because the growing baby requires many times more calcium than the fetus does.
Secretion of “Relaxin” by the Ovaries and Placenta. Anothersubstance besides the estrogens and progesterone, a hormone called relaxin, is secreted by the corpus luteum of the ovary and by placental tissues. Its secretion is increased by a stimulating effect of human chorionic gonadotropin at the same time that the corpus luteum and the placenta secrete large quantities of estrogens and progesterone.
Relaxin is a polypeptide having a molecular weight of about 9000. This hormone, when injected, causes relax-ation of the ligaments of the symphysis pubis in the estrous rat and guinea pig. This effect is weak or possi-bly even absent in pregnant women. Instead, this role is probably played mainly by the estrogens, which also cause relaxation of the pelvic ligaments. It has also been claimed that relaxin softens the cervix of the pregnant woman at the time of delivery.
Related Topics