Chapter 4 | 8th Science - Heat | 8th Science : Chapter 4 : Heat
Chapter: 8th Science : Chapter 4 : Heat
Heat
UNIT 4
HEAT

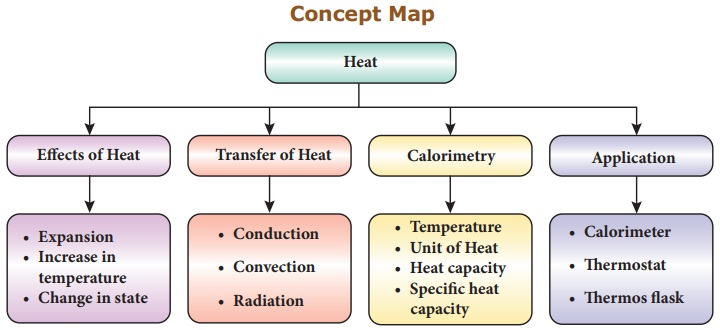
Learning Objectives
After the completion
of this lesson students will be able to:‹
* understand the
effects of heat.
* explain the transfer
of heat.
* know about
calorimetry.
* calculate heat
capacity and specific heat capacity of substances.
* list out the
functions of thermostat.
* know about the
working of thermos flask.
Introduction
All the substances in our
surrounding are made up of atoms and molecules. These atoms and molecules are
always at vibratory motion. Due to this motion, substances have an energy known
as heat energy. This energy flows from hot substances to cold substances or
from hot region to cold region of a substance. When heat energy is supplied to
any substance it increases the energy of the atoms and molecules in it and so
they start to vibrate. These atoms and molecules which vibrate make other atoms
and molecules to vibrate. Thus, heat energy is transferred from one part of the
substance to other part. We can see this heat energy transfer in our daily life
also. Heat energy brings about lot of changes. You will learn about them in
this lesson. You will also study about transfer of heat and measurement of heat
change.
Related Topics