Heat | Chapter 4 | 8th Science - Transfer of Heat | 8th Science : Chapter 4 : Heat
Chapter: 8th Science : Chapter 4 : Heat
Transfer of Heat
Transfer of Heat
If heat energy is supplied to any
substance, it will be transferred from one part of the substance to another
part. It takes place in different ways depending on the state of the substance.
Three ways of heat transfer are:
* Conduction
* Convection
* Radiation
1. Conduction
Activity 4
Take some hot water in
a cup and put a silver spoon in it. Leave the spoon inside the water for some
time. Now touch the other end of the spoon. Do you feel the heat?

Answer:
(i) Yes, we feel hot.
(ii) It is because heat in the hot water is transferred from one
end to other end of the spoon.
(iii) In solid substances such as silver spoon, atoms are
arranged very closely.
(iv) So heat transfer takes place from the higher temperature
region to lower temperature region.
(v) This is due to conduction.
How did the other end of the spoon
become hot? It is because heat in the hot water is transferred from one end to
other end of the spoon. In solid substances such as silver spoon, atoms are
arranged very closely. Hot water molecules which are vibrating transfer the
heat energy to the atoms in the spoon and make them vibrate.
Those atoms make other atoms to
vibrate and thus heat is transferred to the other end of the spoon.
In conduction heat transfer takes
place between two ends of the same solid or through two solid substances that
are at different temperatures but in contact with one another. Thus, we can
define conduction as the process of heat transfer in solids from the region of
higher temperature to the region of lower temperature without the actual
movement of atoms or molecules.
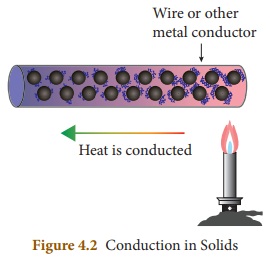
All metals are good conductors of heat. The substances
which does not conduct heat easily are called bad conductors or insulators.Wood, cork, cotton, wool, glass,
rubber, etc are insulators.
Conduction in daily life
* We cook food in vessels made up of
metals. When the vessel is heated, heat is transferred from the metal to the
food.
* When we iron dresses, heat is
transferred from the iron to the cloth.
* Handles of cooking utensils are
made up of plastic or wood because they are poor conductors of heat.
* The temperature inside igloo (snow
house) is warm because snow is a poor conductor of heat.
2. Convection
Activity 5
Take some water in a
vessel and heat it on a stove. Touch the surface of the water. It will be cold.
Touch it after some time. It will be hot now. How did the heat which was
supplied at the bottom reach the top?
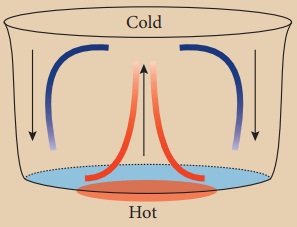
Answer:
(i) When water in the vessel is heated, water molecules at the
bottom receive heat energy and move upward.
(ii) Then the molecules at the top comes down and get heated.
(iii) This kind of heat transfer is known as convection.
When water in the vessel is heated,
water molecules at the bottom receive heat energy and move upward. Then the
molecules at the top comes down and get heated.
This kind of heat transfer is known
as convection. This is how air in the atmosphere is also heated. Thus, the form
of heat transfer from places of high temperature to places of low temperature
by the actual movement of molecules is called convection. Convection takes
place in liquids and gases.
Convection in daily life
* Formation
of land breeze and sea breeze is due to convection of air.
* Wind
flows from one region to another region by convection.
* In hot
air balloons heat is transferred by convection and so the balloon raises.
* In
refrigirators, cool air moves downward and replaces the hot air because of
convection.
3. Radiation
Radiation is the third form of heat
transfer. By conduction, heat is transferred through solids, by convection heat
is transferred through liquids and gases, but by radiation heat can be
transferred through empty space even through vacuum. Heat energy from the Sun
reaches the Earth by this form of heat transfer. Radiation is defined as the
way of heat transfer from one place to another in the form of electromagnetic
waves.
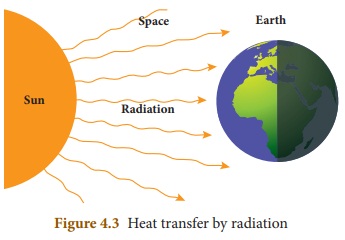
Radiation in daily life
* Heat energy from the Sun reaches
the Earth by radiation.
* While standing near fire we feel
the heat which is transferred as radiation.
* Black surfaces absorb heat
radiation. So that the bottom of the cooking vessels are painted black.
* White colour reflects heat
radiation. That’s why we are advised to wear white cloth during summer.
Heat transfer by
radiation is visible to our eyes. When a substance is heated to 500°C the
radiation begins to become visible to the eye as a dull red glow, and it is
sensed as warmth by the skin. Further heating rapidly increases the amount of
radiation, and its perceived colour becomes orange, yellow and finally white.
Related Topics