Heat | Chapter 4 | 8th Science - Calorimeter | 8th Science : Chapter 4 : Heat
Chapter: 8th Science : Chapter 4 : Heat
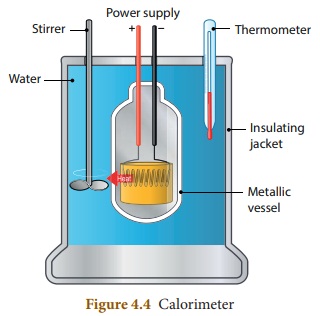
Calorimeter
Calorimeter
A calorimeter is a device used to
measure the amount of heat gained or lost by a substance. It consists of a
vessel made up of metals like copper or aluminium which are good conductors of
heat and electricity.
The metallic vessel is kept in an
insulating jacket to prevent heat loss to the environment. There are two holes
in it. Through one hole a thermometer is inserted to measure the temperature of
the contents. A stirrer is inserted through another hole for stirring the
content in the vessel. The vessel is filled with liquid which is heated by
passing current through the heating element. Using this device we can measure
the heat capacity of the liquid in the container.
The world’s first
ice-calorimeter was used in the year 1782 by Antoine Lavoisier and Pierre-Simon
Laplace, to determine the heat generated by various chemical changes.
Related Topics