Chapter: Biotechnology Applying the Genetic Revolution: Recombinant Proteins
Expression of Proteins by Insect Cells
EXPRESSION OF PROTEINS BY
INSECT CELLS
Mammalian cells are
relatively delicate and have complex nutritional requirements. This makes them
both difficult and expensive to grow in culture compared to bacteria or yeasts.
However, cultured insect cells are relatively robust and can be grown in
simpler media than mammalian cells. Consequently, insect-based expression
systems have been developed. These have the additional advantage of providing
posttranslational modifications that are very similar to those found in
mammalian cells.
The vectors used in cultured insect cells are
almost all derived from a family of viruses, the baculoviruses, which infect only insects (and related invertebrates
such as arachnids and crustaceans).
Baculoviruses are unusual in forming packages of virus particles, known as polyhedrons. Some infected cells
release single virus particles that can infect neighboring cells within the same insect. But when the host insect is dead or
dying, packages of virus particles embedded in a protein matrix are released
instead. The matrix protein is known as polyhedrin,
and the polyhedron structure protects the virus particles while they are
outside the host organism in the
environment. When swallowed by another insect, the polyhedrin is dissolved by
digestion and the polyhedron falls apart. This releases individual virus
particles that can infect the cells of the new host insect.
The polyhedrin gene has an
extremely strong promoter, and late during infection, the polyhedrin protein is
made in massive amounts. Because polyhedrin is not actually needed for virus
infection of cultured insect cells, the polyhedrin promoter may be used to
express recombinant proteins. The polyhedrin coding sequence is removed and
replaced by cDNA encoding the protein to be expressed.
There are many different types of
baculoviruses, and the one most often used is multiple nuclear polyhedrosis
virus (MNPV). This infects many
insects and replicates well in many cultured insect cell lines. A popular cell
line used to propagate this baculovirus is from the fall armyworm (Spodoptera frugiperda). Yields of polyhedrin— and therefore of a recombinant protein using the polyhedrin
promoter—are especially high in this cell line.
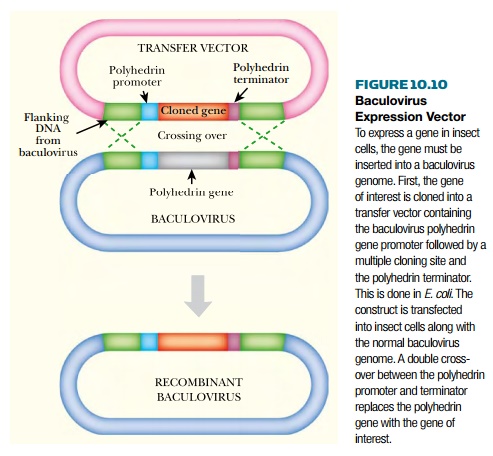
Because the expression vector is a virus,
construction is carried out in two stages (Fig. 10.10). In the first stage, a
“transfer” vector is used to carry the cDNA version of the cloned gene. The
transfer vector is an E. coli plasmid that carries a segment of MNPV DNA, which
initially included the polyhedrin gene and flanking sequences. The polyhedrin
coding sequence was then replaced with a multiple cloning site. When the cloned
gene is inserted, it is under control of the polyhedrin promoter. Construction
up to this point is done in E. coli. In the second stage, the segment
containing the cloned gene is recombined onto the baculovirus, thus replacing
the polyhedrin gene. To achieve this, insect cells are transfected with both
the transfer vector and with MNPV virus DNA. A double-crossover event generates
the required recombinant baculovirus.
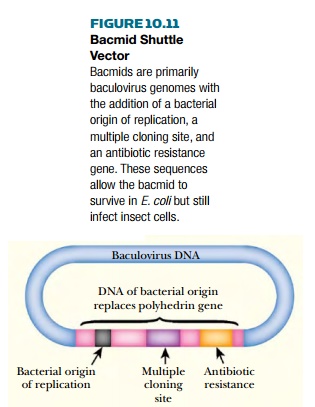
Because this procedure sometimes gives undesirable
results, other insect vectors have been constructed. One possibility is a
shuttle vector that replicates as a plasmid in E. coli and as a virus in insect
cells. Such baculovirus-plasmid hybrids are referred to as bacmids (Fig.
10.11). They consist of an almost entire baculovirus genome into which a
segment of DNA from an E. coli plasmid has been inserted. This region carries a
bacterial origin of replication, a selective marker, and a multiple cloning
site. The inserted segment replaces the polyhedrin gene of the baculovirus. The
cloned gene is inserted into the MCS giving a recombinant bacmid. Bacmid DNA is
then purified from E. coli and transfected into insect cells, where it
replicates as a virus.
The recombinant baculovirus may then be used to
infect cultured insect cells and the desired protein is produced over several
days. Several hundred eukaryotic proteins have been successfully made
Related Topics