Chapter: Biotechnology Applying the Genetic Revolution: Recombinant Proteins
Avoiding Toxic Effects of Protein Overproduction
AVOIDING
TOXIC EFFECTS OF PROTEIN OVERPRODUCTION
Although higher yields are
usually desirable, overproduction of a recombinant protein may harm the host
cell. In bacteria, when too much protein is manufactured too fast, the surplus
forms inclusion bodies. These are
dense crystals of misfolded and nonfunctional protein (see below). Thus
expression systems for recombinant proteins have features to control when and
how much protein the host cell makes. Two common expression systems are the pET
and pBAD systems for E. coli. These
have control mechanisms to switch recombinant protein production on or off, and
the pBAD system can also modulate the amount of protein produced.
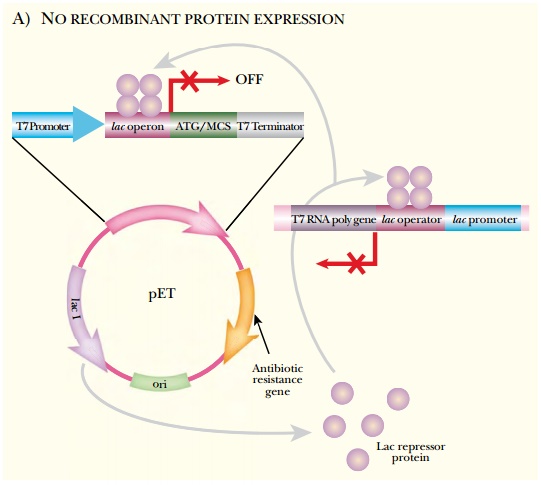
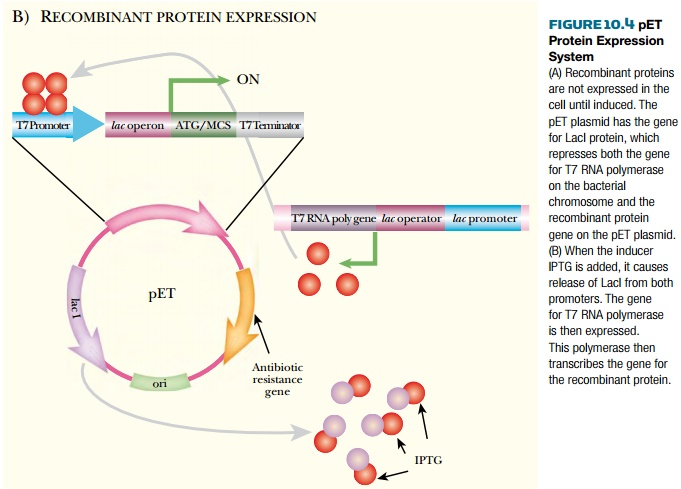
The pET vectors have a hybrid
T7/lac promoter, multiple cloning
site, and T7 terminator. Transcription from the hybrid T7/lac promoter requires T7 RNA polymerase. The host E. coli has the gene for T7 RNA
polymerase engineered into the chromosome, but the lac operon repressor, LacI, represses the gene. When IPTG is
added, this induces release of the LacI protein and expression of the gene. T7
RNA polymerase is then made and binds to the T7/lac hybrid promoter, and the cloned protein of interest is
manufactured (Fig. 10.4).
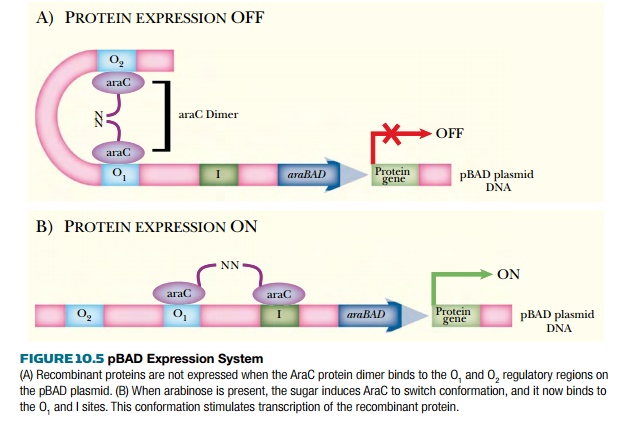
The pBAD system is based on the arabinose operon. This is induced by adding arabinose, which binds to the AraC regulatory protein. Activated AraC exits the O2 site and binds to the I site (Fig. 10.5). This activates transcription of the cloned gene. The pBAD system is modulated by the amount of arabinose added to the culture. If a lot of recombinant protein is needed, then more arabinose is added. However, if the recombinant protein is toxic to the host cells, then less arabinose is added, and less recombinant protein is made. This effect is based on the culture, not the cell. Each cell manufactures the recombinant protein in an “all-or-none” fashion. Low levels of arabinose will not activate every cell in the culture, whereas higher levels of arabinose do activate every cell.
Related Topics