Chapter: Basic Radiology : Brain and Its Coverings
Exercise: Brain Tumors
EXERCISE 12-3.
BRAIN TUMORS
12-6. In Case 12-6, what is the most likely diagnosis (Figure
12-20 A-C)?
A.
Extra-axial brain tumor
B.
Intra-axial brain tumor
C.
Frontal contusion
D.
Subdural hematoma
E.
Encephalocele
12-7. In Case 12-7, what is the most
likely cause of the patient’s symptoms (Figure 12-21 A, B)?

A.
Multiple sclerosis
B.
Inner ear abnormality
C.
Intraventricular meningioma
D.
Hematoma
12-8.
In Case 12-8, what is the most likely explanation for the patient’s
mental status changes (Figure 12-22 A, B)?

A.
Metastatic disease
B.
Intracranial hemorrhage
C.
Small infarcts
D.
Sarcoidosis
Radiologic Findings
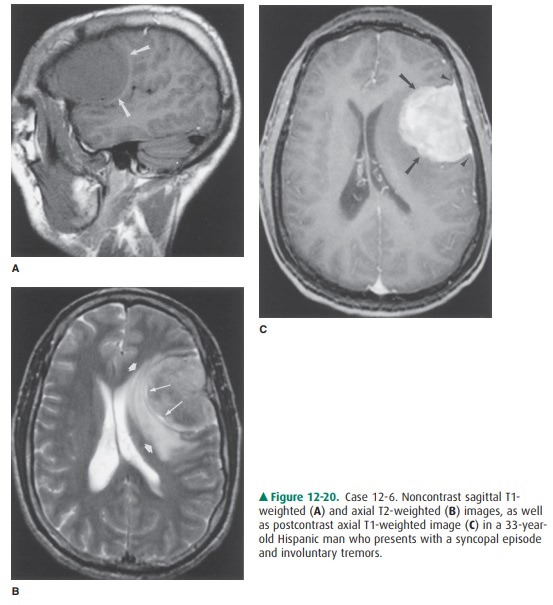
12-6. In this case, the sagittal T1-weighted image before
contrast administration shows an extra-axial, left frontal convexity mass
(Figure 12-20 A, arrows). This homogenous-appearing, smoothly marginated, mass
(arrows) is isointense to the normal gray matter (Fig-ure 12-20 A), and is
sometimes difficult to differenti-ate from normal brain tissue on unenhanced T1
images. On T2-weighted imaging, the mass has a het-erogeneous appearance, but
is predominantly isoin-tense to gray matter (Figure 12-20 B). The mass is
circumscribed by a thin rim (pseudocapsule) of in-creased T2 signal (long
arrows), as well as marginated by a more peripherally located band of T2 signal
hy-perintensity along its medial and posterior borders (short arrows). There is
distortion of the adjacent brain parenchyma, with compression of the left
lat-eral ventricle, and a mild shift of the midline struc-tures to the right.
Following intravenous GdDTPA administration, the mass enhances uniformly
(ar-rows), and dural tails are identified (arrowheads), al-lowing easy
identification (Figure 12-20 C). These features are fairly typical of a
meningioma (A is the correct answer to Question 12-6).
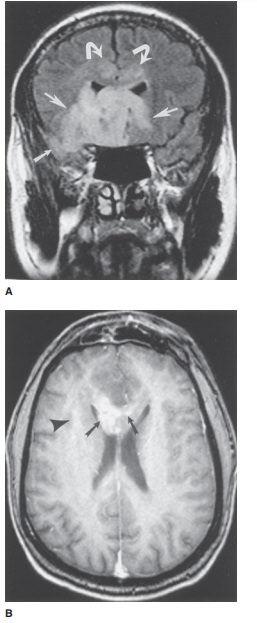
12-7. In this case, a coronal T2 FLAIR-weighted MR image
(Figure 12-21 A) demonstrates a large area of signal hyperintensity involving
the inferior frontal regions (large white arrows) and right temporal lobe
(small white arrow), with extension into the corpus callo-sum (curved arrows).
On the infused axial view, at the level of the body of the corpus callosum
(Figure 12-21 B), subtle, ill-defined enhancement is present within the right
cerebral hemisphere (arrowhead) with patchy enhancement (arrows) extending into
the body of the corpus callosum. This is one appear-ance of a malignant brain
tumor, in this case, an anaplastic oligodendroglioma (E is the correct an-swer
to Question 12-7).
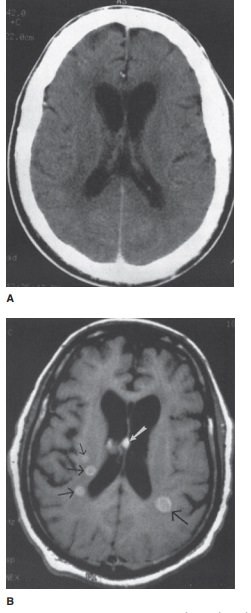
12-8. In this case, a contrast-enhanced axial CT scan shows no
definite abnormality (Figure 12-22 A). A gadolin-ium-enhanced axial T1-weighted
MR image shows multiple enhancing lesions (arrows) within the brain parenchyma
(Figure 12-22 B). In a patient with known lung cancer, metastatic disease is
the most likely explanation for multiple intracranial enhanc-ing lesions (A is
the correct answer to Question 12-8).
Discussion
Brain tumors can be classified in
a variety of ways. The tradi-tional classification of intracranial neoplasms is
based on his-tology. In this system, brain tumors are either primary (they
arise from the brain and its linings) or secondary (they arise from somewhere
outside the CNS, ie, metastases). Primarytumors, which account for
approximately two-thirds of all brain neoplasms, can be subdivided into glial
and nonglial tumors. Secondary tumors, especially from lung and breast cancer,
account for the remaining one-third of brain neo-plasms. Metastases are most
commonly parenchymal, but can also involve the skull and meninges.
Brain tumors can also be
classified according to patient age and general tumor location (ie, adult or
child, supraten-torial or infratentorial). Finally, brain tumors can be
classi-fied according to the specific anatomic region involved. For example, we
can generate lists of brain tumors that specifi-cally affect the pineal or the
pituitary regions.
Case 12-6 illustrates a useful
principle for interpreting studies of patients with suspected brain tumors. It
is very im-portant to first decide whether a mass is within the brain
parenchyma (intra-axial) or outside the brain (extra-axial). Extra-axial masses
usually turn out to be meningiomas, many of which can be removed surgically
with a very low in-cidence of recurrence. Intra-axial masses frequently turn
out to be astrocytomas, and the prognosis is less favorable.
The patient in Case 12-6 has an
extra-axial, dural-based, frontal convexity mass that markedly enhances with
Gd-DTPA. Meningiomas comprise 15% to 20% of intracranial tumors, predominantly
occur in females, and exhibit a peak age incidence of 45 years. They are the
most common nonglial primary CNS tumors. They can occur anywhere within the
head but typically occur along the dural venous si-nuses. The parasagittal
region and cerebral convexities are the most common locations. Anterior basal
or olfactory groove meningiomas account for 5% to 10% of intracranial
menin-giomas. Anosmia results from involvement of the olfactory tracts by the
tumor. These expansile lesions are slow-grow-ing, and the ensuing mass effect
on the adjacent brain parenchyma is gradual. The absence of reactive edema in a
subset of these lesions can be seen as a result of their slow growth. These
masses usually demonstrate intense and uni-form enhancement, independent of
tumor size. A layer of thickened dural enhancement (“dural tail”) is commonly
seen extending away from the base of the meningioma. In many cases, this
finding represents reactive thickening with-out tumor involvement.
Case 12-7 demonstrates a large,
infiltrating (aggressive or high-grade) glioma involving the majority of the
right frontotemporal lobe, with extension into the corpus callo-sum. Although
there is some overlap of the MR imaging features characteristically seen with
these invasive neo-plasms and their less aggressive (lower-grade) counterparts,
the imaging features of higher-grade neoplasms, on the whole, are distinctly
different from those seen with lower-grade lesions. High-grade gliomas, namely
anaplastic astro-cytomas and oligodendrogliomas (as in this case), as well as
glioblastoma multiforme (the most highly malignant glioma), demonstrate
heterogeneous signal characteristics, generally a reflection of the variable
cellularity, in additionto the presence of necrosis, hemorrhage, and cystic
foci. Calcification and hemorrhage are more common in oligo-dendrogliomas,
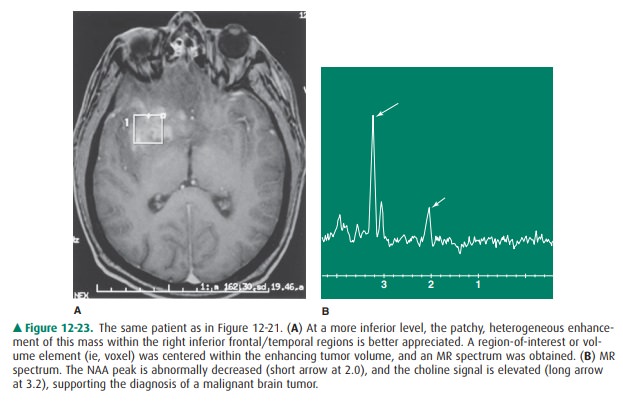
often accompanied by cyst formation and necrosis. The spectroscopic findings of
decreased NAA and increased choline suggest decreased neuronal/axonal den-sity
and increased breakdown of cell membranes (Figure 12-23 A, B).
Oligodendrogliomas account for
about 5% of primary gliomas, occurring most frequently within the frontal lobe
and often involving the cortex. The majority of patients present with seizures.
On the other hand, glioblastoma multiforme is the most common primary malignant
brain neoplasm and oc-curs most frequently in patients over 50 years of age.
Patients with glioblastoma multiforme present with neurologic deficits or
new-onset seizures. The prognosis in these latter cases is dis-mal;
postoperative survival averages 8 months.
On T2-weighted scans, these
high-grade masses usually exhibit heterogeneous signal characteristics, with
areas of high T2 signal attributable to tumor tissue, necrosis, cysts, and
reactive edema, whereas regions of low signal may reflect hemorrhage or calcification.
The corresponding tissue pathology of this region often shows tumor cells
residing within and extending beyond the surrounding edema. En-hancement is
highly variable within anaplastic oligodendrogliomas. Other types of malignant
gliomas, such as glioblastoma multiforme, typically demonstrate intense
en-hancement. The corpus callosum is often involved by a high-grade glial
tumor, which may grow medially from an adjacent hemispheric source or may arise
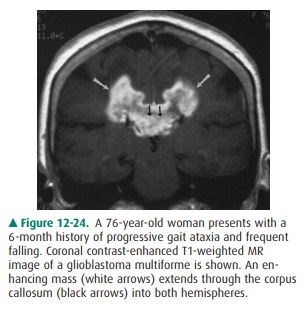
independently within this structure. “Wings” may extend symmetrically or
asymmetri-cally into both cerebral hemispheres, exhibiting a butterfly-type
appearance (Figure 12-24), appropriately termed butterfly glioma. Perfusion
studies on high-grade gliomas generally show increased blood flow and volume, reflecting
the increased vascular density and permeability of these tu-mors. In contrast,
low-grade gliomas may appear only as a re-gion of amorphous signal abnormality
(most obvious on T2-weighted images), often without associated enhancement or
perfusion abnormality.
Case 12-8 illustrates a very
important point to remem-ber when working up patients with suspected metastatic
disease to the brain: MR imaging is considerably more sen-sitive than CT in
detecting metastases. This is not a trivial point, because surgical resection
of single, not multiple, brain lesions is sometimes performed. Conversely, the
suc-cessful application of radiotherapy protocols relies on sen-sitively and
accurately detecting the entire metastatic tumor burden. Metastatic disease to the
brain has a variety ofmanifestations, the most common being parenchymal
involve-ment. Typical hematogenous brain metastases demonstrate solid or
ringlike enhancement on CT or MR scans, occur near gray matter/white matter
junctions, and are usually surrounded by a marked amount of edema. They most
commonly metastasize from lung or breast primaries.
Related Topics