Chapter: Basic Radiology : Brain and Its Coverings
Brain and Its Coverings - Radiology Techniques
TECHNIQUES
Radiologic modalities useful in
evaluating the brain and its coverings can be divided into two major groups:
anatomic modalities and functional modalities. Anatomic modalities, which
provide information mostly of a structural nature, include plain films of the
skull, CT, MR imaging, cerebral arteriography (CA), and ultrasonography (US).
On the other hand, SPECT and PET imaging, CT perfusion, DW and PW MR imaging,
fMRI, and MRS are primarily functional modalities, which give information about
brain perfusion or metabolism. Some techniques provide both anatomic and
functional information. For example, cerebral arteriography depicts blood
vessels supplying the brain but also allows us to estimate brain circulation
time. Ultrasound of the carotid bifurcation is another modality that provides
both anatomic and functional information. A routine sonogram of the carotid
bifurcation gives anatomic data that, when combined with Doppler data, readily
provides information about blood flow.
The following discussion of
current neuroradiologic tech-niques emphasizes relative examination cost and
patient risk, along with the advantages and disadvantages of each tech-nique.
The normal imaging appearance of the brain and its coverings is also
illustrated.
Plain Radiographs
Plain radiographs of the skull
are obtained by placing a pa-tient’s head between an x-ray source and a
recording device (ie, x-ray film). Whereas bones of the skull attenuate a large
number of x-rays to create an image, soft tissues such as scalp or brain are
poorly visualized, if at all. Another difficulty in plain film interpretation
results from the spherical shape of the skull, leading to multiple superimposed
structures. The resultant skull radiograph primarily gives information about
the bones of the skull, but no direct information about the intracranial
contents. Indirect information about intracra-nial abnormalities can sometimes
be obtained from the skull plain radiograph, although this information can be
quite subtle, even in the setting of advanced disease. Skull plain radiographs
have been largely replaced today by more sensitive techniques such as CT or MR
imaging. Even in the setting of suspected skull fracture, plain radiographs are
rarely indicated, because CT scans also show the fracture, as well as any
intracranial abnormality that might require treatment. Currently, plain
radiographs of the skull serve a very limited role in routine neuroimaging and
are only briefly discussed.
Computed Tomography
CT scans consist of
computer-generated cross-sectional im-ages obtained from a rotating x-ray beam
and detector sys-tem. Advances in scanning technology now permit simultaneous
acquisition of multiple images during a single rotation of the x-ray tube (eg,
currently up to 256 slices) during a breath-hold. The resultant images, unlike
plain films, exquisitely depict and differentiate between soft tis-sues, thus
allowing direct visualization of intracranial con-tents and abnormalities
associated with neurologic diseases. The contrast or brightness (“window” or
“level,” respec-tively) of these images can be adjusted to highlight
particu-lar tissues.
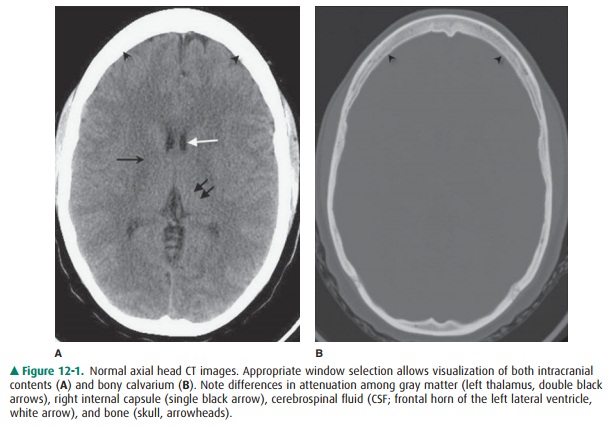
Typically, a head CT consists of images adjusted to em-phasize soft-tissue detail (soft-tissue windows) as well as im-ages adjusted to visualize bony detail (bone windows) (Figure 12-1). As stated earlier, CT image generation is dependent on variable attenuation of the x-ray beam based on the density of structures it passes through (eg, bones of the skull base are very dense and attenuate a large percentage of the x-ray beam). Therefore, cortical bone appears white (has a high at-tenuation value or Hounsfield unit), whereas air within the paranasal sinuses appears black (has a low attenuation value) (Figure 12-1). Cerebral white matter has a slightly lower Hounsfield number than does cerebral gray matter and con-sequently appears slightly darker than gray matter on a head CT scan (Figure 12-1A). Intracranial pathologic conditions can be either dark (low attenuation) or bright (high attenua-tion), depending on the particular abnormality. For example, acute intracranial hemorrhage is typically very bright, whereas an acute cerebral infarction demonstrates low atten-uation when compared to the surrounding normal brain be-cause of the presence of edema.
The CT technologist can change
the slice thickness and angulation, among other technical factors, to alter the
way an image appears. Images are typically obtained axially in helical fashion,
with acquisition of a volumetric data set. Current scanner technology allows
the axial data set to be reformatted in coronal, sagittal, or oblique planes or
as a 3-D image, with little, if any, loss of resolution. CT examinations may be
per-formed after intravenous administration of an iodinated contrast agent, especially
when MRI is contraindicated or unavailable. These agents “light up” or enhance
normal blood vessels and dural sinuses, as well as intracranial structures that
lack a blood-brain barrier (BBB), such as the pituitary gland, choroid plexus,
or pineal gland. Pathologic conditions that interrupt the BBB (such as
neoplasm, infection, or cere-bral infarction) also demonstrate enhancement
after contrast material administration. For this reason, lesions that may be
invisible on a noncontrast study are often obvious on the contrast-enhanced
scan.
The intravenous administration of
a contrast bolus can be appropriately timed to maximize vascular opacification
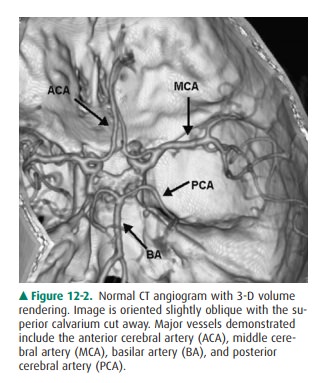
of the arterial or venous circulation (CTA or CTV, respectively). These high
spatial resolution 3-D CTA images (Figure 12-2) of the cervical and
intracranial vasculature are routinely em-ployed to quantify vessel stenosis
due to atherosclerotic dis-ease, to assess for vascular injury related to
trauma, or to detect cerebral aneurysm in the patient with subarachnoid
hemorrhage.
In particular, CTA has become a
standard component of evaluating the acute stroke patient. CTA accurately
identifies the location and extent of large vessel occlusions and can be
supplemented by a more detailed, quantitative evaluation of the cerebral
microvascular hemodynamics (CT perfusion) during the early phase of bolus
passage. Software analysis of this tailored CTA data produces maps of
capillary-level cere-bral perfusion, typically measured by mean transit time
(MTT), cerebral blood flow (CBF), and cerebral blood vol-ume (CBV). In the
setting of cerebral infarction, these pa-rameters can help interpret the
infarct “core” (CBV) versus the ischemic “penumbra” (MTT and CBF). Evaluation
of po-tential mismatch between the infarct core and surrounding penumbra serves
as the rationale for instituting various reperfusion techniques.
Another recent application of CTA
is in the screening eval-uation of blunt cerebrovascular injury, including
closed head injuries, seatbelt abrasion (or other soft-tissue injury) of the
anterior neck, basilar skull fracture extending through the carotid canal, and
cervical vertebral body fracture. It is an ac-curate technique for detecting
internal carotid artery (ICA) dissections and for assessing stenoses, although
evaluation is difficult in areas of surrounding dense bone as a result of
as-sociated “streak artifact.” However, this noninvasive, relatively short
imaging procedure rivals conventional angiographic methods, as it requires no
patient transfer and can sensitively identify vascular injury in relation to
other associated brain insults, cervical spine injury, or facial or basilar
skull fractures.
High-resolution data acquisition
during the venous phase following intravenous contrast administration (CT
venogra-phy) can be used to identify dural sinuses and cerebral veins, evaluate
for dural venous sinus thrombosis, and distinguish partial sinus obstruction
from venous occlusion in the setting of adjacent brain masses. CT venography
can also differenti-ate slow flow from thrombosis, which may occasionally be
difficult with MR techniques.
The major advantages of CT are
that it is inexpensive, is widely available, can be used in patients with
MR-incom-patible hardware, and allows a relatively quick assessment of
intracranial contents in the setting of a neurological deficit. The images
obtained are very sensitive to the pres-ence of acute hemorrhage and
calcification, and images re-vealing exquisite bony detail of the skull and
skull base can be acquired. Because of the configuration of the scanner,
patients are reasonably accessible for monitoring during the examination.
CT scanners do have a number of
disadvantages, how-ever. Patients are exposed to ionizing radiation and
iodine-based contrast agents (although lower doses of contrast are needed with
newer multidetector scanners). Imaging arti-facts can interfere with accurate
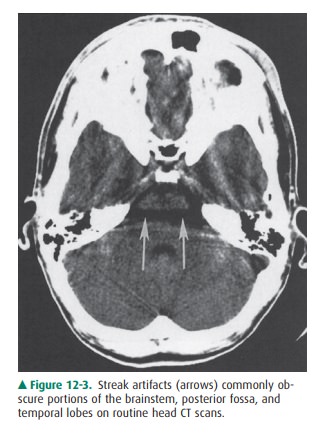
interpretation. In particular, images of the brainstem and posterior fossa are
often de-graded by “streak artifacts” from dense bone (Figure 12-3). Streak
artifacts from metallic objects (eg, fillings, braces, surgical clips) can also
obscure abnormalities. Images can be severely degraded by patient motion.
Fortunately, unlike MR scans, individual CT images degraded by motion can be rap-idly
reacquired.
Magnetic Resonance Imaging
One of the most exciting
developments in radiology during the past 30 years has been the growth of
magnetic resonance imaging (MRI), which is currently the mainstay of clinical
neuroimaging. The concept of nuclear magnetic resonance (NMR), initially used
for probing the physiochemical struc-ture of molecules, was first described in
the 1930s, but it took more than 40 years before the translation of NMR
phenom-ena could be used for clinical imaging.
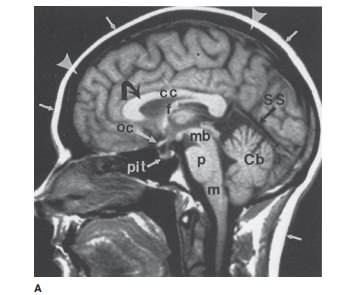
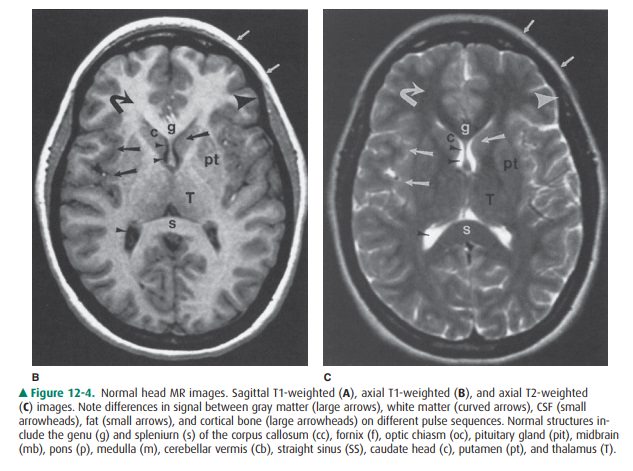
MR examinations, like CT scans,
consist of computer-reconstructed cross-sectional images (Figure 12-4). In MR
imaging, however, unlike CT scans or plain radiographs, the information collected
is not x-ray beam attenuation. The MR image is a visual display of NMR data
collected princi-pally from nuclei within body tissues—especially hydrogen
nuclei within water and fat molecules. Intrinsic tissue relax-ation occurs by
two major pathways, called longitudinal, or T1, and transverse, or T2, decay.
MR imaging sequences that emphasize T1 decay are commonly referred to as
T1-weighted; sequences that accentuate T2 relaxation proper-ties are called
T2-weighted (Figure 12-4). Most MR scans of the brain use both of these
sequences, because certain ab-normalities may only be obvious on one or the
other. T2-weighted images are usually easy to identify because fluid (eg,
cerebrospinal, globe vitreous) is very bright; fluid on a T1-weighted scan is
usually dark. Fat is bright on T1-weighted scans, but darker on T2-weighted
images. On the other hand, both cortical bone and air are very dark on all
imaging sequences. Brain tissue has intermediate intensity; vessels can have
almost any signal, depending on the veloc-ity of flowing blood.
The most commonly used clinically
approved contrast agent for MR imaging is gadopentetate-dimeglumine or Gd-DTPA,
which is very well tolerated and generally safe, al-though caution must be used
in patients with renal impairment because of the associated risk of developing
nephrogenic systemic sclerosis. Its major use in the CNS is to improve lesion
detectability by “lighting up” pathologic conditions that either lack a BBB or
have a disrupted BBB.
Conventional MR imaging depicts
excellent soft-tissue contrast. Traditionally, long image acquisition times,
image artifacts related to patient motion, and the increased cost of scanning
due to limited patient throughput have ham-pered the clinical utility of MR
imaging. Over the past 15 years, technical advances in gradient technology,
coil design, image reconstruction algorithms, contrast adminis tration
protocols, and data acquisition strategies have accel-erated the development
and implementation of fast imaging methods. These techniques, including fast
gradient echo imaging, fast spin echo imaging, FLAIR (fluid-attenuated
inversion recovery), and echo planar imaging, have enabled substantial
reductions in imaging time. Images may be ac-quired during a single breath-hold
on a clinical scanner, eliminating respiratory and motion artifacts. Vessel
con-spicuity can be enhanced by application of fat-suppression sequences, which
eliminate unwanted signal from back-ground tissues. These improvements have led
to a vast range of applications that were previously impractical, in-cluding
high-resolution MRA, DW and PW MR imaging, MRS, fMRI, and real-time monitoring
of interventional procedures.
Since its first clinical
application nearly 15 years ago, MRA has proven to be a useful tool for evaluation
of the cer-vical or intracranial carotid vasculature. MRA represents a class of
techniques that utilize the MR scanner to noninva-sively generate
three-dimensional images of the carotid or vertebral-basilar circulations.
Although a detailed discussion of these techniques is beyond the scope of this topic,
sev-eral comments are noteworthy. These methods permit dis-tinction between
blood flow and adjacent soft tissue, with or without administration of
intravenous contrast. As noted earlier, revolutionary developments have
permitted MRA images to be rapidly acquired with ever-improving temporal and
spatial resolution.
Presently, MRA serves as one of
the first-line studies for evaluation of arterial occlusive disease and for
screening of intracranial aneurysms. These methods have largely re-placed
conventional arteriographic studies for evaluation of atherosclerotic disease,
except in cases of critical steno-sis (> 70%). In these instances, the
degree of luminal nar-rowing may be overestimated by MRA and may require
verification with CTA, catheter-based study, or Doppler ul-trasound. Moreover,
aneurysms detected on an intracranial MRA typically require a catheter-based
study for detailing aneurysm size and orientation, for establishing the
location of adjacent vessels and collateral flow, and for confirming suspicious
vascular dilatation, as well as for detecting the presence of vasospasm or
additional aneurysms that may not be readily apparent on the MRA study. In an
increasing number of cases, catheter-based studies will additionally be
performed for coil embolization (obliteration) of detected aneurysms, rather
than surgical clipping.
Molecular diffusion, the random
translational move-ment of water and other small molecules in tissue, is
ther-mally driven and is referred to as Brownian motion. Over a given time
period, these random motions, expressed as molecular displacements, can be
detected using specifically designed diffusion-sensitive MR sequences. A common
ap-plication of diffusion imaging is the detection of early is-chemic
infarction, where the infarcted tissue “lights up” because of a “restricted
diffusion” state within the intracellu-lar compartment. Other applications of
diffusion-sensitive sequences include differentiating cysts from solid tumors,
as well as evaluating inflammatory/infectious conditions (en-cephalitis,
abscess) or white matter abnormalities (hypertensive encephalopathy).
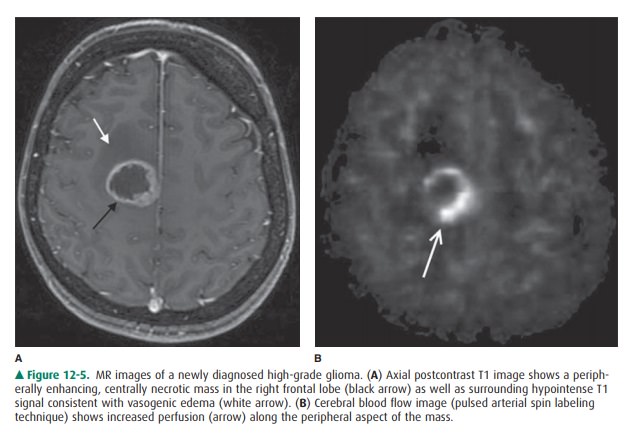
Perfusion MR imaging measures
cerebral blood flow at the capillary level of an organ or tissue region. Perfusion-weighted
MR imaging has applications in the evaluation of a number of disease states,
including cerebral ischemia and reperfusion, brain tumors (Figure 12-5),
epilepsy, and blood flow deficits in Alzheimer’s disease. In addition, the
close spa-tial coupling between brain activity and CBF permits the ap-plication
of perfusion MR techniques to imaging brain function. MR perfusion imaging is
technically complex and requires advanced scanner and postprocessing software
for image generation. Various methods can be employed includ-ing contrast bolus
technique (analogous to CT perfusion) or arterial spin labeling (ASL). ASL uses
a radiofrequency pulse to “label” protons flowing in the cervical arteries and
that signal is subsequently imaged as those protons flow into the cerebrum. One
of the major advantages of ASL is that itrequires no contrast administration,
which is of great benefit in patients with renal impairment.
Functional MR imaging is an
important brain mapping technique that uses fast imaging techniques to depict
re-gional cortical blood flow changes in space and time during performance of a
particular task (eg, flexion of the index fin-ger). The utilization of this
technique to localize brain activ-ity is historically based on measurable
increases in cerebral blood flow (and blood volume) with increased neural
activ-ity, referred to as neurovascular coupling. The hemodynamic response to a
stimulus is not instantaneous, but on the order of a few seconds. Consequently,
fMRI techniques are consid-ered an indirect approach to imaging brain function,
but provide excellent spatial resolution and can be precisely matched with
anatomic structures. Changes in blood oxy-genation and perfusion can be imaged
using fMRI tech-niques, which has become the most widely used modality for
depicting regional brain activation in response to sensorimo-tor or cognitive
tasks.
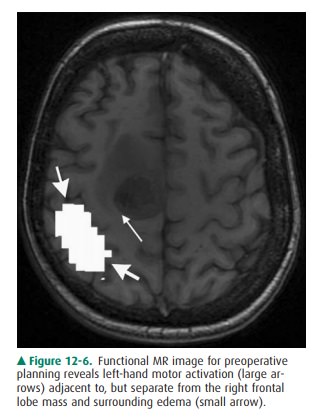
An important clinical application
of fMRI is presurgical mapping, whereby eloquent brain cortex can be defined in
relation to mass lesions (Figure 12-6). This allows for thejudicious selection
of an appropriate management strategy (surgical versus nonsurgical) according
to the functional nature of the adjacent brain tissue. A second application
in-volves determination of the cerebral hemisphere responsible for language and
memory tasks in a patient with complex partial seizures, prior to undergoing
temporal lobectomy. Additionally, several groups have reported successful
functional activation studies for lateralizing language preoperatively uti-lizing
fMRI.
MR spectroscopy (MRS) provides
qualitative and quan-titative information about brain metabolism and tissue
composition. This functional analysis is based on detecting variations in the
precession frequencies of spinning pro-tons in a magnetic field. One factor
influencing the preces-sion or resonance frequency is the chemical environment
of the individual proton. Protons in different cerebral metabolites can be
sensitively discriminated on this basis, and the position of these metabolites
can be displayed as a spectrum. The x-axis position of a given metabolite
reflects the degree of “chemical shift” of the metabolite with re-spect to a
designated reference metabolite and is expressed in units of parts per million
(or ppm). The area under thepeak is determined by the number of protons that
con-tribute to the MR signal.
The major metabolites detected in
the CNS are N-acetyl aspartate (NAA),
a neuronal marker; choline, a marker for cellularity and cell membrane
turnover; creatine, a marker for energy metabolism; and lactate, a marker for
anaerobic metabolism. In addition to these metabolites, others have been
assessed, including alanine, glutamine, myoinositol, and succinate, using
various MR strategies. Presently, MRS is being used in clinical practice to
provide functional informa-tion regarding many CNS abnormalities, and
complements the conventional MR imaging study. A common applica-tion relates to
the pre- and posttreatment evaluation of brain tumors, with MRS playing an
important role in as-sessing for residual or recurrent tumor following surgical
resection.
MR imaging offers a number of
advantages over CT in the workup of patients with neurologic disease. Its
soft-tis-sue contrast resolution is superior to that of CT, and le-sions that
may be subtle or invisible on CT are frequently obvious on MR imaging. MR
imaging also allows acquisi-tion of multiplanar views in the sagittal, axial,
coronal, and oblique projections that may be impossible to obtain with CT.
Furthermore, MR imaging gives information about blood flow without the need for
a contrast agent, and bony streak artifacts that obscure lesions of the
brainstem and cerebellum on CT scans are not present on MR images. Finally, MR
imaging does not expose the patient to ionizing radiation.
Cerebral Arteriography
Cerebral arteriography involves
the injection of water-soluble contrast material into a carotid or vertebral
artery. Contrast material is injected into the desired vessel via a small
catheter, which has been introduced into the body through the femoral or
brachial artery. Information about the arterial, capillary, or venous
circulation of the brain is recorded on se-rial plain films or, most commonly,
digitized for viewing on a monitor or for storage within a computer (Figure
12-7).
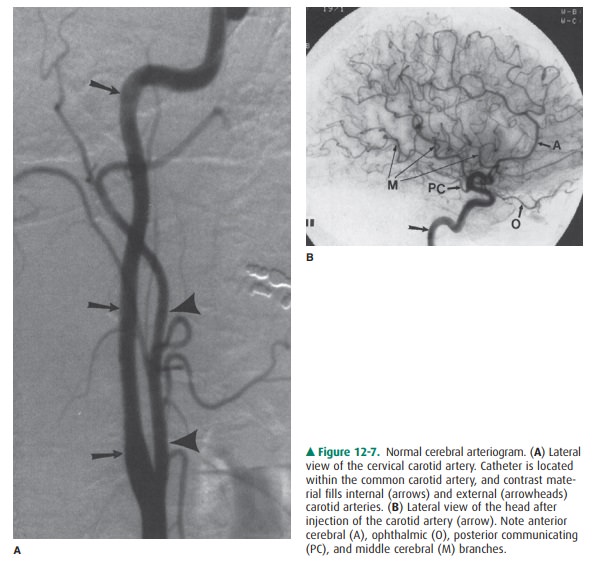
Cerebral arteriograms are
expensive (two to three times as much as MR examinations) and are relatively
more risky procedures than other noninvasive neuroradiologic studies. The major
risk of the procedure is stroke, which may occur in one of every 1,000
patients. Stroke during cerebral arteri-ography occurs either from an embolic
event (eg, inadver-tent injection of air, thrombus formation on the catheter
tip, atherosclerotic plaque dislodged by catheter manipula-tion) or from
catheter-related local vessel trauma (eg, dis-sections or occlusions).
Although CT angiography has
largely replaced catheter angiography for most routine diagnostic evaluations,
catheter angiography is invaluable in the workup of vascular diseases affecting
the CNS. Specifically, it remains the gold standard for assessing vasculitis
and is indispensable in eval-uating and treating cerebral aneurysms and certain
intracra-nial vascular malformations or fistulas. It is a useful adjunct to
cross-sectional imaging (CTA, MRA, or US) to assess vas-cular stenosis as well
as carotid or vertebral artery integrity after trauma to the neck, especially
in the setting of acute neurological deficit. Finally, it is unsurpassed for
showing vascular anatomy of the brain and is, therefore, useful as a
preoperative road map.
The field of interventional
neuroradiology continues to grow and exert considerable impact on the diagnosis
and treatment of certain CNS diseases. New catheter designs and materials,
recently developed endovascular devices (ex-tracranial/intracranial stents),
and an increasing number of trained specialists performing endovascular
procedures have led to novel therapeutic applications and approaches for
managing previously untreatable conditions. Endovascular diagnostic and
therapeutic procedures, based on fundamental cerebral arteriography principles,
have gained widespread
acceptance and, in some cases, rival traditional neurosurgi-cal approaches in terms of complication rates, clinical out-comes, and long-term survival benefit. Although a full discussion of these techniques is beyond the scope of this topic, they include pharmacologic and mechanical throm-bolysis of intracranial clot in the setting of acute infarction or dural sinus thrombosis; embolization (obliteration) of intracranial aneurysms using thrombosing material (ie, coils); carotid artery angioplasty and/or stent placement for critical stenotic narrowing or radiation-induced arterial stricture; preoperative or definitive devascularization of a hypervascular mass or arteriovenous malformation; em-bolization of small, bleeding external carotid artery branches in epistaxis; balloon occlusion tests of the carotid artery; and endovascular treatment of vasospasm. Em-bolization materials include particulate emboli, liquid adhe-sive glues, and various coils.
Ultrasonography
Ultrasonography is the diagnostic
application of ultrasound to the human body. Major applications of
ultrasonography in CNS disease include gray-scale imaging and Doppler
evaluation of carotid artery patency and flow in the setting of
atherosclerosis, assessment of vasospasm in the setting of sub-arachnoid
hemorrhage using transcranial Doppler, screeningevaluation of intracranial
abnormalities in the newborn and young infant (Figure 12-8), and detection of
intracranial hemorrhage in premature infants prior to extracorporeal membrane
oxygenation therapy. Ultrasound has also been used intraoperatively to
demonstrate the spinal cord and sur-rounding structures during spine surgery
and to define tumor and cyst margins during craniotomies.
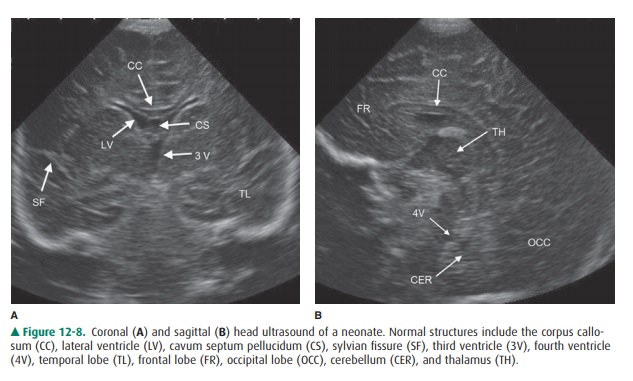
Transcranial Doppler is a
recently developed tool in the evaluation of cerebrovascular disorders. It uses
low-fre-quency sound waves to adequately penetrate the skull and produces
spectral waveforms of the major intracranial vessels for evaluation of flow
velocity, direction, amplitude, and pul-satility. Present clinical applications
include diagnosis of cerebral vasospasm, evaluation of stroke and transient
is-chemic attack, detection of intracranial emboli, serial moni-toring of
vasculitis in children with sickle cell disease, and assessment of intracranial
pressure and cerebral blood flow changes in patients with head injury or mass
lesions.
Ultrasound examinations, although moderately expen-sive, are virtually risk-free to the patient, involve no ioniz-ing radiation, and are portable (ie, can be performed at the bedside). However, examination quality and therefore di-agnostic accuracy are operator-dependent. Also, the heavy reliance of ultrasonography on the presence of an adequate “acoustic window” through which an examination can be performed diminishes its usefulness in examining thebrain after the fontanelles close in infancy. Finally, to the untrained eye, anatomic structures and pathologic processes as depicted by US are not as readily apparent as they are on CT or MR images.
Single Photon Emission Computed Tomography
SPECT uses a rotating gamma
camera to reconstruct cross-sectional images of the distribution of a
radioactive pharma-ceutical that has been administered to a patient (usually
intravenously). For brain imaging, radioactive iodine (123I) or technetium
(99mTc) is combined with a compound that rap-idly crosses the BBB
and localizes within brain tissue in pro-portion to regional blood flow. The
rotating gamma camera detects gamma rays emitted by the radiopharmaceutical and
produces cross-sectional images of the brain that are really a map of brain
perfusion (Figure 12-9). SPECT imaging also gives indirect information about
brain metabolism, because perfusion is usually highest to parts of the brain
with high metabolic activity and lowest to areas with low metabolic de-mand.
Normal SPECT examinations demonstrate activity concentrated primarily in areas
of high perfusion/metabolism, such as the cortical and deep gray matter (Figure
12-9).
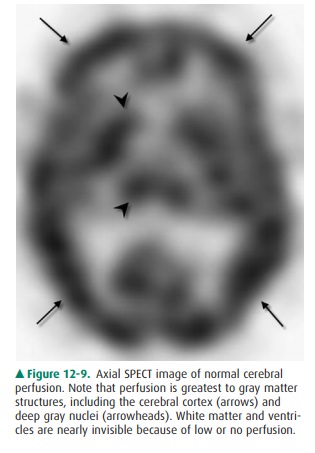
SPECT studies are moderately expensive (as much as or more than brain MR imaging), and, as expected, they pro-vide limited anatomic information. SPECT also exposes patients to ionizing radiation. Because patients rarely have allergic reactions to the radiopharmaceuticals used, the ex-amination is of low risk. Although SPECT provides critical information regarding regional cerebral perfusion, partic-ularly in the setting of stroke, this information can be more readily obtained during CTA/CT perfusion or MR perfu-sion acquisitions. SPECT has also been used with varying degrees of success in the workup of patients with epilepsy or dementia.
Positron Emission Tomography
PET scans consist of
computer-generated cross-sectional im-ages of the distribution and local
concentration of a radio-pharmaceutical. This technique is very similar to
SPECT imaging; however, there are differences in the type of camera and
radiopharmaceuticals used. PET studies use radiophar-maceuticals labeled with a
cyclotron-produced positron emitter, which are very expensive to produce and
have a very short half-life (on the order of seconds to minutes). The most
widely used radiotracer is 18F-deoxyglucose. PET scanning with this
agent gives a measurement of brain glucose metab-olism. Areas of high metabolic
activity (ie, cerebral cortex, deep gray nuclei) demonstrate greater
radiopharmaceutical uptake than do areas of low metabolic activity, such as
white matter or cerebrospinal fluid (Figure 12-10). The bones of the skull and
scalp soft tissues are, for the most part, invisible. Other agents are useful
in assessing regional cerebral blood flow, neuroreceptor function, and the
like.
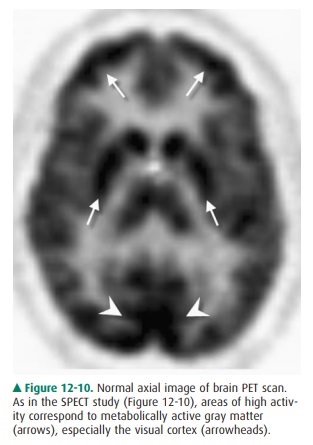
Since the previous edition, PET
scans have become much more widely available, although they remain expensive.
The expense, in large part, is related to the cost of imaging equipment and in
the production or delivery of radiophar-maceuticals. Although patients
undergoing PET examina-tions are exposed to ionizing radiation, the overall
risk to the patient is low. Anatomic resolution, although not as good as with
CT or MR imaging, is better than with SPECT imaging. The major advantage of PET
imaging is that it is extremely versatile, providing in vivo information about
brain perfusion, glucose metabolism, receptor density and, ultimately, brain
function.
PET provides useful information
in the setting of stroke, epilepsy, dementia, and tumors. At present, the two
main in-dications are in the workup of patients with complex partial seizures
and in identifying tumor recurrence in patients who have undergone surgery,
radiation therapy, or both, for brain tumors.
Related Topics