Chapter: Organic Chemistry: Carboxylic acids and carboxylic acid derivatives
Enolate reactions - Carboxylic acids and carboxylic acid derivatives
ENOLATE REACTIONS
Key Notes
Enolates
Esters
contain acidic α protons
which can be removed with a strong base to form enolate ions. A bulky base is
used to prevent the possibility of nucleophilic substitution taking place.
Diethyl malonate can be converted to a stable enolate ion using sodium ethoxide
as base.
Alkylations
Enolate
ions can be alkylated with alkyl halides. Diethyl malonate can be alkylated
twice, hydrolyzed, and decarboxylated to give a disubstituted ethanoic acid.
Claisen condensation
Two
esters can be condensed together to form a β-ketoester. The reaction involves the formation
of an enolate ion from one molecule of ester, which then undergoes nucleophilic
substitution with another ester molecule. Mixed Claisen condensations are
possible with two different esters or between an ester and a ketone.
Enolates
Enolate ions can be formed from aldehydes and
ketones containing protons on an α-carbon. Enolate ions can also be formed from
esters if they have protons on an α-carbon (Fig. 1). Such protons are slightly
acidic and can be removed on treatment with a powerful base such as lithium
diisopropylamide (LDA). LDA acts as a base rather than as a nucleophile since
it is a bulky molecule and this prevents it attacking the carbonyl group in a
nucleophilic substitution reaction.
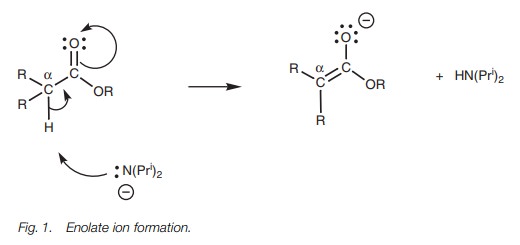
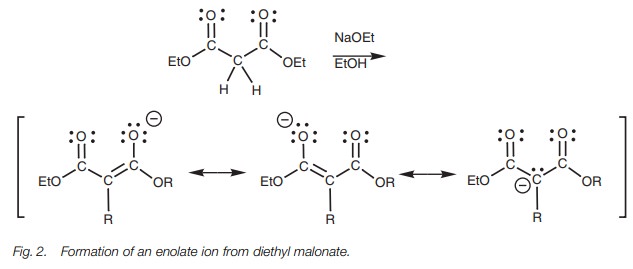
Formation of enolate ions is easier if there
are two esters flanking the α−carbon since the α−proton will be more acidic. The acidic proton in diethyl malonate
can be removed with a weaker base than LDA (e.g. sodium ethoxide; Fig. 2). The enolate ion is more stable
since the charge can be delocalized over both carbonyl groups.
Alkylations
Enolate ions can be alkylated with alkyl
halides through the SN2 nucleophilic substitution of an alkyl
halide.
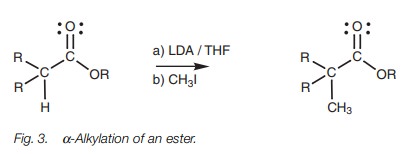
Although simple esters can be converted to
their enolate ions and alkylated, the use of a molecule such as diethyl
malonate is far more effective. This is because the α−protons of diethyl malonate (pKa
10–12) are more acidic than the α−protons of a simple ester such as ethyl acetate
(pKa 25) and can be
removed by a milder base. It is possible to predict the base required to carry
out the deprotonation reaction by considering the pKa value of the conjugate acid for that base. If this pKa is higher than the pKa value of the ester, then
the deprotonation reaction is possible. For example, the conjugate acid of the
ethoxide ion is ethanol (pKa
16) and so any ester having a pKa
less than 16 will be deprotonated by the ethoxide ion. Therefore, diethyl
malonate is deprotonated but not ethyl acetate. A further point worth noting is
that the ethoxide ion is strong enough to deprotonate the diethyl malonate
quantita-tively such that all the diethyl malonate is converted to the enolate
ion. This prevents the possibility of any competing Claisen reaction (see
below) since that reaction requires the presence of unaltered ester. Diethyl
malonate can be converted quantitatively to its enolate with ethoxide ion,
alkylated with an alkyl halide, treated with another equivalent of base, then
alkylated with a second different alkyl halide (Fig. 4). Subsequent hydrolysis and decarboxyla-tion of the diethyl
ester results in the formation of the carboxylic acid. The decarboxylation
mechanism (Fig. 5) is dependent on
the presence of the other carbonyl group at the β−position.
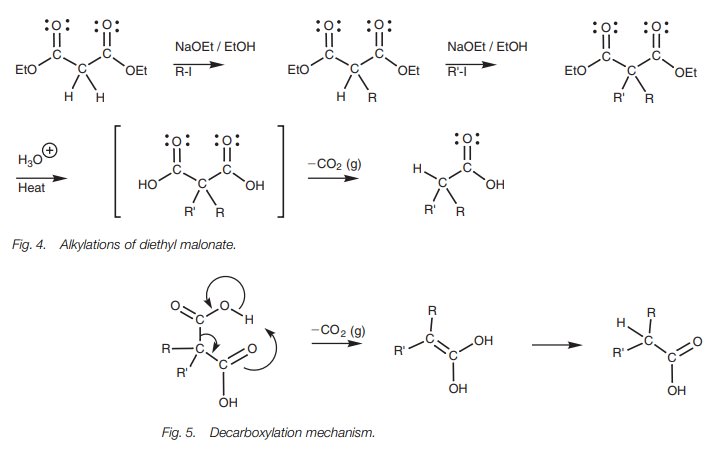
The final product can be viewed as a
disubstituted ethanoic acid. In theory, this product could also be synthesized
from ethyl ethanoate. However, the use
of diethyl malonate is superior since the presence of two carbonyl groups
allows easier formation of the intermediate enolate ions.
Claisen condensation
The Claisen reaction involves the condensation
or linking of two ester molecules to form a β-ketoester (Fig. 6). This
reaction can be viewed as the ester equivalent of the Aldol reaction. The
reaction involves the formation of an enolate ion from one ester molecule,
which then undergoes nucleophilic substitution with a second ester molecule (Fig. 7, Step 1). The ethoxide ion which
is formed in step 2 removes anα−proton from the β−ketoester in step 3 to form a stable enolate ion and this drives
the reaction to completion. The final product is isolated by protonating the
enolate ion with acid. Two different esters can be used in the Claisen
condensation as long as one of the esters has no α−protons and cannot form an enolate ion (Fig. 8). β−Diketones can be synthesized from the mixed
Claisen condensation of a ketone with an ester (Fig. 9). Again, it is advisable to use an ester which cannot form
an enolate ion to prevent competing Claisen condensations.
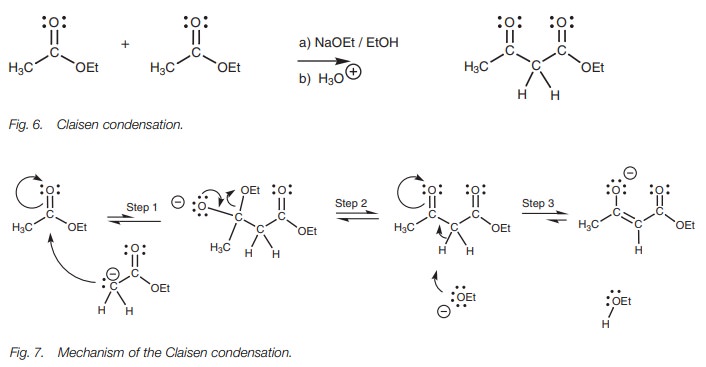
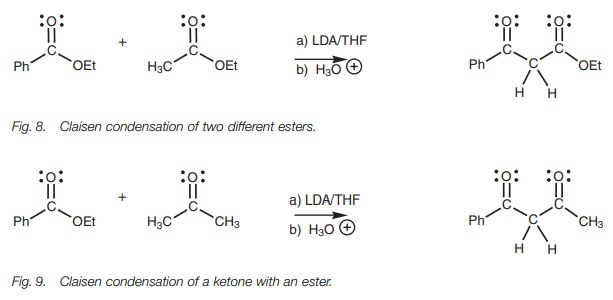
In both these last two examples, a very strong
base is used in the form of LDA such that the enolate ion is formed
quantitatively (from ethyl acetate and acetone respectively). This prevents the
possibility of self-Claisen condensation and limits the reaction to the crossed
Claisen condensation.
Related Topics