Chapter: Biochemistry: Biosynthesis of Nucleic Acids: Replication
DNA Polymerase from E. coli
DNA Polymerase from E. coli
The first DNA polymerase discovered was found in E. coli.DNA polymerase catalyzes the successive addition of each new nucleotide to the growing chain.
At least
five DNA polymerases are present in E.
coli. Three of them have been studied more extensively, and some of their
properties are listed in Table 10.1. DNA polymerase I (Pol I) was discovered
first, with the subsequent discovery of polymerases II (Pol II) and polymerase
III (Pol III). Polymerase I consists of a single polypeptide chain, but
polymerases II and III are multisubunit proteins that share some common
subunits. Polymerase II is not required for replication; rather, it is strictly
a repair enzyme. Recently, two more polymerases, Pol IV and Pol V, were discovered.
They, too, are repair enzymes, and both are involved in a unique repair
mechanism called the SOS response Two important considerations regarding the
polymerases are the speed of the synthetic reaction (turnover number) and the processivity, which is the number of
nucleotides joined before the enzyme dissociates from the template (Table
10.1).

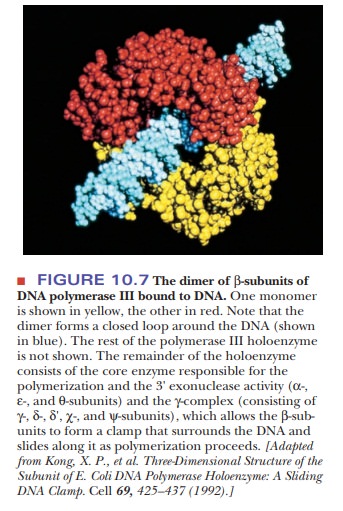
Polymerase III consists of a core enzyme responsible for the polymerization and 3' exonuclease activity-consisting of α-, ε-, and θ-subunits-and a number of other subunits, including a dimer of α-subunits responsible for DNA bind-ing, and the γ-complex-consisting of γ-, δ-, δ', χ-, and ψ-subunits-which allows the β-subunits to form a clamp that surrounds the DNA and slides along it as polymerization proceeds (Figure 10.7). Table 10.2 gives the subunit composi-tion of the DNA polymerase III complex. All of these polymerases add nucleo-tides to a growing polynucleotide chain but have different roles in the overall replication process. As can be seen in Table 10.1, DNA polymerase III has the highest turnover number and a huge processivity compared to polymerases I and II.
If DNA
polymerases are added to a single-stranded DNA template with all the
deoxynucleotide triphosphates necessary to make a strand of DNA, no reaction
occurs. It was discovered that DNA polymerases cannot catalyze de novo
synthesis. All three enzymes require the presence of a primer, a short oligonucleotide strand to which the growing
polynucleotide chain is covalently attached in the early stages of replication.
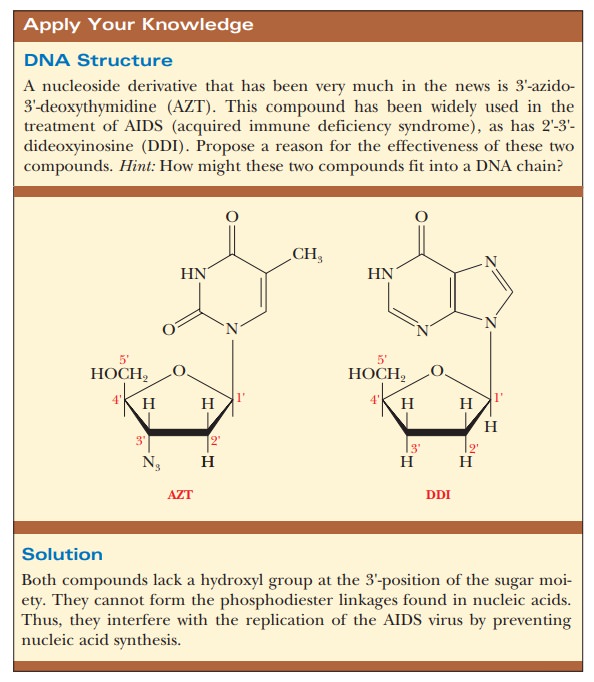
In essence, DNA polymerases must have a nucleotide with a free 3'-hydroxyl
already in place so that they can add the first nucleotide as part of the
growing chain. In natural replication, this primer is RNA.
DNA
polymerase reaction requires all four deoxyribonucleoside triphos-phates-dTTP,
dATP, dGTP, and dCTP. Mg2+ and a DNA template are also
necessary. Because of the requirement for an RNA primer, all four
ribonucleo-side triphosphates-ATP, UTP, GTP, and CTP-are needed as well; they
are incorporated into the primer. The primer (RNA) is hydrogen-bonded to the
template (DNA); the primer provides a stable framework on which the nascent chain
can start to grow. The newly synthesized DNA strand begins to grow by forming a
covalent linkage to the free 3'-hydroxyl group of the primer.
It is
now known that DNA polymerase I has a specialized function in replication-repairing
and “patching” DNA-and that DNA polymerase III is the enzyme primarily
responsible for the polymerization of the newly formed DNA strand. The major
function of DNA polymerases II, IV, and V is as repair enzymes. The exonuclease
activities listed in Table 10.1 are part of the proofreading-and-repair
functions of DNA polymerases, a process by which incorrect nucleotides are
removed from the polynucleotide so that the cor-rect nucleotides can be
incorporated. The 3' - > 5' exonuclease activity, which all three
polymerases possess, is part of the proofreading
function; incorrect nucleotides are removed in the course of replication and
are replaced by the correct ones. Proofreading is done one nucleotide at a
time. The 5' - > 3' exo-nuclease activity clears away short stretches of nucleotides
during repair, usu-ally involving
several nucleotides at a time. This is also how the RNA primers are removed.
The proofreading-and-repair function is less effective in some DNA polymerases.
Related Topics