Chapter: Biotechnology Applying the Genetic Revolution: Aging and Apoptosis
Control of Apoptotic Pathways in Development
CONTROL
OF APOPTOTIC PATHWAYS IN DEVELOPMENT
Although controlling the onset of
apoptosis is very complex, the ramifications of losing control are dire. Too
much apoptosis or inappropriate activation of apoptosis can destroy fully
functioning cells. Death of too much tissue can kill a developing organism. Not
enough apoptosis, especially during development, can create surplus tissue and
disrupt the normal operation of tissues and organisms. Many disease states may
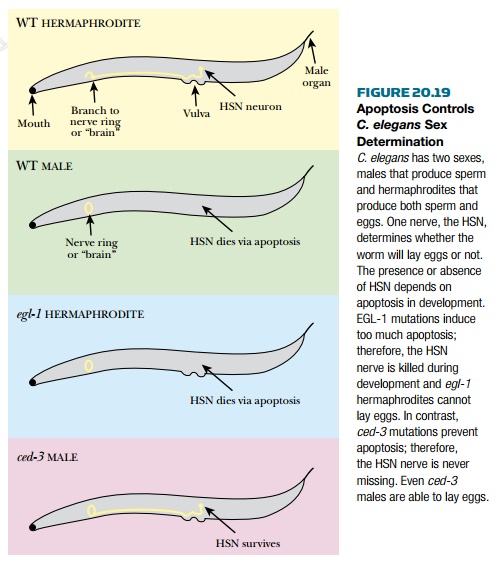
arise from inappropriate or defective apoptosis. In C. elegans sex
development depends on apoptosis. C. elegans comes in two “sexes,”
males, which produce only sperm, and hermaphrodites, which produce both sperm
and eggs. No true females are produced. The decision to become male or
hermaphrodite hinges on apoptosis ( Fig. 20.19 ). Two neurons control the
muscles around the vulva so that eggs can be laid. If the neurons are present,
the worm is a hermaphrodite. If the neurons are absent, the worm cannot lay
eggs, essentially making it a male. The presence or absence of these
hermaphrodite-specific neurons (HSN) depends on apoptosis. Defects in ced-3 or
elg-1 affect whether the worm is male or hermaphrodite. Without elg-1
, too much apoptosis occurs; therefore, all worms are missing the HSN and
no egg-laying worms are produced. Conversely, without ced-3 , apoptosis
cannot occur and the HSN survive in all worms.
Apoptosis is tightly regulated during an
immune response. During infection, the body responds to the attack by increasing
the number of white cells of the immune system. When the infection is past, the
body eliminates the surplus immune cells via apoptosis. Immune cells use the
death receptor pathway to trigger apoptosis (see earlier discussion). Too much
apoptosis would deplete our immune system of essential cells and disable the
immune response. During HIV infection, the number of T-cells plummets to
dangerously low levels, leaving the patient open to many secondary infections.
One theory is that HIV kills T cells by inducing apoptosis.
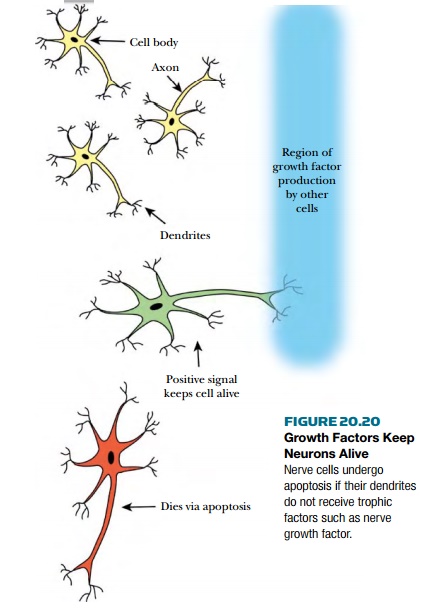
The nervous system is another tissue
that is highly sensitive to apoptosis. During development a large number of
neuronsundergo apoptosis. One theory suggests that neurons die if they do not
receive a “keep on living” signal or trophic factor . If a developing neuron reaches
its correct destination, it will receive the trophic factor. If the neuron
fails to reach its target it gets no trophic factor and enters apoptosis by default.
If neurons are cultured in a laboratory dish, removal of one trophic factor, nerve
growth factor , induces the cells to undergo apoptosis ( Fig. 20.20 ). Addition
of caspase inhibitors blocks cell death, proving the cells were dying via
apoptosis. During mouse development, embryos with defective genes for either
caspase-3 or caspase-9 die. Lack of apoptosis in the developing neural system
is the main cause for death in both cases. In the adult brain, apoptosis of
neurons causes irreparable damage because neurons do not regenerate. Extensive
apoptosis may play a role in manydiseases, such as Alzheimer’s (see later
discussion), Parkinson’s disease, Huntington’s disease, and amyotrophic lateral
sclerosis (ALS). The exact role of apoptosis in these diseases is still being
investigated.
Related Topics