Chapter: Biochemistry: Integration of Metabolism: Cellular Signaling
Calcium Ion as a Second Messenger
Calcium Ion as a Second Messenger
Calcium
ion (Ca2+) is involved in another ubiquitous second-messenger
scheme. Much of the calcium-mediated response depends on release of Ca2+
from intracellular reservoirs, similar to the release of Ca2+ from
the sarcoplasmic reticulum in the action of the neuromuscular junction. A
component of the inner layer of the phospholipid bilayer, phosphatidylinositol 4,5-bisphosphate
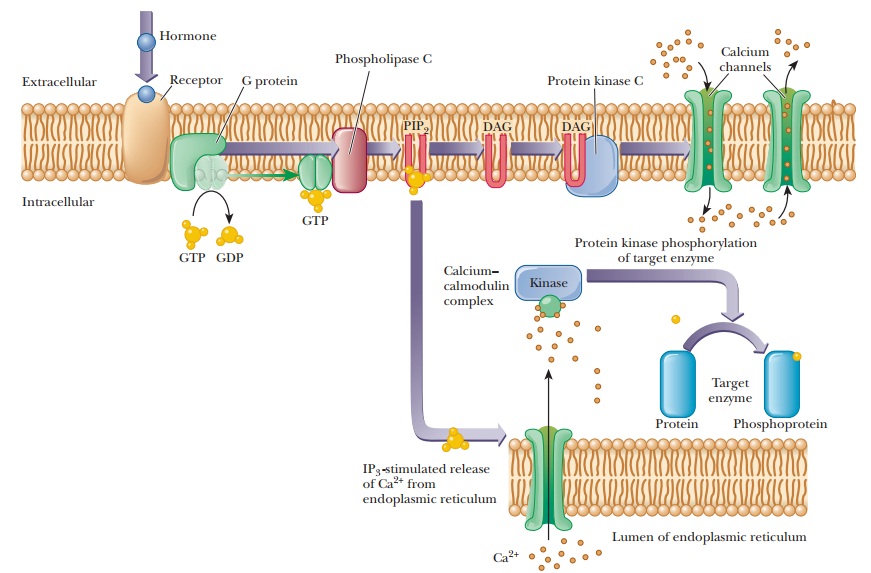
(PIP2), is also required in this scheme (Figure 24.12).
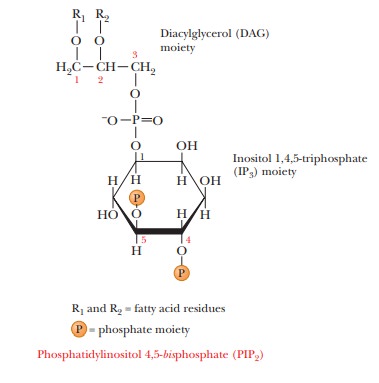
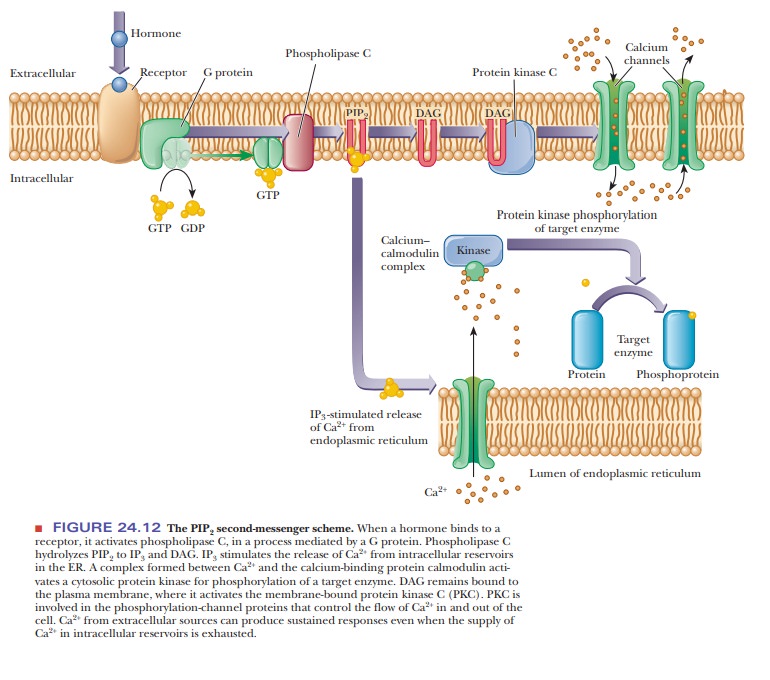
When the external trigger binds to its receptor on the cell membrane, it activates phospholipase C, which hydrolyzes PIP2 to inositol 1,4,5-triphosphate (IP3) and a diacylglycerol (DAG), in a process mediated by a differentmember of the family of G proteins. The IP3 is the actual second messenger. It diffuses through the cytosol to the endoplasmic reticulum (ER), where it stimulates the release of Ca2+. A complex is formed between the calcium-binding protein calmodulin and Ca2+. This calcium–calmodulin complex activates a cyto-solic protein kinase, which phosphorylates target enzymes in the same fashion as in the cAMP second-messenger scheme. DAG also plays a role in this scheme; it is nonpolar and diffuses through the plasma membrane. When DAG encoun-ters the membrane-bound protein kinase C, it too acts as a second messenger by activating this enzyme (actually a family of enzymes).
Protein kinase C also phosphorylates target enzymes, including channel proteins that control the flow of Ca2+ into and out of the cell. By controlling the flow of Ca2+, this second-messenger system can produce sustained responses even when the supply of Ca2+ in the intracellular reservoirs becomes exhausted.
Related Topics