Chapter: Biochemistry: Living Cell
Buffers
Buffers
A buffer is a mixture of a weak acid and its
salt with a strong base (eg) A mixture of acetic acid and sodium acetate.
HAC + NaAC -----> Na+ +H+
+ 2AC-
where HAC = Acetic acid; NaAC = Sodium acetate.
A buffer solution is one which resists a change
in its pH value (hydrogen ion concentration) on dilution or on addition of an
acid or alkali. The process by which added H+ and OH-
ions are removed so that pH remains constant is known as buffer action.
For (eg) if alkali is added to the above
mentioned buffer it forms NaAC and no free H+ or OH- will
be available.
[Na+ + H+ + 2AC-]
+ NaOH ------> 2NaAC + H2O
If an acid is added to the buffer it will form
NaCl and no free H+ or OH- will be available.
[Na+ + H+ + 2AC-]
+ HCl --------> NaCl + 2 HAC
In either cases there is no change in hydrogen
ion concentration i.e. it resist the change in pH of the solution
Examples of buffer –
A mixture of
·
Glycine
and HCl
·
Potassium
dihydrogen phosphate and dipotassium hydrogen phosphate.
·
Sodium
bicarbonate and sodium carbonate.
Uses of buffer
·
Bufers
are used for preparing standard solutions in which it is always desired to
maintain a constant pH.
·
These
are used to maintain H+ concentration which is necessary for optimal activity
of enzymes.
·
Buffers
regulate acid-base balance by restricting pH change in body fluids and tissues,
since they are capable of absorbing H+ ions and OH- ions when an acid or an
alkali is formed during metabolic activities.
Buffers of blood
The important buffers present in blood are
·
Bicarbonate
buffer
·
Phosphate
buffer
·
Protein
buffer
·
Hemoglobin
buffer
Bicarbonate buffer
It is the most important buffer in blood plasma
and consist of bicarbonate [HCO3-] and carbonic acid [H2CO3]
This buffer is efficient in maintaining the pH of blood plasma to 7.4 against
the acids produced in tissue metabolism (eg) phosphoric acid, lactic acid,
aceto acetic acid and b-hydroxy butyric acid. These acids are
converted to their anions and the bicarbonate is converted to carbonic acid a weak acid.
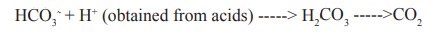
CO2 thus formed is expirated through
lungs during respiration.
Phosphate buffer
The phosphate buffer consists of dibasic
phosphate [HPO42-] and monobasic phosphate (H2PO4-).
Its pKa value is about 6.8. It is more effective in the pH range 5.8 to 7.8.
Plasma has a ratio of 4 between [HPO42-] : [H2PO4-].
Therefore pH = pKa + log { [HPO42-] /[H2PO4-]
}
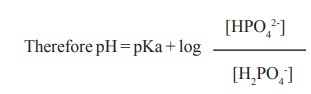
pH = 6.8 + log 4 = 7.4 [7.4
is the normal pH of blood]
Protein buffer
The protein buffers are very important in the
plasma and in the intracellular fluids but their concentration is very low in
CSF, lymph and interstitial fluids.
They exist as anions serving as conjugate bases
(Pr-) at the blood pH 7.4 and form conjugate acids (HPr) accepting H+. They
have the capacity to buffer some H2CO3 in the blood.
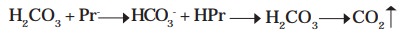
H2CO3 + Pr- -- > HCO3- + HPr - - > H2CO3 - - > CO2
Hemoglobin buffer
They are involved in buffering CO2
inside erythrocytes. The buffering capacity of hemoglobin depends on its
oxygenation and deoxygenation. Inside the erythrocytes, CO2 combines
with H2O to form H2CO3 under the action of
carbonic anhydrase. At the blood pH 7.4, H2CO3
dissociates into H+ and HCO3- and needs immediate buffering.
Oxyhemoglobin (HbO2-) on the other side loses O2 to form
deoxyhemoglobin (Hb-) which remains undissociated (HHb) by accepting H+ from
the ionization of H2CO3. Thus, Hb- buffers H2CO3
in erythrocytes.
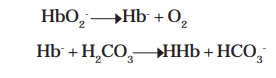
Related Topics