Chapter: Clinical Anesthesiology: Perioperative & Critical Care Medicine: Anesthetic Complications
Anesthetic Complications: Allergic Reactions
ALLERGIC REACTIONS
Hypersensitivity (or allergic) reactions are
exagger-ated immunological responses to antigenic stimula-tion in previously
sensitized persons. The antigen, or
allergen, may be a protein, polypeptide, or smaller molecule. Moreover,
the allergen may be the sub-stance itself, a metabolite, or a breakdown
product. Patients may be exposed to antigens through the respiratory tract,
gastrointestinal tract, eyes, skin and from previous intravenous,
intramuscular, or peritoneal exposure.
Anaphylaxis occurs when inflammatory agents
are released from basophils and mast cells as a result of an antigen
interacting with the immunoglobulin (Ig) E. Anaphylactoid reactions manifest
themselves in the same manner as anaphylactic reactions, but are not the result
of an interaction with IgE. Direct activation of complement and IgG-mediated
com-plement activation can result in similar inflamma-tory mediator release and
activity.
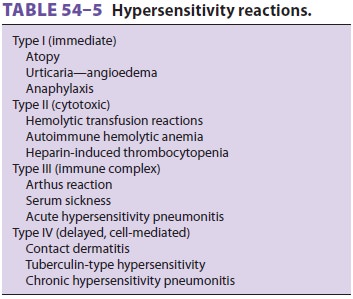
Depending on the antigen and the immune
sys-tem components involved, hypersensitivity reactions are classically divided
into four types (Table 54–5).
In many cases, an allergen (eg, latex) may cause more than one type of
hypersensitivity reaction. Type I reactions involve antigens that cross-link
IgE antibodies, triggering the release of inflamma-tory mediators from mast
cells. In type II reactions, complement-fixing (C1-binding) IgG antibodies bind
to antigens on cell surfaces, activating the classic complement pathway and
lysing the cells. Examples of type II reactions include hemolytic transfusion
reactions and heparin-induced thrombocytopenia. Type III reactions occur when
antigen–antibody(IgG or IgM) immune complexes are deposited in tissues,
activating complement and generating che-motactic factors that attract
neutrophils to the area. The activated neutrophils cause tissue injury by
releasing lysosomal enzymes and toxic products. Type III reactions include
serum sickness reactions and acute hypersensitivity pneumonitis. Type IV
reactions, often referred to as delayed hypersensitiv-ity reactions, are
mediated by CD4 + T lymphocytes that have been sensitized to a specific antigen by prior
exposure. Prior TH1 response causes expres-sion of a T-cell receptor protein
that is specific for the antigen. Reexposure to the antigen causes these
lymphocytes to produce lymphokines—interleukins (IL), interferon (IFN), and
tumor necrosis factor-γ (TNF-γ)—that attract and
activate inflammatory mononuclear cells over 48–72 hr. Production of IL-1 and
IL-6 by antigen-processing cells amplifies clonal expression of the specific
sensitized T cells and attracts other types of T cells. IL-2 secretion
trans-forms CD8+
cytotoxic T cells into killer cells; IL-4 and IFN-γ cause macrophages to undergo epithe-lioid transformation, often
producing granuloma. Examples of type IV reactions are those associated with
tuberculosis, histoplasmosis, schistosomiasis, and hypersensitivity pneumonitis
and some autoim-mune disorders, such as rheumatoid arthritis and Wegener’s
granulomatosis.
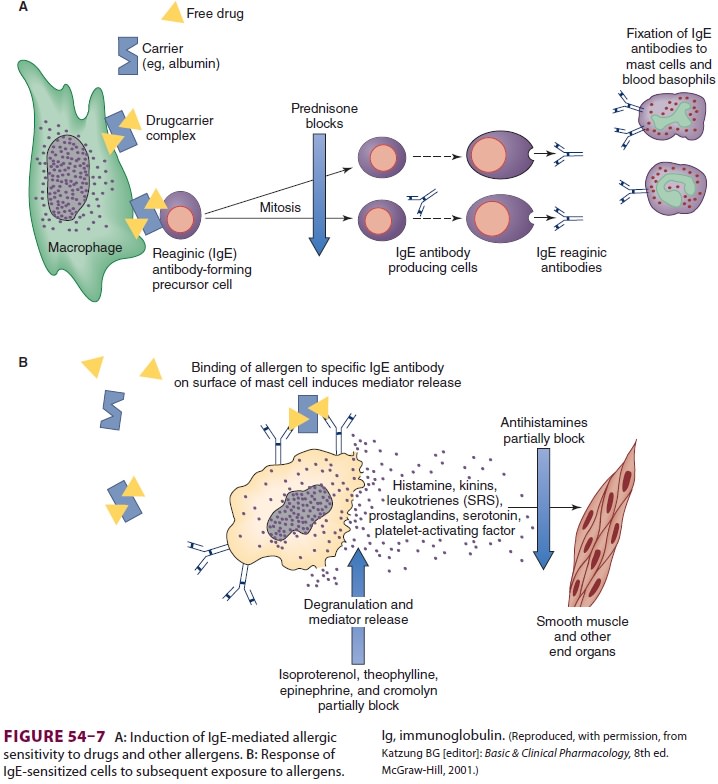
1.Immediate Hypersensitivity Reactions
Initial exposure of a susceptible person to
an antigen induces CD4+ T cells to lymphokines that activateand transform specific B
lymphocytes into plasma cells, producing allergen-specific IgE antibodies (Figure
54–7). The Fc portion of these antibodies then
associates with high affinity receptors on the cell surface of tissue mast
cells and circulating baso-phils. During subsequent reexposure to the antigen,
it binds the Fab portion of adjacent IgE antibodies on the mast cell surface,
inducing degranulation and release of inflammatory lipid mediators and
addi-tional cytokines from the mast cell. The end result is the release of
histamine, tryptase, proteoglycans (heparin and chondroitin sulfate), and
carboxy-peptidases. Prostaglandin (mainly prostaglandinD2) and leukotriene (B4, C4, D4,
E4,
and platelet-activating factor) synthesis is also increased. The combined
effects of these mediators can produce arteriolar vasodilatation, increased
vascular perme-ability, increased mucus secretion, smooth muscle contraction,
and other clinical manifestations of type I reactions.
Type I hypersensitivity reactions are classified as atopic or nonatopic.
Atopic disorders typically affect the skin or respiratory tract and include
aller-gic rhinitis, atopic dermatitis, and allergic asthma. Nonatopic
hypersensitivity disorders include urti-caria, angioedema, and anaphylaxis;
when these reactions are mild, they are confined to the skin (urticaria) or
subcutaneous tissue (angioedema), but when they are severe, they become
generalized and a life-threatening medical emergency (ana-phylaxis). Urticarial
lesions are characteristically well-circumscribed skin wheals with raised
ery-thematous borders and blanched centers; they are intensely pruritic.
Angioedema presents as deep, nonpitting cutaneous edema from marked
vasodi-latation and increased permeability of subcutaneous blood vessels. When
angioedema is extensive, it can be associated with large fluid shifts; when it
involves the pharyngeal or laryngeal mucosa, it can rapidly compromise the
airway.
2. Anaphylactic Reactions
Anaphylaxis isanexaggerated response to anallergen (eg, antibiotic) that is mediated by a type hypersensitivity reaction. The syndrome appears within minutes of exposure to a specific antigen in a sensitized person and characteristically presents as acute respiratory distress, circulatory shock, or both. Death may occur from asphyxiation or irreversible circulatory shock. The incidence of anaphylactic reactions during anesthesia has been estimated at a rate of 1:3500 to 1:20000 anesthetics. Mortality from anaphylaxis can be as frequent as 4% of cases with brain injury, occurring in another 2% of surviving patients. A French study evaluating 789 anaphylactic and anaphylactoid reactions reported that the most common sources of perioperative anaphylaxis were neuromuscular blockers (58%), latex (17%), and antibiotics (15%).
The most important mediators of anaphylaxis
are histamine, leukotrienes, basophil kallikrein (BK-A,) and
platelet-activating factor. They increase vascular permeability and contract
smooth muscle. H1-receptor activation contracts bronchial smooth muscle, whereas H2-receptor activation causesvasodilatation,
enhanced mucus secretion, tachycar-dia, and increased myocardial contractility.
BK-A cleaves bradykinin from kininogen; bradykinin increases vascular
permeability and vasodilatation and contracts smooth muscle. Activation of
Hage-man factor can initiate intravascular coagulation.
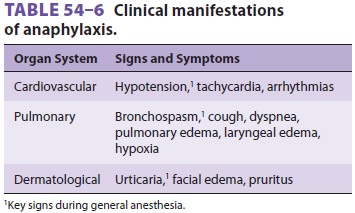
Eosinophil chemotactic factor of anaphylaxis,
neutrophil chemotactic factor, and leukotriene B4 attract inflammatory cells
that mediate additional tissueinjury. Angioedema of the pharynx, larynx, and
trachea produce upper airway obstruction, whereas bronchospasm and mucosal
edema result in lower airway obstruction. Histamine may preferentially
constrict large airways, whereas leukotrienes pri-marily affect smaller
peripheral airways. Transuda-tion of fluid into the skin (angioedema) and
viscera produces hypovolemia and shock, whereas arte-riolar vasodilatation
decreases systemic vascular resistance. Coronary hypoperfusion and arterial
hypoxemia promote arrhythmias and myocardial ischemia. Leukotriene and prostaglandin
media-tors may also cause coronary vasospasm. Prolonged circulatory shock leads
to progressive lactic acido-sis and ischemic damage to vital organs. Table
54–6 summarizes important manifestations of anaphylac-tic reactions.
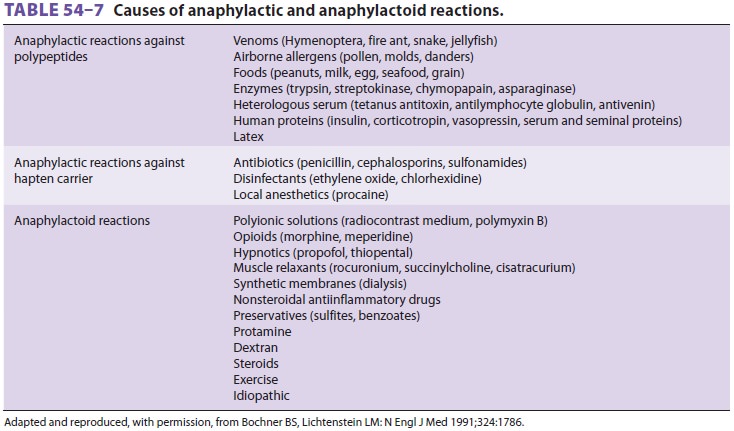
Anaphylactoid reactions resemble anaphylaxis
but do not depend on IgE antibody interaction with antigen. A drug can directly
release histamine from mast cells (eg, urticaria following high-dose morphine sulfate) or activate complement. Despite differing
mechanisms, anaphylactic and anaphylactoid reactions typically are clinically
indistin-guishable and equally life-threatening. Table 54–7 lists common causes of anaphylactic and anaphylac-toid reactions.
Factors that may predispose patients to these
reactions include pregnancy, known atopy, and pre-vious drug exposure. Such
reactions are more com-mon in younger than older patients. Laboratory
identification of patients who have experienced an adverse allergic reaction or
who may be particularly

susceptible is often aided by intradermal
skin testing, leukocyte or basophil degranulation testing (hista-mine release
test), or radio-allergosorbent testing (RAST). The latter is capable of
measuring the level of drug-specific IgE antibody in the serum. Serum tryptase
measurement is helpful in confirming the diagnosis of an anaphylactic reaction.
Prophylactic pretreatment with histamine receptor antagonists and
corticosteroids decreases the severity of the reaction. Treatment must be
immediate and tailored to the severity of the reaction ( Table
54–8).
3.Allergic Reactions to Anesthetic Agents
True anaphylaxis due to anesthetic agents is
rare; anaphylactoid reactions are much morecommon. Risk factors associated with
hypersensitiv-ity to anesthetics include female gender, atopic his-tory,
preexisting allergies, and previous anesthetic exposures. Muscle relaxants are
the most common cause of anaphylaxis during anesthesia, with an esti-mated
incidence of 1 in 6500 patients. They account for almost 60% of perioperative
anaphylactic reac-tions. In many instances, there was no previous exposure to
muscle relaxants. Investigators suggest that over-the-counter drugs, cosmetics,
and food products, many of which contain tertiary or quater-nary ammonium ions,
can sensitize susceptible indi-viduals. A French study found that, in decreasing
order of frequency, rocuronium, succinylcholine, and atracurium were most often
responsible; this likely reflects the propensity to cause anaphylaxis, together
with frequency of use.Although rarer, hypnotic agents can also be responsible
for some allergic reactions. The inci-dence of anaphylaxis for thiopental and
propofol is 1 in 30,000 and 1 in 60,000, respectively. Allergic reactions to
etomidate, ketamine, and benzodi-azepines are exceedingly rare. True
anaphylactic reactions due to opioids are far less common than nonimmune
histamine release. Similarly, ana-phylactic reactions to local anesthetics are
much less common than vasovagal reactions, toxic reac-tions to accidental
intravenous injections, and side effects from absorbed or intravenously
injected epi-nephrine. IgE-mediated reactions to ester-type local anesthetics,
however, are well described secondary to reaction to the metabolite,
para-aminobenzoic acid, In contrast, true anaphylaxis due to amide-type local
anesthetics is very rare; in some instances, the preservative (paraben or
methylparaben) was believed to be responsible for an apparent anaphy-lactoid
reaction to a local anesthetic. Moreover, the cross-reactivity between
amide-type local anesthet-ics seems to be low. There are no reports of
anaphy-laxis to volatile anesthetics.
4. Latex Allergy
The severity of allergic reactions to
latex-containing products ranges from mild contact dermatitis to
life-threatening anaphylaxis. Latex allergy is the second most common cause of
anaphylaxis during anesthe-sia. Most serious reactions seem to involve a direct
IgE-mediated immune response to polypeptides in natural latex, although some
cases of contact derma-titis may be due to a type IV sensitivity reaction to
chemicals introduced in the manufacturing process. Nonetheless, a relationship
between the occurrence of contact dermatitis and the probability of future
anaphylaxis has been suggested. Chronic exposure to latex and a history of
atopy increases the risk of sen-sitization. Healthcare workers and patients
undergo-ing frequent procedures with latex items (eg, repeated urinary bladder
catheterization, barium enema examinations) should therefore be considered at
increased risk. Patients with spina bifida, spinal cord injury, and
congenitalabnormalities of the genitourinary tract have an increased incidence
of latex allergy. The incidence of latex anaphylaxis in children is estimated
to be 1 in 10,000. A history of allergic symptoms to latex should be sought in
all patients during the preanes-thetic interview. Foods that cross-react with
latex include mango, kiwi, chestnut, avacado, passion fruit, and banana.
IL-18 and IL-13 single nucleotide polymor-phisms may affect the
sensitivity of individuals to latex and promote allergic responses.
Anaphylactic reactions to latex may be
con-fused with reactions to other substances (eg, drugs, blood products)
because the onset of symptoms can be delayed for more than 1 hr after initial
exposure. Treatment is the same as for other forms of anaphy-lactic reactions.
Skin-prick tests, intradermal tests, basophil histamine-release tests, and RAST
have been used to evaluate high-risk patients. Prevent-ing a reaction in
sensitized patients includes phar-macological prophylaxis and absolute
avoidance of latex. Preoperative administration of H1 and H2 histamine
antagonists and steroids may provide some protection, although their use is
controver-sial. Although most pieces of anesthetic equipment are now
latex-free, some may still contain latex (eg, gloves, tourniquets, some
ventilator bellows, intravenous injection ports, and older reusable face
masks). An allergic reaction has even been docu-mented from inhalation of latex
antigen contained within aerosolized glove powder. Manufacturers of
latex-containing medical products must label their products accordingly. Only
devices specifi-cally known not to contain latex (eg, polyvinyl or neoprene
gloves, silicone endotracheal tubes or laryngeal masks, plastic face masks) can
be used in latex-allergic patients. Rubber stoppers should be removed from drug
vials prior to use, and injec-tions should be made through plastic stopcocks,
if latex has not been eliminated from containers and injection ports.
5. Allergies to Antibiotics
Many true drug allergies in surgical patients
are due to antibiotics, mainly β-lactam antibiotics, such as penicillins and cephalosporins. Although 1%
to 4% of β-lactam administrations
result in allergic reac-tions, only 0.004% to 0.015% of these reactionsresult
in anaphylaxis. Up to 2% of the general population is allergic to penicillin,
but only 0.01% of penicillin administrations result in anaphylaxis.
Cephalosporin cross-sensitivity in patients with penicillin allergy is
estimated to be 2% to 7%, buthistory of an anaphylactic reaction to penicil-lin
increases the cross-reactivity rate up to 50%. Patients with a prior history of
an anaphylactic reaction to penicillin should therefore not receive a
cephalosporin. Although imipenem exhibits similar cross-sensitivity, aztreonam
seems to be antigen-ically distinct and reportedly does not cross-react with
other β-lactams. Sulfonamide allergy is also relatively common in surgical
patients. Sulfa drugs include sulfonamide antibiotics, furosemide,
hydro-chlorothiazide, and captopril. Fortunately, the fre-quency of
cross-reactivity among these agents is low.
Like cephalosporins, vancomycin is commonly
used for antibiotic prophylaxis in surgical patients. Unfortunately, it is
associated with adverse reac-tions. An anaphylactoid-type reaction, “red man
syndrome,” consists of intense pruritus, flushing, and erythema of the head and
upper torso, often with arterial hypotension; this syndrome seems to be related
to a rapid rate of administration more than to dose or allergy. Isolated
systemic hypoten-sion is a much more frequent side effect and seems to be
primarily mediated by histamine release, because pretreatment with H1 and H2 antihistamines can prevent hypotension, even with rapid rates of
administration.
Immunologic mechanisms are associated with other perioperative
pathologies. Transfusion-related lung injury may be secondary to the activity
of anti-bodies in the donor plasma, producing a hyper-sensitivity reaction that
results in lung infiltrates and respiratory failure. IgG antibody formation
directed at heparin–PF4 complexes results in plate-let activation, thrombosis,
and heparin-induced thrombocytopenia.
Related Topics