Chapter: Basic Concept of Biotechnology : Animal Biotechnology
Animal Biotechnology
Animal
Biotechnology
Animal
biotechnology is a broad field encompassing the polarities of fundamental and
applied research, including molecular modeling, gene manipulation, development
of diagnostics and vaccines and manipulation of tissue. It accounts for the use
of biotechnology tools, including molecular markers, stem cells, and tissue
engineering. Molecular markers are increasingly being used to identify and
select the particular genes that lead to desirable traits and it is now
possible to select superior germ plasma and disseminate it using artificial
insemination, embryo transfer and other assisted reproductive technologies.
These technologies have been used in the genetic improvement of livestock.
Transgenesis offers considerable opportunity for advances in medicine and
agriculture. In livestock, the ability to insert new genes for such economically
important characteristics as fecundity, resistance to or tolerance of other
environmental stresses would represent a major breakthrough in the breeding of
commercially superior stock. Another opportunity that transgenic technology
could provide is in the production of medically important proteins such as
insulin and clotting factors in the milk of domestic livestock. A comprehensive
evaluation of strategies for developing, testing, breeding and disseminating
transgenic livestock in the context of quantitativeimprovement of economic
traits is being done. Genetic improvement of livestock depends on access to
genetic variation and effective methods for exploiting this variation. Genetic
diversity constitutes a buffer against changes in the environment and is a key
in selection and breeding for adaptability and production in a range of
environments. Animal cell culture technology in today's scenario has become
indispensable in the field of life sciences, which provides a basis to study
regulation, proliferation, differentiation, and to perform genetic
manipulation. It requires specific technical skills to carry out successfully.
Application of tissue culture includes the study and understanding of
intracellular activity, intracellular flux, pharmacology, cell-cell
interaction, cell products, toxicology, tissue engineering, genomics, and
immunology. Knowledge acquired from these studies can be used in the biomedical
applications.
· Culture Media: The
culture medium is the most importantcomponent of the culture environment,
because it provides the necessary nutrients, growth factors, and hormones for
cell growth, as well as regulating the pH and the osmotic pressure of the
culture. Although initial cell culture experiments were performed using natural
media obtained from tissue extracts and body fluids, the need for
standardization, media quality, and increased demand led to the development of
defined media. The three basic classes of media are basal media, reduced-serum
media, and serum-free media, which differ in their requirement for
supplementation with serum.
· Media Components Balanced Salt
Solutions: A balanced saltsolution (BSS) is
composed of inorganic salts and may include sodium carbonate and, in some
cases, glucose. Commercial complete media will list which BSS formulation was
used.
Serum: Serum
is vitally important as a source of growth andadhesion factors, hormones,
lipids and minerals for the culture of cells in basal media. In addition, serum
also regulates cell membrane permeability and serves as a carrier for lipids,
enzymes, micronutrients, and trace elements into the cell. However, using serum
in media has a number of disadvantages including high cost, problems with
standardization, specificity, variability, and unwanted effects such as
stimulation or inhibition of growth and/or cellular function on certain cell
cultures. If the serum is not obtained from reputable source, contamination can
also pose a serious threat to successful cell culture experiments. Always check
new batches of serum before use. The quality and the composition can vary
greatly from batch to batch. Serum is inactivated by incubating it for 30 min
at +56oC. Originally, heating was used to inactivate complements for
immunoassays, but it may also have other, undocumented effects.
· Other Supplements: In
addition to serum, tissue extracts and digestshave traditionally been used to
supplement tissue culture media. The most common ones are amino acid
hydrolysates (from beef heart) and embryo extract (chick embryo).
· Basal Media:
The majority of cell lines grow well in basal media,which contain amino acids,
vitamins, inorganic salts, and a carbon source such as glucose, but these basal
media formulations must be further supplemented with serum.
· Reduced-Serum Media: Another
strategy to reduce the undesiredeffects of serum in cell culture experiments is
to use reduced-serum media. Reduced-serum media are basal media formulations
enriched with nutrients and animal-derived factors, which reduce the amount of
serum that is needed.
Serum-Free Media:
Serum-free media (SFM) circumvents issues withusing animal sera by replacing
the serum with appropriate nutritional and hormonal formulations. Serum-free
mediaformulations exist for many primary cultures and cell lines, including
recombinant protein producing lines of Chinese Hamster Ovary (CHO), various
hybridoma cell lines, the insect lines Sf9 and Sf21 (Spodopterafrugiperda), and for cell lines that act as hosts for
viral production (e.g., 293, VERO, MDCK, MDBK), and others. One of the major
advantages of using serum-free media is the ability to make the medium
selective for specific cell types by choosing the appropriate combination of
growth factors. Using serum in a medium has a number of disadvantages: the
physiological variability, the shelf life and consistency, the quality control,
the specificity, the availability, the downstream processing, the possibility
of contamination, the growth inhibitors, the standardization and the costs.
Using serum-free media and defined media supplements (Nutridoma-CS,
Nutridoma-SP and Transferrin) offers three main advantages: The ability to make
a medium selective for a particular cell type. The possibility of switching
from growth-enhancing medium for propagation to a differentiation-inducing
medium.The possibility of bioassays (e.g., protein production) free from
interference with serum proteins (easier downstream processing).
Media Recommendations: Many
continuous mammalian cell linescan be maintained on a relatively simple medium
such as MEM supplemented with serum, and a culture grown in MEM can probably be
just as easily grown in DMEM or Medium 199. However, when a specialized
function is expressed, a more complex medium may be required. Information for
selecting the appropriate medium for a given cell type is usually available in
published literature, and may also be obtained from the source of the cells or
cell banks. If there is no information available on the appropriate medium for
your cell type, choose the growth medium and serum empirically or test several
different media for best results. In general, a good placeto start is MEM for
adherent cells and RPMI-1640 for suspension cells. The conditions listed below
(Table 1) can be used as a guide line when setting up a new mammalian cell
culture. Insect cells are cultured in growth media that are usually more acidic
that those used for mammalian cells such as TNM-FH and Grace’s medium.
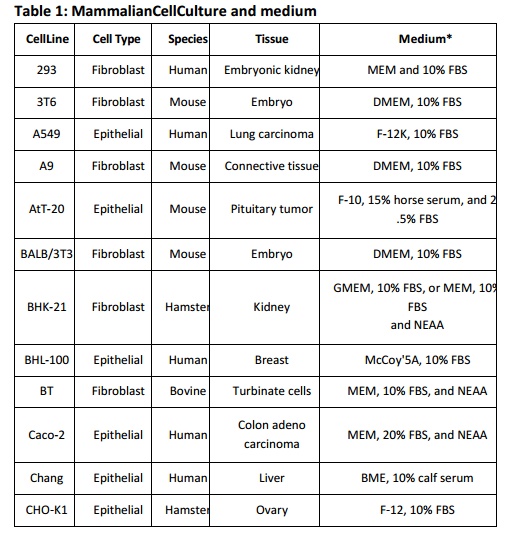
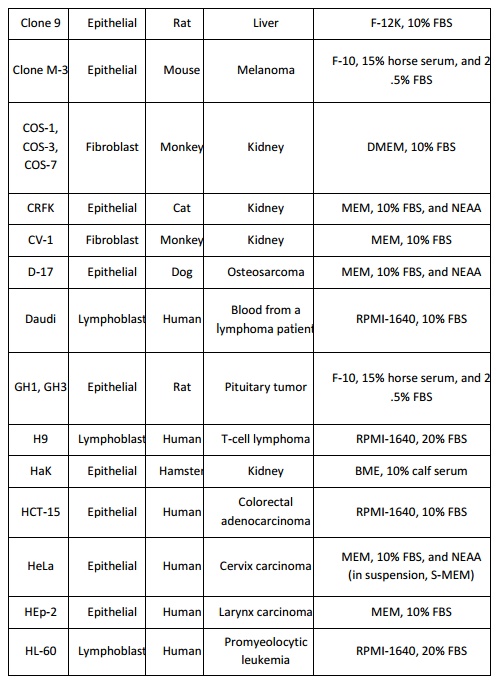
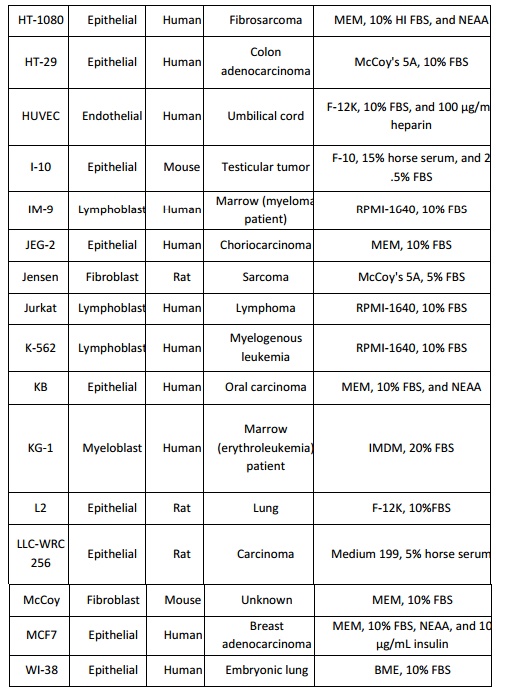

BME:
Basal Medium Eagle; DMEM:
Dulbecco’sModified EagleMedium; FBS:
Fetal Bovine Serum; GMEM: Glasgow
Minimum Essential Medium; IMDM:
Iscove’s Modified Dulbecco’s Medium; MEM:
Minimum Essential Medium; NEAA:
Non-Essential AminoAcids Solution; TNM-FH:Trichoplusiani Medium-Formulation Hink
(i.e. Grace’s Insect Medium, Supplemented)
Related Topics