behaviour, Limitations | Chemistry - Werner's theory of coordination compounds | 12th Chemistry : UNIT 5 : Coordination Chemistry
Chapter: 12th Chemistry : UNIT 5 : Coordination Chemistry
Werner's theory of coordination compounds
Werner's
theory of coordination compounds:
Swiss chemist Alfred
Werner was the first one to propose a theory of coordination compounds to
explain the observed behaviour of them.
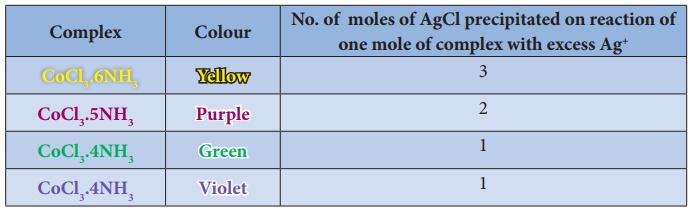
Let us consider the
different coloured complexes of cobalt(III) chloride with ammonia which exhibit
different properties as shown below.
In this case, the
valences of the elements present in both the reacting molecules, cobalt(III)
chloride and ammonia are completely satisfied. Yet these substances react to
form the above mentioned complexes.
To explain this behaviour Werner postulated his theory as follows
1. Most of the elements
exhibit, two types of valence namely primary valence and secondary valence and
each element tend to satisfy both the valences.In modern terminology, the
primary valence is referred as the oxidation state of the metal atom and the
secondary valence as the coordination number. For example, according to Werner,
the primary and secondary valences of cobalt are 3 and 6 respectively.
2. The primary valence of a metal ion is positive in most of the
cases and zero in certain cases. They are always satisfied by negative ions.
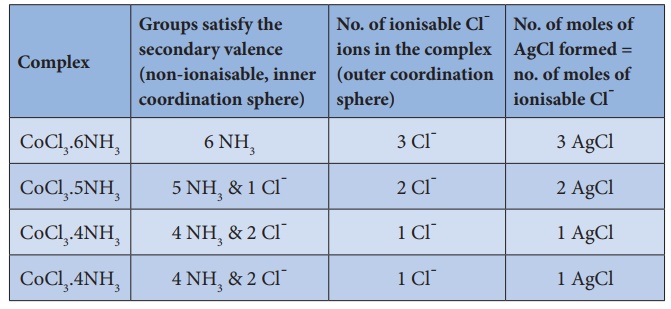
For example in the complex CoCl3.6NH3, The primary
valence of Co is +3 and is satisfied by 3Cl- ions.
3. The secondary valence
is satisfied by negative ions, neutral molecules, positive ions or the
combination of these. For example, in CoCl3.6NH3 the
secondary valence of cobalt is 6 and is satisfied by six neutral ammonia
molecules, whereas in CoCl3.5NH3 the secondary valence of
cobalt is satisfied by five neutral ammonia molecules and a Cl- ion.
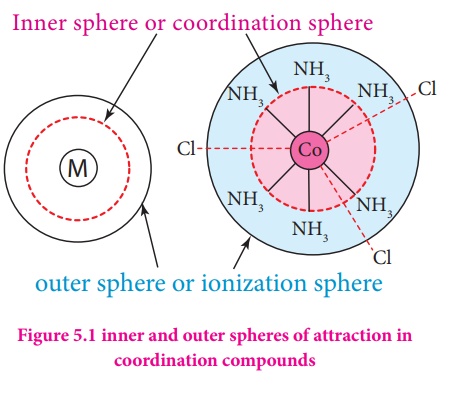
4. According to Werner,
there are two spheres of attraction around a metal atom/ion in a complex. The
inner sphere is known as coordination sphere and the groups present in this
sphere are firmly attached to the metal. The outer sphere is called ionisation
sphere. The groups present in this sphere are loosely bound to the central
metal ion and hence can be separated into ions upon dissolving the complex in a
suitable solvent.
5. The primary valences are non-directional while the secondary
valences are directional. The geometry of the complex is determined by the
spacial arrangement of the groups which satisfy the secondary valence. For
example, if a metal ion has a secondary valence of six, it has an octahedral
geometry. If the secondary valence is 4, it has either tetrahedral or square
planar geometry.
The following table
illustrates the Werner's postulates.
Limitations of Werner’s theory:
Even though, Werner’s
theory was able to explain a number of properties of coordination compounds, it
does not explain their colour and the magnetic properties.
Related Topics