Chapter: Plant Anatomy:An Applied Approach: The leaf
Vascular system in leaf

The vascular system
The specialized cells which conduct water and salts upwards from the roots, and the cells involved in the transport or translocation of the substances synthesized in the leaf mesophyll and other tissues, are grouped together in well-defined strands called vascular bundles. In the leaf, these are seen as the midrib and vein system. Vascular bundles are continuous, either directly or if developed, through the petiole, with the primary system of vascular tissue in the stem. Alternatively, if secondary growth has occurred, leaf bundles may be continuous with the secondary xylem and phloem. The vascular system in leaves may be likened to the system of tributaries which feed into a major river. The xylem system functions in reverse, with larger veins essentially supplying water and dissolved solutes to the smaller veins. The phloem, on the other hand, is the pathway through which assimilated materials are translocated. This pathway functions from the smaller to-wards the larger veins. Assimilates often take particular pathways through a series of small, intermediate and larger veins, before ultimately discharg-ing into the midvein system, which connects to the phloem in the stem. Assimilated material usually follows a pathway from the source (where syn-thesized) to sink (where utilized). According to the source of demand, phlo-em can transport materials in either direction. Translocation in plants involves movement of water and dissolved inorganic nutrients through the xylem from roots to the aerial parts of the plant, and the transport of photo-assimilated material from sites of synthesis (source) to sites of utilization (sink) via the phloem.
The xylem is responsible for apoplastic transport in vascular plants, which is not limited totally to water transport, but in addition, the transport of various macro- and micronutrients, amino acids and other important inorganic substances, from the roots to the stem and, ultimately, the leaf via the apoplastic continuum.
The phloem is responsible for the transport of the major proportion of soluble carbohydrate as well as other essential products. The phloem forms the major long-distance symplastic transport pathway in all vascular plants. Translocation usually takes place from a site of synthesis of assimilated material (called a source) to a site or sites of utilization (called sinks). The assimilated material is translocated in a water-based medium, whichemphasizes the essential inter-relationship between the xylem and phloem, more particularly so in the leaf where most of the phloem loading takes place in mature plants.
![]()
![]() Transpiration is the driving force that facilitates the movement of solutes through the xylem. Transpiration requires that water entry is facilitated at the roots. Efficient transport must occur in a conducting system, in order that water may be moved to other regions of the plant where it is utilized in numerous biochemical and growth-related reactions. The xylem is also the principal pathway through which water is moved from point of entry, to a point of exit, which in higher plants is via stomata, by the process of transpi-ration. Transpiration itself facilitates leaf cooling by evapotranspirational heat loss to the atmosphere. Thus the physiological necessity of regulating transpirational water loss, via the cuticle, the epidermis and the stomata, have led to variations in the surface sculpturing of the cuticle, changes in the size of epidermal cells, and alteration of stomatal cells, related to the habitat preference of the particular species.
Transpiration is the driving force that facilitates the movement of solutes through the xylem. Transpiration requires that water entry is facilitated at the roots. Efficient transport must occur in a conducting system, in order that water may be moved to other regions of the plant where it is utilized in numerous biochemical and growth-related reactions. The xylem is also the principal pathway through which water is moved from point of entry, to a point of exit, which in higher plants is via stomata, by the process of transpi-ration. Transpiration itself facilitates leaf cooling by evapotranspirational heat loss to the atmosphere. Thus the physiological necessity of regulating transpirational water loss, via the cuticle, the epidermis and the stomata, have led to variations in the surface sculpturing of the cuticle, changes in the size of epidermal cells, and alteration of stomatal cells, related to the habitat preference of the particular species.
The vascular tissue within the leaf blade is arranged in a pattern that appears to be under strong genetic control. The phenotypic expression of the genotype varies very little in overall pattern, although the number of bundles may vary in the leaves of plants of any one species found growing under a range of conditions. However, the main features that characterize a particular type of venation are constant enough for use in identification of fragments. It is rare for a family or genus to have a ‘unique’ pattern, but some families can be distinguished by the constancy of a particular type. For example, Melastomataceae have a vein running parallel with the mar-gin in most species.
Vascular patterns have been classified by several different authors. The system proposed by Hickey has proved to be the most popular. Hickey (1973) published his classification system, which details a classification of the ar-chitectural features of dicot leaves. It was updated with Wolf, and presented again in Metcalf and Chalk’s Anatomy of the Dicotyledons, volume 1, 2nd edition. (1979). Hickey and Wolf’s system deals with the placement and form of those elements which contribute directly to the outward expression of leaf structure, including shape, marginal configuration, venation and gland position, and was developed from an extensive survey of both living and fos-sil leaves. This system partially incorporates modifications of two earlier classifications: that of Turrill for leaf shape and that of Von Ettingshausen for venation patterns. After categorization of such features as shape of the whole leaf and of the apex and base, leaves are separated into a number of classes depending on the course of their principal venation. According to Hickey, identification of order of venation, which is fundamental to the ap-plication of classification, is determined by size of a vein at its point of origin and to a lesser extent by its behaviour in relation to that of other orders. This classification includes a description of the areoles, i.e. the smallest areas of![]()
![]() leaf tissue surrounded by veins which form a contiguous field over most of the leaf. Perhaps the most useful aspect of this classification system lies in the fact that most dicot taxa possess consistent patterns of leaf architecture, allowing a rigorous method of describing the features of leaves, which is of immediate usefulness in both modern and fossil plant taxonomic studies. Some of the key aspects of the system are shown in Fig. 6.26.
leaf tissue surrounded by veins which form a contiguous field over most of the leaf. Perhaps the most useful aspect of this classification system lies in the fact that most dicot taxa possess consistent patterns of leaf architecture, allowing a rigorous method of describing the features of leaves, which is of immediate usefulness in both modern and fossil plant taxonomic studies. Some of the key aspects of the system are shown in Fig. 6.26.

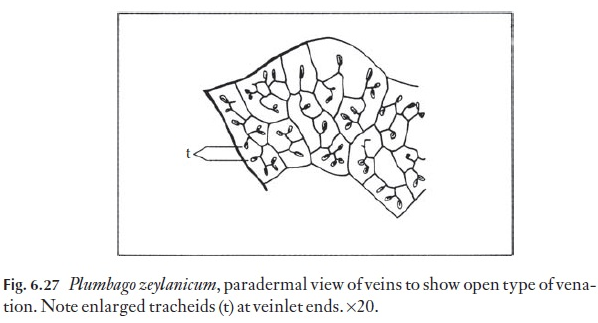
Vascular systems can be studied best in cleared material, which have sub-sequently been stained with safranin. A range of main vein systems is readi-ly observed. The angle at which the veins depart from the midrib can be a useful and relatively constant feature for a species. The nature of the vein endings of the final or smallest order of branching is also used taxo-nomically. Figure 6.27 shows the ‘open’ type of vein ending in Plumbagozeylanicum.
The commonly held idea that all monocotyledons have parallel venation is quickly dispelled by examination of such leaves as Bryonia or Smilax. In addition, monocotyledonous leaves may have large numbers of cross veins, joining the more prominent longitudinally arranged (parallel) vein net-work. These cross veins may be longer than the sum of all the parallel veins. There are also dicotyledons that do not have the so-called net-like type of venation.
In most dicotyledonous leaves (except those which are greatly reduced and needle-like, e.g. Hakea), the phloem pole in a vascular bundle faces the lower (abaxial) leaf surface and the xylem pole the adaxial surface. This is generally true for most species, which have a collateral vascular bundle ar-rangement. However, there are at least 27 families which have bicollateral vascular bundles, that is, they have phloem at either side of the xylem. Ex-amples here are to be found in the pumpkin family (Cucurbitaceae), the Solanaceae (potato, tobacco, tomato), the milkweeds (Asclepiadaceae) and others, such as the Chenopodiaceae and Apocynaceae. In mature leaves of the potato, the smaller minor veins (fifth and sixth order) may contain few living adaxial sieve tube members, whilst all the corresponding abaxial sieve tube members in the same veins are living.
Lamina vein orders
Most leaves have two components in their vascular networks – a major and minor vein system. What makes them different? Simply, the major veins in dicotyledonous foliage leaves occupy much of the cross-sectional area of the leaf, and are often associated with hypodermal collenchymatous or scleren-chymatous strands. Viewed in cross-section, they may even sometimes show signs of a cambial zone. However, this cambial zone displays only limited secondary growth, which is more evident nearer the base of the leaf and (if present) down in the petiole, where the vasculature of the main vascular supply to the leaf assumes a more cauline appearance (that is, it is more
By contrast, minor veins lack associated mechanical supporting tissue. Unlike the major network veins, the minor veins are usually em-bedded within the interface between the palisade and spongy mesophyll lay-ers. As mentioned, minor veins are embedded within a horizontally orientated mesophyll, termed the paraveinal mesophyll in some dicotyledons, which, judging by the relatively high plasmodesmatal frequencies recorded between adjacent cells, is the principal symplastic solute conduction pathway from the palisade and spongy layers, into the surrounding parenchymatous bundle-sheath cells, terminating at the sieve tubes within major and minor veins.
Major vein differentiation
The sequence of events that takes place within the dicotyledonous foliage leaf may be summarized as follows. Once the blade or lamina starts to ex-pand due to anticlinal and periclinal cell division within the marginal and submarginal initials, the procambial strands begin to form – the first of these to become evident is the midrib or main vein. This vein is blocked out acropetally, and vascular tissues differentiate in regular sequence (proto-phloem followed by protoxylem, then metaphloem followed by metaxylem) towards the tip of the still-immature expanding leaf. The major veins of the lamina follow suite, initiating at the base of the leaf, they too, differentiate acropetally. Thus the first-formed of the major lamina veins matures first, and the last-formed (apical) major veins differentiate and mature last.
The minor veins
In dicotyledonous foliage leaves, the minor veins differentiate basipetally, from the apex and the leaf margin, back towards the major vein network. Thus, it is quite feasible for the tip of the developing leaf to mature with ![]()
![]()
![]()
![]() respect to transport, before the base of the leaf. The apex could therefore conceivably export photoassimilated material to the still immature basal part of the leaf, during the overall maturation and development process.
respect to transport, before the base of the leaf. The apex could therefore conceivably export photoassimilated material to the still immature basal part of the leaf, during the overall maturation and development process.
Related Topics