Chapter: 10th Science : Chapter 10 : Types of Chemical Reactions
Types of Chemical Reactions
TYPES OF CHEMICAL
REACTIONS
1. Classification based on the nature of rearrangements of atoms
So far you studied about
a chemical reaction and how it can be described as a chemical equation. A large
number of chemical reactions are taking place around us every day.Are they
taking place in a similar way? No. Each reaction involves different kinds of
atoms and hence the way they react also differs. Thus, based on the manner by
which the atoms of the reactants are rearranged, chemical reactions are
classified as follows.
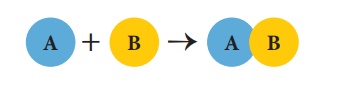
(a) Combination reactions
A combination reaction
is a reaction in which two or more reactants combine to form a compound. It is
otherwise called 'synthesis reaction' or 'composition reaction'. When a
reactant ‘A’ combines with ‘B’, it forms the product ‘AB’. The generalised
scheme of a combination reaction is given below:
Example: Hydrogen gas combines
with chlorine gas to form hydrogen chloride gas.
H2(g) + Cl2(g)
→ 2HCl(g)
Depending on the
chemical nature of the reactants, there are three classes of combination
reactions:
Element + Element → Compound
In this type of
combination reaction, two elements react with one other to form a compound. The
reaction may take place between a metal and a non-metal or two non-metals.
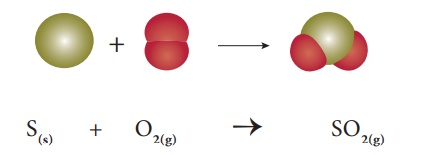
Example 1: When solid sulphur
reacts
with oxygen, it produces
sulphur dioxide. Here both the reactants are non-metals.
Example 2: Sodium, a silvery-white metal, combines with
chlorine, a pale yellow green gas, to form sodium chloride, an edible compound.
Here one of the reactants is a metal (sodium) and the other (chlorine) is a
non-metal.
2Na(s) + Cl2(g)
→ 2NaCl(s)
Test Yourself:
Identify the possible
combination reactions between the metals and non-metals given in the following
table and write their balanced chemical equations:
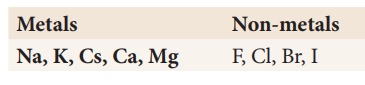
Compound + Element → Compound
In this case, a compound
reacts with an element to form a new compound. For instance, phosphorous
trichloride reacts with chlorine gas and forms phosphorous pentachloride.
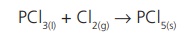
Compound + Compound → Compound
It is a reaction between
two compounds to form a new compound. In the following reaction, silicon
dioxide reacts with calcium oxide to form calcium silicate.
SiO2(s) + CaO(s)
→ CaSiO3(s)
Most of the combination
reactions are exothermic in nature. Because, they involve the formation of new
bonds, which releases a huge amount of energy in the form of heat.
(b) Decomposition reactions
In a decomposition
reaction, a single compound splits into two or more simpler substances under
suitable conditions. It is the opposite of the combination reaction. The generalised
scheme of a decomposition reaction is given below:
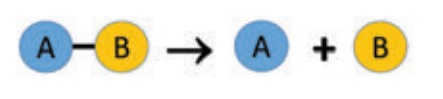
Breaking of bonds is the
major phenomenon in a decomposition reaction and hence it requires energy to
break the bonds, depending on the nature of the energy used in the
decomposition reaction.
There are three main
classes of decomposition reactions. They are
(i) Thermal Decomposition Reactions
(ii) Electrolytic Decomposition Reactions
(iii) Photo
Decomposition Reactions
(i) Thermal Decomposition Reactions
In this type of
reaction, the reactant is decomposed by applying heat. For example, on heating
mercury (II) oxide is decomposed into mercury metal and oxygen gas. As the
molecule is dissociated by the absorption of heat, it is otherwise called
‘Thermolysis’. It is a class of compound to element/element decomposition. i.e.
a compound (HgO) is decomposed into two elements (Hg and Oxygen).
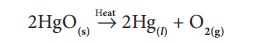
Similarly, when calcium
carbonate is heated, it breaks down in to calcium oxide and carbon dioxide. It
is a type of compound to compound/compound decomposition.
CaCO3(s)
Heat→
CaO(s) + CO2(g)
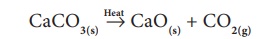
In thermal decomposition
reaction, heat is supplied to break the bonds. Such reactions, in which heat is
absorbed, are called ‘Endothermic reactions’.
(ii) Electrolytic Decomposition Reactions
In some of the decomposition
reactions, electrical energy is used to bring about the reaction. For example,
decomposition of sodium chloride occurs on passing electric current through its
aqueous solution. Sodium chloride decomposes in to metallic sodium and chlorine
gas. This process is termed as ‘Electrolysis’.
2NaCl(aq)→Electricity
→ 2Na(s) + Cl2(g)
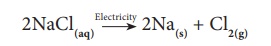
Here, a compound (NaCl)
is converted into elements (Na and chlorine). So it is a type of compound to
element/element decomposition.
(iii) Photo Decomposition Reactions
Light is an another form
of energy, which facilitates some of the decomposition reactions. For example,
when silver bromide is exposed to light, it breaks down into silver metal and
bromine gas. As the decomposition is caused by light, this kind of reaction is
also called ‘Photolysis’.
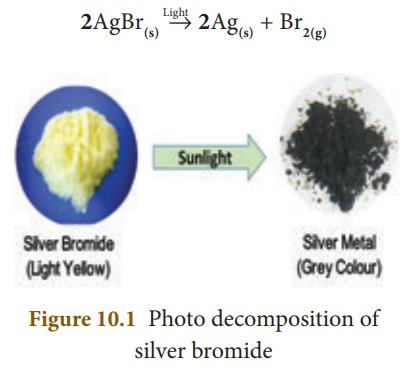
The yellow coloured
silver bromide turns into grey coloured silver metal. It is also a compound to
element/element decomposition.
(c) Single Displacement Reactions
It is a reaction between
an element and a compound. When they react, one of the elements of the
compound-reactant is replaced by the element-reactant to form a new compound
and an element. The general schematic representation of a single displacement
reaction is given as:

‘A’ displaces element
‘B’ from the compound ‘BC’ and hence a single displacement reaction occurs. If
zinc metal is placed in hydrochloric acid, hydrogen gas is evolved. Here,
hydrogen is displaced by zinc metal and zinc chloride is formed.
Zn(s) + 2HCl(aq)
→ ZnCl2(aq) + H2(g)
Fe(s) + CuSO4(aq)
→ FeSO4(aq) + Cu(s)
If an iron nail is
placed in an aqueous solution of copper (II) sulphate as shown in Fig. 10.2,
the iron displaces copper from its aqueous solution and the so formed copper
deposits over the iron nail.

It is easy to propose so
many reactions of this kind with different combinations of reactants. Will they
all occur in practice? No. This is most easily demonstrated with halogens. Let
us consider the following two reactions:
2NaCl(aq) + F2(g)
→ 2NaF(aq) + Cl2(g)
2NaF(aq) + Cl2(g)
→ 2NaCl(aq) + F2(g)
The first reaction
involves the displacement of chlorine from NaCl, by fluorine. In the second
reaction, chlorine displaces fluorine from NaF. Out of these two, the second
reaction will not occur. Because, fluorine is more active than chlorine and
occupies the upper position in the periodic table. So, in displacement
reactions, the activity of the elements and their relative position in the
periodic table are the key factors to determine the feasibility of the
reactions. More active elements readily displace less active elements from
their aqueous solution.
The activity series of
some elements is given below:
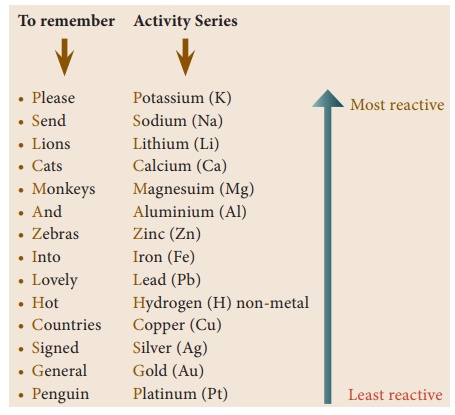
By referring the
activity series, try to answer the following questions:
Which of the metals
displaces hydrogen gas from hydrochloric acid? Silver or Zinc. Give the
chemical equation of the reaction and Justify your answer.
d) Double Displacement Reactions
When two compounds
react, if their ions are interchanged, then the reaction is called double
displacement reaction. The ion of one compound is replaced by the ion of the
another compound. Ions of identical charges are only interchanged, i.e., a
cation can be replaced by other cations. This reaction is also called ‘Metathesis
Reaction’. The schematic representation of a double displacement reaction
is given below:
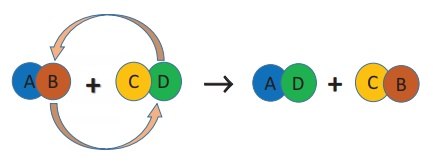
For a double
displacement reaction to take place, one of the products must be a precipitate
or water. By this way, there are major classes of double displacement
reactions. They are:
(i) Precipitation Reactions
(ii) Neutralization Reactions
(i) Precipitation Reactions
When aqueous solutions
of two compounds are mixed, if they react to form an insoluble compound and a
soluble compound, then it is called precipitation reaction. Because the
insoluble compound, formed as one of the products, is a precipitate and hence
the reaction is so called.
When the clear aqueous
solutions of potassium iodide and lead (II) nitrate are mixed, a double
displacement reaction takes place between them.
Pb(NO3)2(aq) + 2KI(aq) → PbI2(s)↓ + 2KNO3(aq)
Potassium and lead
displace or replace one other and form a yellow precipitate of lead (II) iodide
as shown in Fig. 10.3.

(ii) Neutralization Reactions
In your lower classes,
you have learned the reaction between an acid and a base. It is another type of
displacement reaction in which the acid reacts with the base to form a salt and
water. It is called 'neutralization reaction' as both acid and base neutralize
each other.
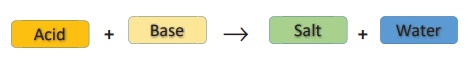
Reaction of sodium
hydroxide with hydrochloric acid is a typical neutralization reaction. Here,
sodium replaces hydrogen from hydrochloric acid forming sodium chloride, a
neutral soluble salt.
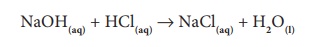
Similarly, when ammonium
hydroxide reacts with nitric acid, it forms ammonium nitrate and water.
(e) Combustion Reactions
A combustion reaction is
one in which the reactant rapidly combines with oxygen to form one or more
oxides and energy (heat). So in combustion reactions, one of the reactants must
be oxygen. Combustion reactions are majorly used as heat energy sources in many
of our day to day activities. For instance, we use LPG gas for domestic cooking
purposes. We get heat and flame from LPG gas by its combustion reaction of its
constituent gases. LPG is a mixture of hydrocarbon gases like propane, butane,
propylene, etc. All these hydrocarbons burn with oxygen to form carbon dioxide
and water.
C3H8(g)
+ 5O2(g) → 3CO2(g) + 4H2O(g)
+ Heat
Propane

Since heat is evolved,
it is an exothermic reaction. As oxygen is added, it is also an oxidation. So,
combustion may be called as an exothermic oxidation. If a flame is formed (as
shown in Fig. 10.4), then it is called burning.

Which of the following
is a combustion?
(i) Digestion of Food
(ii) Rusting of iron
Many thousands of
reactions fall under these five categories and further you will learn in detail
about these reactions in your higher classes.
2. Classification based on the direction of the reaction
You know that innumerable
changes occur every day around us. Are all they permanent? For example, liquid
water freezes into ice, but then ice melts into liquid water. In other words,
freezing is reversed. So, it is not a permanent change. Moreover, it is a
physical change. Physical changes can be reversed easily. Can chemical changes
be reversed? Can the products be converted into reactants? Let us consider the
burning of a wood. The carbon compounds present in the wood are burnt into
carbon dioxide gas and water. Can we get back the wood immediately from carbon
dioxide and water? We cannot. So, it is a permanent change. In most of the
cases, we cannot. But, some chemical reactions can be reversed. Our mobile
phone gets energy from its lithium ion battery by chemical reactions. It is
called discharging. On recharging the mobile, these chemical reactions are
reversed. Thus, chemical reactions may be reversed under suitable conditions.
Hence, they are grouped into two categories such as reversible and irreversible
reactions.

Reversible Reactions
A reversible reaction is
a reaction that can be reversed, i.e., the products can be converted back to
the reactants. A reversible reaction is represented by a double arrow with
their heads in the direction opposite to each other. Thus, a reversible
reaction can be represented by the following equation:
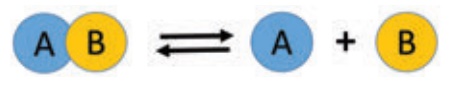
Explanation: Here, the compound ‘AB’ undergoes decomposition
to form the products ‘A’ and ‘B’. It is the forward reaction. As soon as
the products are formed, they combine together to form ‘AB’. It is the backward
reaction. So, the reaction takes place in both the directions. Do you think
then that no products are formed in the aforesaid reaction? If you think so,
you are wrong. Because, even though the reaction takes place in both the
directions, at the initial stage the rates (speed) of these reactions are not
equal. Consider the following decomposition reaction of phosphorous
pentachloride into phosphorous trichloride and chlorine.
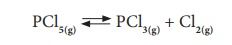
The forward reaction is
the decomposition of PCl5 and the backward reaction is the
combination of PCl3 and Cl2. Initially, the forward
reaction proceeds faster than the backward reaction. After sometimes, the speed
of both the reactions become equal. So, PCl5 cannot be completely
converted into the products as the reaction is reversed. It is a reversible
reaction. The actual measurements of the given reaction show that the reaction
is at equilibrium, but the amount of PCl5 is more than that of PCl3
and Cl2.
Thus, more amount of products can be obtained in a reversible reaction by the periodical removal of one of the products or the periodical addition of the reactants.
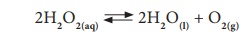
Irreversible Reactions
The reaction that cannot
be reversed is called irreversible reaction. The irreversible reactions
are unidirectional, i.e., they take place only in the forward direction.
Consider the combustion of coal into carbon dioxide and water.
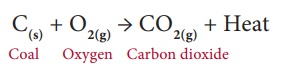
In this reaction, solid
coal burns with oxygen and gets converted into carbon dioxide gas and water. As
the product is a gas, as soon as it is formed it escapes out of the reaction
container. It is extremely hard to decompose a gas into a solid. Thus, the
backward reaction is not possible in this case. So, it is an irreversible
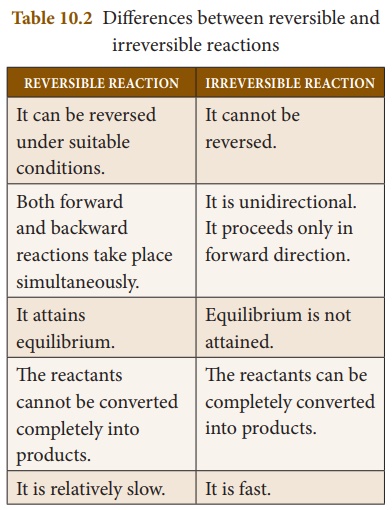
reaction. Table 10.2 provides the main differences between a reversible and an
irreversible reaction:
You will learn more
about these reactions in your higher classes.
Related Topics