Chapter: Medical Physiology: Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
Tubular Reabsorption Includes Passive and Active Mechanisms
Tubular Reabsorption Includes Passive and Active Mechanisms
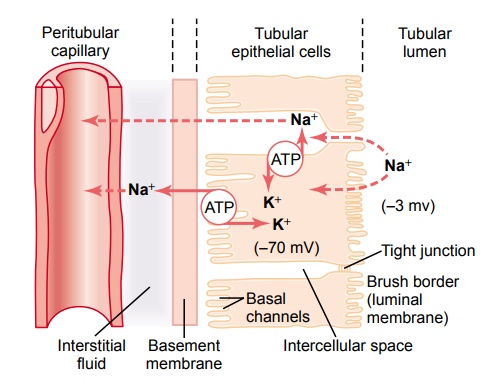
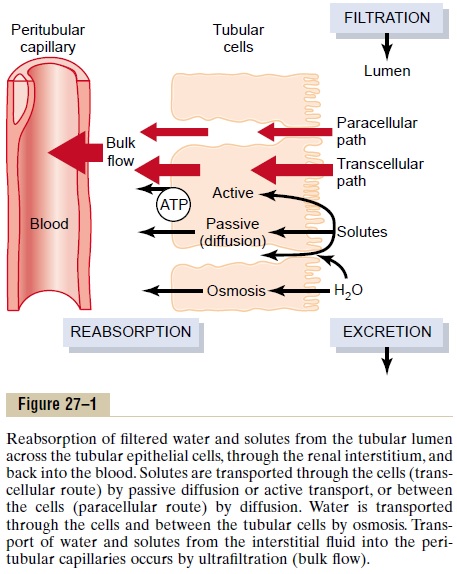
For a substance to be reabsorbed, it must first be trans-ported (1) across the tubular epithelial membranes into the renal interstitial fluid and then (2) through the peritubular capillary membrane back into the blood (Figure 27–1). Thus, reabsorption of water and solutes includes a series of transport steps. Reabsorption across the tubular epithelium into the interstitial fluid includes active or passive transport by way of the same basic mechanisms discussed for transport across other membranes of the body. For instance, water and solutes can be transported either through the cell membranes themselves (transcellular route) or through the junctional spaces between the cells (para-cellular route). Then, after absorption across thetubular epithelial cells into the interstitial fluid, water and solutes are transported the rest of the way through the peritubular capillary walls into the blood by ultra-filtration (bulk flow) that is mediated by hydrostaticand colloid osmotic forces. The peritubular capillaries behave very much like the venous ends of most other capillaries because there is a net reabsorptive force that moves the fluid and solutes from the interstitium into the blood.
Active Transport
Active transport can move a solute against an elec-trochemical gradient and requires energy derived from metabolism. Transport that is coupled directly to an energy source, such as the hydrolysis of adenosine triphosphate (ATP), is termed primary active trans-port. A good example of this is the sodium-potassium ATPase pump that functions throughout most parts of the renal tubule. Transport that is coupled indirectly to an energy source, such as that due to an ion gradient, is referred to as secondary active transport. Reabsorp-tion of glucose by the renal tubule is an example of secondary active transport. Although solutes can be reabsorbed by active and/or passive mechanisms by the tubule, water is always reabsorbed by a passive (nonactive) physical mechanism called osmosis, which means water diffusion from a region of low solute con-centration (high water concentration) to one of high solute concentration (low water concentration).
Solutes Can Be Transported Through Epithelial Cells or Between Cells. Renal tubular cells, like other epithelial cells, areheld together by tight junctions. Lateral intercellular spaces lie behind the tight junctions and separate the epithelial cells of the tubule. Solutes can be reabsorbed or secreted across the cells by way of the transcellularpathway or between the cells by moving across thetight junctions and intercellular spaces by way of the paracellular pathway. Sodium is a substance that moves through both routes, although most of the sodium is transported through the transcellular pathway. In some nephron segments, especially the proximal tubule, water is also reabsorbed across the paracellular pathway, and substances dissolved in the water, especially potassium, magnesium, and chlo-ride ions, are carried with the reabsorbed fluid between the cells.
Primary Active Transport Through the Tubular Membrane Is Linked to Hydrolysis of ATP.The special importance ofprimary active transport is that it can move solutes against an electrochemical gradient. The energy for thisactive transport comes from the hydrolysis of ATP by way of membrane-bound ATPase; the ATPase is also a component of the carrier mechanism that binds and moves solutes across the cell membranes. The primary active transporters that are known include sodium-potassium ATPase, hydrogen ATPase, hydrogen-potassium ATPase, and calcium ATPase.
A good example of a primary active transport system is the reabsorption of sodium ions across the proximal tubular membrane, as shown in Figure 27–2. On the basolateral sides of the tubular epithelial cell, the cell membrane has an extensive sodium-potassium ATPase system that hydrolyzes ATP and uses the released energy to transport sodium ions out of the cell into the interstitium. At the same time, potassium is transported from the interstitium to the inside of the cell. The operation of this ion pump maintains low intracellular sodium and high intracellular potassium concentrations and creates a net negative charge of about -70 millivolts within the cell. This pumping of sodium out of the cell across the basolateral membrane of the cell favors passive diffusion of sodium across the luminal membrane of the cell, from the tubular lumeninto the cell, for two reasons: (1) There is a concen-tration gradient favoring sodium diffusion into the cell because intracellular sodium concentration is low (12 mEq/L) and tubular fluid sodium concentration is high (140 mEq/L). (2) The negative, -70-millivolt, intracellular potential attracts the positive sodium ions from the tubular lumen into the cell.
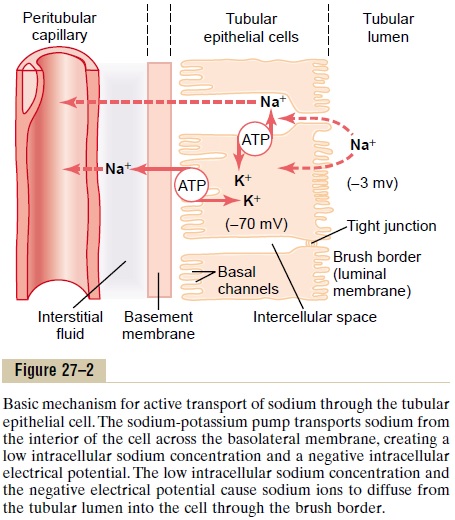
Active reabsorption of sodium by sodium-potassium ATPase occurs in most parts of the tubule. In certain parts of the nephron, there are additional provisions for moving large amounts of sodium into the cell. In the proximal tubule, there is an extensive
There are also sodium carrier proteins that bind sodium ions on the luminal surface of the membrane and release them inside the cell, providing facilitated diffusion of sodium through the membrane into the cell. These sodium carrier pro-teins are also important for secondary active transport of other substances, such as glucose and amino acids, as discussed later.
Thus, the net reabsorption of sodium ions from the tubular lumen back into the blood involves at least three steps:
1. Sodium diffuses across the luminal membrane (also called the apical membrane) into the cell down an electrochemical gradient established by the sodium-potassium ATPase pump on the basolateral side of the membrane.
2. Sodium is transported across the basolateral membrane against an electrochemical gradient by the sodium-potassium ATPase pump.
3. Sodium, water, and other substances are reabsorbed from the interstitial fluid into the peritubular capillaries by ultrafiltration, a passive process driven by the hydrostatic and colloid osmotic pressure gradients.
Secondary Active Reabsorption Through the Tubular Membrane.
In secondary active transport, two or more substances interact with a specific membrane protein (a carrier molecule) and are transported together across the membrane. As one of the substances (for instance, sodium) diffuses down its electrochemical gradient, the energy released is used to drive another substance
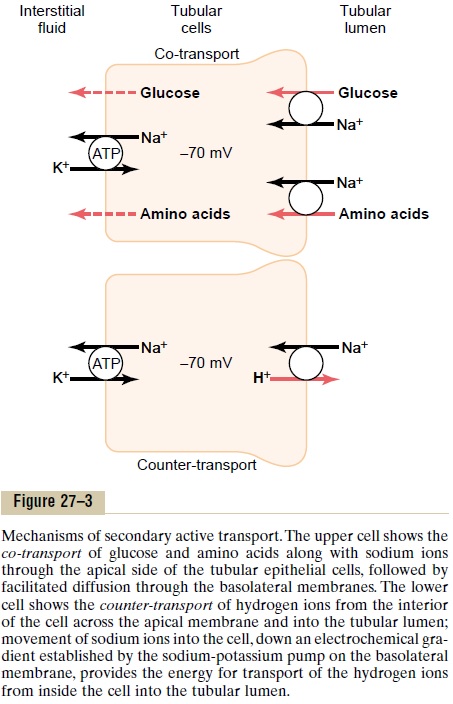
(for instance, glucose) against its electrochemical gradient. Thus, secondary active transport does not require energy directly from ATP or from other high-energy phosphate sources. Rather, the direct source of the energy is that liberated by the simultaneous facil-itated diffusion of another transported substance down its own electrochemical gradient.
Figure 27–3 shows secondary active transport of glucose and amino acids in the proximal tubule. In both instances, a specific carrier protein in the brush border combines with a sodium ion and an amino acid or a glucose molecule at the same time. These trans-port mechanisms are so efficient that they remove vir-tually all the glucose and amino acids from the tubular lumen. After entry into the cell, glucose and amino acids exit across the basolateral membranes by facili-tated diffusion, driven by the high glucose and amino acid concentrations in the cell.
Although transport of glucose against a chemical gradient does not directly use ATP, the reabsorption of glucose depends on energy expended by the primary active sodium-potassium ATPase pump in the basolateral membrane. Because of the activity of this pump, an electrochemical gradient for facilitated diffusion of sodium across the luminal membrane is maintained, and it is this downhill diffusion of sodium to the interior of the cell that provides the energy for the simultaneous uphill transport of glucose across the luminal membrane. Thus, this reabsorption of glucose is referred to as “secondary active transport” because glucose itself is reabsorbed uphill against a chemical gradient, but it is “secondary” to primary active trans-port of sodium.
Another important point is that a substance is said to undergo “active” transport when at least one of the steps in the reabsorption involves primary or second-ary active transport, even though other steps in the reabsorption process may be passive. For glucose reabsorption, secondary active transport occurs at the luminal membrane, but passive facilitated diffusion occurs at the basolateral membrane, and passive uptake by bulk flow occurs at the peritubular capillaries.
Secondary Active Secretion into the Tubules. Some sub-stances are secreted into the tubules by secondary active transport. This often involves counter-transport of the substance with sodium ions. In counter-transport, the energy liberated from the downhill movement of one of the substances (for example, sodium ions) enables uphill movement of a second substance in the opposite direction.
One example of counter-transport, shown in Figure 27–3, is the active secretion of hydrogen ions coupled to sodium reabsorption in the luminal membrane of the proximal tubule. In this case, sodium entry into the cell is coupled with hydrogen extrusion from the cell by sodium-hydrogen counter-transport. This transport is mediated by a specific protein in the brush border of the luminal membrane. As sodium is carried to the interior of the cell, hydrogen ions are forced outward in the opposite direction into the tubular lumen.
Pinocytosis—An Active Transport Mechanism for Reabsorption of Proteins. Some parts of the tubule, especially theproximal tubule, reabsorb large molecules such as proteins by pinocytosis. In this process, the protein attaches to the brush border of the luminal membrane, and this portion of the membrane then invaginates to the interior of the cell until it is completely pinched off and a vesicle is formed containing the protein. Once inside the cell, the protein is digested into its con-stituent amino acids, which are reabsorbed through the basolateral membrane into the interstitial fluid. Because pinocytosis requires energy, it is considered a form of active transport.
Transport Maximum for Substances That Are Actively Reab- sorbed. For most substances that are actively reab-sorbed or secreted, there is a limit to the rate at which the solute can be transported, often referred to as the transport maximum. This limit is due to saturationof the specific transport systems involved when the amount of solute delivered to the tubule (referred to as tubular load) exceeds the capacity of the carrier proteins and specific enzymes involved in the trans-port process.
The glucose transport system in the proximal tubule is a good example. Normally, measurable glucose does not appear in the urine because essentially all the fil-tered glucose is reabsorbed in the proximal tubule. However, when the filtered load exceeds the capabil-ity of the tubules to reabsorb glucose, urinary excre-tion of glucose does occur.
In the adult human, the transport maximum for glucose averages about 375 mg/min, whereas the fil-tered load of glucose is only about 125 mg/min (GFR x plasma glucose = 125 ml/min x 1 mg/ml). With large increases in GFR and/or plasma glucose concentration that increase the filtered load of glucose above 375 mg/ min, the excess glucose filtered is not reabsorbed and passes into the urine.
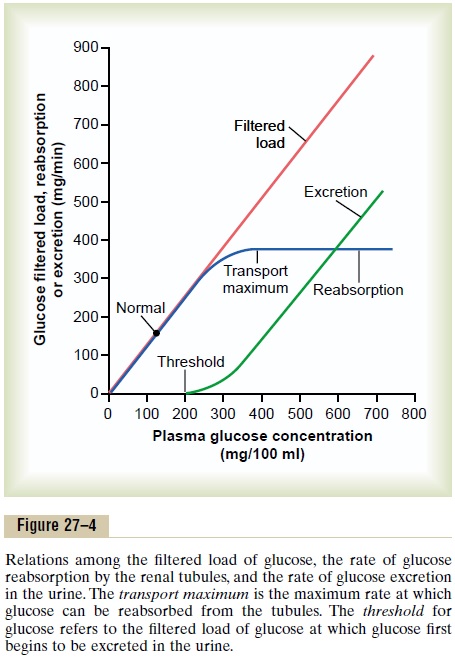
Figure 27–4 shows the relation between plasma con-centration of glucose, filtered load of glucose, tubular transport maximum for glucose, and rate of glucose loss in the urine. Note that when the plasma glucose concentration is 100 mg/100 mL and the filtered load is at its normal level, 125 mg/min, there is no loss of glucose in the urine. However, when the plasma con-centration of glucose rises above about 200 mg/100 ml, increasing the filtered load to about 250 mg/min, a small amount of glucose begins to appear in the urine. This point is termed the threshold for glucose. Notethat this appearance of glucose in the urine (at the threshold) occurs before the transport maximum is reached. One reason for the difference betweenthreshold and transport maximum is that not all nephrons have the same transport maximum for glucose, and some of the nephrons excrete glucose before others have reached their transport maximum.
The overall transport maximum for the kidneys, which is normally about 375 mg/min, is reached when all nephrons have reached their maximal capacity to reab-sorb glucose.
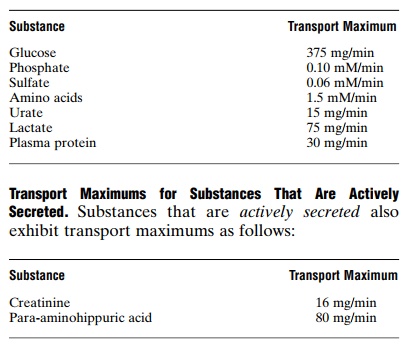
The plasma glucose of a healthy person almost never becomes high enough to cause excretion of glucose in the urine, even after eating a meal. However, in uncontrolled diabetes mellitus, plasma glucose may rise to high levels, causing the filtered load of glucose to exceed the transport maximum and resulting in urinary glucose excretion. Some of the important transport maximums for substances activelyreabsorbed by the tubules are as follows:
Substances That Are Actively Transported but Do Not Exhibit a Transport Maximum. The reason that actively trans-ported solutes often exhibit a transport maximum is that the transport carrier system becomes saturated as the tubular load increases. Substances that are passivelyreabsorbed do not demonstrate a transport maximum because their rate of transport is determined by other factors, such as (1) the electrochemical gradient for diffusion of the substance across the membrane, (2) the permeability of the membrane for the substance, and (3) the time that the fluid containing the substance remains within the tubule. Transport of this type is referred to asgradient-time transport because the rate of transport depends on the electrochemical gradient and the time that the substance is in the tubule, which in turn depends on the tubular flow rate.
Some actively transported substances also have char-acteristics of gradient-time transport. An example issodium reabsorption in the proximal tubule. The main reason that sodium transport in the proximal tubule does not exhibit a transport maximum is that other factors limit the reabsorption rate besides the maximum rate of active transport. For example, in the proximal tubules, the maximum transport capacity of the basolateral sodium-potassium ATPase pump is usually far greater than the actual rate of net sodium reabsorption. One of the reasons for this is that a sig-nificant amount of sodium transported out of the cell leaks back into the tubular lumen through the epithelial tight junctions. The rate at which this back-leak occurs depends on several factors, including (1) the permeability of the tight junctions and (2) the interstitial physical forces, which determine the rate of bulk flow reabsorption from the interstitial fluid into the peritubular capillaries. Therefore, sodium trans-port in the proximal tubules obeys mainly gradient-time transport principles rather than tubular maximum transport characteristics. This means that the greater the concentration of sodium in the proxi-mal tubules, the greater its reabsorption rate. Also, the slower the flow rate of tubular fluid, the greater the percentage of sodium that can be reabsorbed from the proximal tubules.
In the more distal parts of the nephron, the epithelial cells have much tighter junctions and transport much smaller amounts of sodium. In these segments, sodium reabsorption exhibits a transport maximum similar to that for other actively transported substances. Furthermore, this transport maximum can be increased in response to certain hormones, such as aldosterone.
Passive Water Reabsorption by Osmosis Is Coupled Mainly to Sodium Reabsorption
When solutes are transported out of the tubule by either primary or secondary active transport, their concentrations tend to decrease inside the tubule while increasing in the renal interstitium. This creates a concentration difference that causes osmosis of water in the same direction that the solutes are trans-ported, from the tubular lumen to the renal intersti-tium. Some parts of the renal tubule, especially the proximal tubule, are highly permeable to water, and water reabsorption occurs so rapidly that there is only a small concentration gradient for solutes across the tubular membrane.
A large part of the osmotic flow of water occurs through the so-called tight junctions between the epithelial cells as well as through the cells themselves. The reason for this, as already discussed, is that the junctions between the cells are not as tight as their name would imply, and they allow significant diffusion of water and small ions. This is especially true in the proximal tubules, which have a high permeability for water and a smaller but significant permeability to most ions, such as sodium, chloride, potassium, calcium, and magnesium.
As water moves across the tight junctions by osmosis, it can also carry with it some of the solutes, a process referred to as solvent drag. And because the reabsorption of water, organic solutes, and ions is coupled to sodium reabsorption, changes in sodium reabsorption significantly influence the reabsorption of water and many other solutes.
In the more distal parts of the nephron, beginning in the loop of Henle and extending through the col-lecting tubule, the tight junctions become far less per-meable to water and solutes, and the epithelial cells also have a greatly decreased membrane surface area. Therefore, water cannot move easily across the tubular membrane by osmosis. However, antidiuretic hormone (ADH) greatly increases the water permeability in the distal and collecting tubules, as discussed later.
Thus, water movement across the tubular epithe-lium can occur only if the membrane is permeable to water, no matter how large the osmotic gradient. In the proximal tubule, the water permeability is always high, and water is reabsorbed as rapidly as the solutes. In the ascending loop of Henle, water permeability is always low, so that almost no water is reabsorbed, despite a large osmotic gradient. Water permeability in the last parts of the tubules—the distal tubules, col-lecting tubules, and collecting ducts—can be high or low, depending on the presence or absence of ADH.
Reabsorption of Chloride, Urea, and Other Solutes by Passive Diffusion
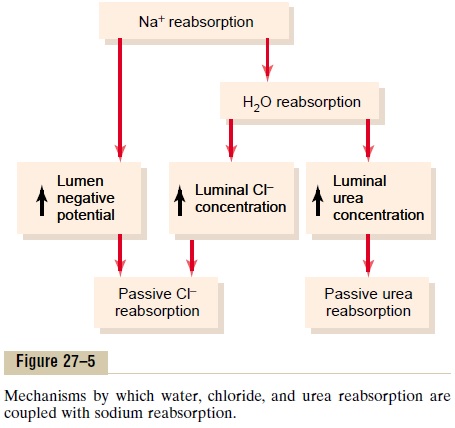
When sodium is reabsorbed through the tubular epithelial cell, negative ions such as chloride are trans-ported along with sodium because of electrical poten-tials. That is, transport of positively charged sodium ions out of the lumen leaves the inside of the lumen negatively charged, compared with the interstitial fluid. This causes chloride ions to diffuse passively through the paracellular pathway. Additional reab-sorption of chloride ions occurs because of a chloride concentration gradient that develops when water is reabsorbed from the tubule by osmosis, thereby con-centrating the chloride ions in the tubular lumen (Figure 27–5). Thus, the active reabsorption of sodium is closely coupled to the passive reabsorption of chlo-ride by way of an electrical potential and a chloride concentration gradient.
Chloride ions can also be reabsorbed by secondary active transport. The most important of the secondary active transport processes for chloride reabsorption involves co-transport of chloride with sodium across the luminal membrane.
Urea is also passively reabsorbed from the tubule, but to a much lesser extent than chloride ions. As water is reabsorbed from the tubules (by osmosis coupled to sodium reabsorption), urea concentration in the tubular lumen increases (see Figure 27–5). This creates a concentration gradient favoring the reab-sorption of urea. However, urea does not permeate the tubule as readily as water. In some parts of the nephron, especially the inner medullary collecting duct, passive urea reabsorption is facilitated by specific urea transporters. Yet only about one half of the ureathat is filtered by the glomerular capillaries is reab-sorbed from the tubules. The remainder of the urea passes into the urine, allowing the kidneys to excrete large amounts of this waste product of metabolism.
Another waste product of metabolism, creatinine, is an even larger molecule than urea and is essentially impermeant to the tubular membrane. Therefore, almost none of the creatinine that is filtered is reab-sorbed, so that virtually all the creatinine filtered by the glomerulus is excreted in the urine.
Related Topics