Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Mycobacteria
Tuberculosis : Clinical Aspects
TUBERCULOSIS : CLINICAL ASPECTS
MANIFESTATIONS
Primary Tuberculosis
Primary tuberculosis is either asymptomatic or manifest only by fever and malaise. Radi-ographs may show infiltrates in the mid-zones of the lung and enlarged draining lymph nodes in the area around the hilum. When these lymph nodes fibrose and sometimes calcify, they produce a characteristic picture (Ghon complex) on radiograph. In approximately 5% of patients, the primary disease is not controlled and merges into the reactivation type of tuberculosis, or it disseminates to many organs to produce active miliary tuberculosis. The latter may result from a necrotic tubercle eroding into a small blood vessel.
Reactivation Tuberculosis
Approximately 10% of those recovering from a primary infection develop clinical disease sometime during their lifetime. In Western countries, reactivation of previous quiescent lesions occurs most often after the age of 50 and is more common in men. Reactivation is associated with a period of immunosuppression precipitated by malnutrition, alcoholism, diabetes, old age, and a dramatic change in the individual’s life, such as loss of a spouse. In areas in which the disease is more common, reactivation tuberculosis is more fre-quently seen in young adults experiencing the immunosuppression that accompanies pu-berty and pregnancy. Recently, reactivation and progressive primary tuberculosis among younger adults have increased as a complication of AIDS.
Cough is the universal symptom. It is initially dry, but as the disease progresses sputum is produced, which even later is mixed with blood (hemoptysis). Fever, malaise, fatigue, sweating, and weight loss all progress with continuing disease. Radiographically, infiltrates appearing in the apices of the lung coalesce to form cavities with progressive destruction of lung tissue. Less commonly, reactivation tuberculosis can also occur in other organs, such as the kidneys, bones, lymph nodes, brain, meninges, bone marrow, and bowel. Dis-ease at these sites ranges from a localized tumor-like granuloma (tuberculoma) to a fatal chronic meningitis. Untreated, the progressive cough, fever, and weight loss of pulmonary tuberculosis creates an internally consuming fire that usually takes 2 to 5 years to cause death. The course in AIDS and other CMI-compromised patients is more rapid.
DIAGNOSIS
Tuberculin Test
The tuberculin skin test measures DTH to tuberculoprotein. PPD is standardized biologically against an international reference preparation, and its activity expressed in tuberculin units (TU). Most initial skin tests employ 5 TU (intermediate strength). When an unusually high degree of hypersensitivity or eye or skin tuberculosis is suspected, then 1 TU (first strength) or less is used initially to avoid the risk of an excessive reaction locally or at the site of a mycobacterial lesion.
The test most commonly performed involves intradermal injection that is read 48 to 72 hours later. An area of measured induration of 10 mm or more accompanied by ery-thema constitutes a positive reaction, although smaller areas of induration and erythema indicate a lesser degree of sensitization to mycobacterial proteins. No induration indicates a negative reaction. A positive PPD test indicates that the individual has been infected at some time with M. tuberculosis or with a strongly cross-reacting mycobacterium of an-other species. It carries no implication about the activity of the infection, which may have been simply a primary complex contracted 20 years previously.
A negative PPD test in a healthy individual indicates that he or she has not been in-fected with M. tuberculosis, is in the prehypersensitive stage of a primary infection, or has finally lost tuberculin sensitivity along with disappearance of antigen from an old primary complex. Patients with severe disseminated disease, those on steroid or immuno-suppressive drugs, or those with certain other diseases such as AIDS and measles, may also become anergic. They lose their tuberculin hypersensitivity and become more sus-ceptible to the disease. Induration below the 10-mm diameter criterion for positivity indi-cates low-level sensitization, which may be attributable to M. tuberculosis infection or to a cross-reacting mycobacterial infection.
The clinical value of the PPD test depends on the occurrence of primary infection in different age groups. Now, primary infection is sufficiently uncommon in much of the Western world that a negative test is frequently important in excluding tuberculosis. A positive test in infancy or childhood has significance in diagnosis and can often be used to trace a household or school source of infection. Epidemiologic surveys of tuberculin reac-tivity indicate trends in the incidence of infection and constitute the simplest way of mon-itoring the effectiveness of control measures.
Laboratory Diagnosis
If present in sufficient numbers, acid-fast bacilli can be detected microscopically in direct smears of clinical specimens or in smears of material concentrated for culture (see be-low). Smears are stained by the Ziehl – Neelsen procedure or one of its modifications, in-cluding the fluorescence staining method. About 65% of culture-positive sputum samples yield positive smears from concentrated specimens. These procedures are not specific for M. tuberculosis because other mycobacteria may have a similar morphology and may beetiologic agents of disease, members of the normal flora, or external contaminants. Their significance depends on the specimen. Acid-fast bacilli in sputum are highly significant for mycobacterial infection. A clean-voided male urine specimen, on the other hand, is often contaminated with Mycobacterium smegmatisfrom the prepuce, and the finding of acid-fast bacilli does not per se indicate infection. Bronchoscopy equipment and nasotra-cheal tubes or their lubricants are prone to contamination with free-living mycobacteria, and false conclusions have been drawn from smears of such preparations. The polymerase chain reaction has been reported to be useful in the direct diagnosis of tuberculosis by a number of investigators. To date, none of these techniques are practical for routine use in the clinical laboratory.
Cultural confirmation of a tentative diagnosis of tuberculosis is thus essential, and the organism must be isolated for identification and susceptibility testing. Specimens from protected sites, such as cerebrospinal fluid, bone marrow, pleural fluid, and ureteric urine, can be seeded directly to culture media used for M. tuberculosis isolation. Those samples inevitably contaminated with normal flora, such as sputum, gastric aspirations (cultured when sputum is not available, for example, in young children), or voided urine, are treated with alkali, acid, or a detergent germicide under conditions that kill the normal flora but allow many mycobacteria to survive because of their resistance to these agents. The most commonly used treatment now employs N-acetylcysteine to dissolve mucus, combined with the antibacterial effect of a weak sodium hydroxide solution. The material is concentrated by centrifugation or filtration, neutralized or washed, and inoculated onto culture media.
Cultures on solid media usually take 3 weeks or longer to show visible colonies. Growth may be detected radiometrically in about half the time by using liquid oleic acid – albumin broth containing 14C-labeled palmitic acid, which is metabolized by mycobacteria to liberate 14CO2. The labeled CO2 is detected in the space above the medium using an automated sampling procedure. Incorporation of a specific inhibitor of M. tuberculosis in a parallel vial increases the specificity of the test.
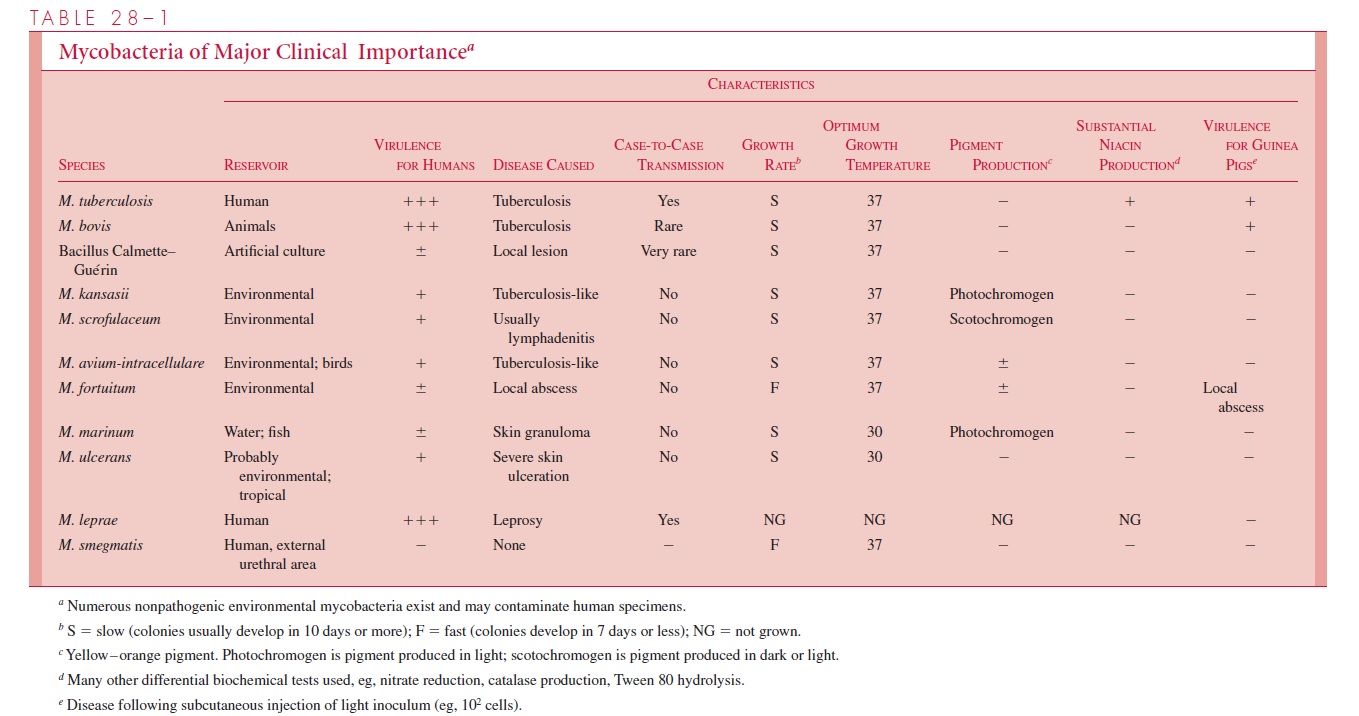
Whichever procedure is used, specific identification of an isolated mycobacterium is essential. It may be achieved with a number of cultural and biochemical tests, including those shown in Table 28 – 1, but the process usually takes several weeks. More rapid re-sults can be obtained by high-resolution gas chromatographic analysis of fatty acids in mycobacterial colonies or by testing for homology between genetic probes of labeled my-cobacterial DNA and ribosomal RNA extracted from the strain under test. Specific probes are now available commercially for detecting M. tuberculosis and the Mycobacteriumavium – intracellulare complex.
Susceptibility testing is important with newly diagnosed cases. When sufficient num-bers of acid-fast bacilli are seen on direct smears, the treated clinical specimen can be seeded directly onto antimicrobic-containing media for susceptibility tests, thereby saving several weeks. If numbers are scanty, the initiation of tests must await primary isolation. More rapid test results can be obtained by incorporating antimicrobics into the medium used for radiometric detection of mycobacterial growth. These results show good concor-dance with conventional tests and are available 1 to 2 weeks earlier.
TREATMENT
M. tuberculosis is susceptible to several effective antimicrobics (Table 28 – 2). Isoniazid,ethambutol, rifampin, pyrazinamide, streptomycin, and combinations of these agents constitute the primary drugs of choice for treatment of tuberculosis. All of these, except ethambutol, are bactericidal. Isoniazid and rifampin are active against both intra- and ex-tracellular organisms, and pyrazinamide, a nicotinamide analog, acts at the acidic pH found within cells. Streptomycin does not penetrate into cells and is thus active only against extracellular organisms. M. tuberculosis is also susceptible to other drugs that may be used to replace those of the primary group if they are inappropriate because of resistance or drug toxicity. The fluoroquinolones, such as ciprofloxacin and ofloxacin, are active against M. tuberculosis and penetrate well into infected cells. Their role in the treatment of tuberculosis is under evaluation. Isoniazid and ethambutol act on the my-colic acid (isoniazid) and LAM (ethambutol) elements of mycobacterial cell wall syn-thesis. The molecular targets of the other agents have yet to be defined except for the general antibacterial agents (rifampin, streptomycin, fluoroquinolones).

Mutational resistance to antituberculous drugs occurs at frequencies of 10 7 to 10 10. For example, mutation in a gene coding for a catalase-peroxidase enzyme causes failure of the conversion of isoniazid to its biologically active form. Such mutants often come to predominate and produce clinical relapse particularly when a single drug is used. Adequate, continuous treatment with two or three antituberculous drugs with different modes of action greatly reduces the probability a mutant will be expressed, because the chance of a doubly resistant mutant in a lesion’s organism population is very low. The proportion of infections with strains resistant to first-line drugs varies between 5 and 15% but appears to be increasing in many locales, particularly among individuals who have been treated previously. Of particular concern is the establishment in the last decade of strains resistant to both isoniazid and rifampin, the mainstays of primary treat-ment. Susceptibility tests are required to guide drug selection.
Treatment with multiple antimicrobics to which the organism is susceptible usually renders the patient noninfectious within 1 or 2 weeks, which has shifted the care of tuber-culous patients from isolation hospitals and sanatoriums to the home or the general hospi-tal. After an initial intense phase of systemic chemotherapy, treatment is usually continued with oral antimicrobics for several months. Until recently, therapy with two oral agents, isoniazid and ethambutol, was continued for a total of 18 to 24 months. Studies have now demonstrated that therapy can be shortened to 9 months when isoniazid and rifampin are used concomitantly and to 6 months when pyrazinamide is added as a third agent. In patients whose organisms display resistance to one or more of these drugs, and in those with HIV infection, a more prolonged treatment course is used. The effectiveness of chemotherapy on most forms of tuberculosis has been dramatic and has greatly reduced the need for surgical procedures such as pulmonary lobectomy. Failure of chemotherapy is often associated with lack of adherence to the regimen by the patient, the presence of resistant organisms, or both.
PREVENTION
Prophylactic chemotherapy, usually with isoniazid alone, is now used in situations in which known or suspected primary tuberculous infection poses the risk of clinical disease. Isoniazid can be used alone in prophylaxis because the load of tubercle bacilli in a subclinical primary lesion is small in relation to that in reactivation tuberculosis, and experience has shown that the development of subsequent clinical disease from isoniazid-resistant strains se-lected by prophylaxis can be discounted. Unfortunately, isoniazid may cause a form of hepatitis, and the risk increases progressively after 20 years of age. Its use in older sub-jects involves balancing risk against potential benefit and requires monitoring with liver function tests.
At present, the bacillus Calmette-Guérin (BCG) vaccine (named for its originators, Calmette and Guérin) is the only available vaccine. It has been used for prophylaxis of tu-berculosis in various countries since 1923; administration is usually intradermal. It is a live vaccine derived originally from a strain of M. bovis that was attenuated by repeated subculture. Since then, it has had a checkered history, with results in different controlled trials ranging from ineffectiveness to 80% protection. In most studies, however, it has substantially decreased the highly lethal miliary and meningeal forms of tuberculosis among young children. On the basis of these results, massive immunization campaigns sponsored by the World Health Organization have been organized in underdeveloped countries.
BCG is used only in tuberculin-negative subjects. Successful vaccination leads to a mi-nor local lesion, self-limiting multiplication of the organism locally and in draining lym-phatic vessels, and development of tuberculin hypersensitivity. The latter results in loss of the PPD test as a diagnostic and epidemiologic tool, and when infection rates are low, as they are now in most Western countries, this loss may offset the possible immunity pro-duced. In general, tuberculosis rates in the West have declined as rapidly in countries that have not used the BCG vaccine as in those that have adopted mass vaccination with its occasional complications. Its potential value in these countries is restricted to population groups at particular risk. Its role in developing countries remains a matter of some contention. The BCG vaccination is contraindicated for individuals in whom cell-mediated immune mechanisms are compromised, such as those infected with the HIV.
Related Topics