Chapter: 11th Zoology : Chapter 6 : Respiration
Transport of gases(oxygen and Carbon-dioxide) in Respiration
Transport of
gases
1. Transport of oxygen
Molecular
oxygen is carried in blood in two ways: bound to haemoglobin within the red
blood cells and dissolved in plasma. Oxygen is poorly soluble in water, so only
3% of the oxygen is transported in the dissolved form. 97% of oxygen binds with
haemoglobin in a reversible manner to form oxyhaemoglobin (HbO2).
The rate at which haemoglobin binds with O2 is regulated by the
partial pressure of O2. Each haemoglobin carries maximum of four
molecules of oxygen. In the alveoli high pO2, low pCO2,
low temperature and less H+ concentration, favours the formation of
oxyhaemoglobin, whereas in the tissues low pO2, high pCO2,
high H+ and high temperature favours the dissociation of oxygen from
oxyhaemoglobin.
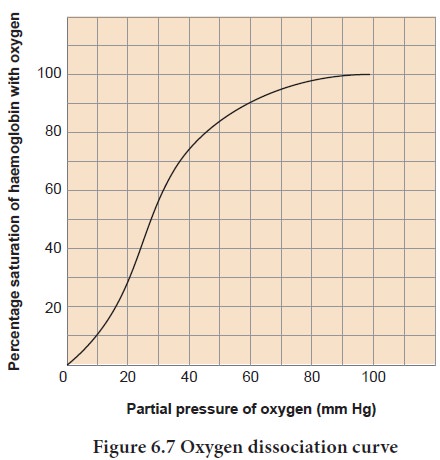
A sigmoid
curve (S–shaped) is obtained whenpercentagesaturationofhaemoglobin with oxygen
is plotted against pO2. This curve is called oxygenhaemoglobin
dissociation curve (Figure 6.7). This S–shaped curve has a steep slope for pO2
values between 10 and 50mmHg and then flattens between 70 and 100 mm Hg.
Under
normal physiological conditions, every 100mL of oxygenated blood can deliver
about 5mL of O2 to the tissues.
2. Transport of
Carbon–dioxide
Blood
transports CO2 from the tissue cells to the lungs in three ways
i. Dissolved in plasma About 7 – 10% of CO2 is transported in a dissolved form in the plasma.
ii.Bound to haemoglobin About 20 –
25% of dissolved CO2 is
bound and carried in the RBCs as
carbaminohaemoglobin (Hb CO2)

iii. As bicarbonate ions in plasma about 70%
of CO2 is transported as
bicarbonate ions This is influenced
by pCO2 and the degree of haemoglobin oxygenation. RBCs contain a
high concentration of the enzyme, carbonic anhydras, Whereas small amounts of
carbonic anhydrase is present in the plasma.
At the
tissues the pCO2 is high due to catabolism and diffuses into the
blood to form HCC3– and H+ ions. When CO2 diffuses into
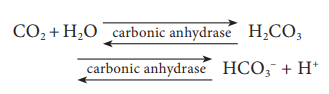
the RBCs, it combines with water forming carbonic acid (H2CC3)
catalyzed by carbonic anhydrase. Carbonic acid is unstable and dissociates into
hydrogen and bicarbonate ions.
Carbonic
anhydrase facilitates the reaction in both directions.
The HCC3–
moves quickly from the RBCs into the plasma, where it is carried to the lungs.
At the alveolar site where pCO2 is low, the reaction is reversed
leading to the formation of CO2 and water. Thus CO2
trapped as HCC3– at the tissue level it is transported to
the alveoli and released out as CO2. Every 100mL of deoxygenated
blood delivers 4mL of CO2 to the alveoli for elimination.
Bohr effect and Haldane effect
Increase
in PCO2 and decrease in pH decrease the affinity of haemoglobin for
oxygen and shifts the oxyhaemoglobin dissociation curve to the right and
facilitates unloading of oxygen from hemoglobin in the tissue. This effect of pCO2
and pH on the oxyhaemoglobin dissociation curve is called the Bohr small
effect.
The
Haldane effect, on the other hand describes how oxygen concentrations
determines hemoglobin’s affinity for carbon dioxide. The amount of carbon
dioxide transported in blood is remarkably affected by the degree oxygenation
of the blood. The lower the partial pressure of. O2 lower is the
affinity of haemoglobin saturation with oxygen hence more CO2 is
carried in the blood. This phenomenon is called Haldane effect. This effects CO2
exchanges in both the tissues and lungs.
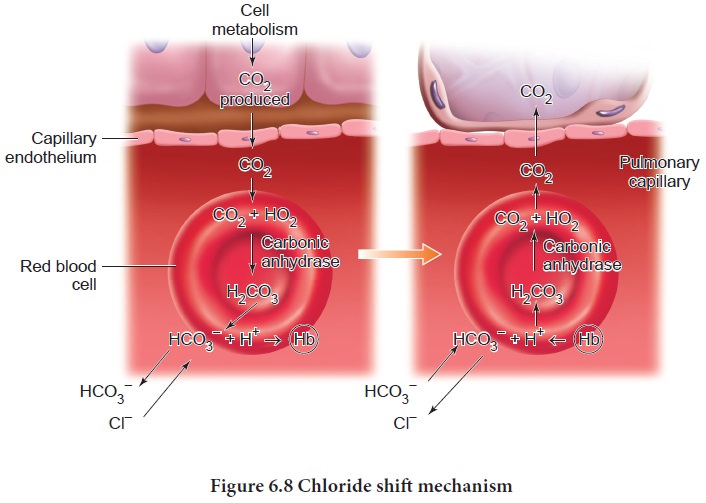
In the
lungs the process is reversed as the blood moves through the pulmonary
capillaries, its PCO2 declines from 45mm Hg to 40mm Hg. For this to
occur carbondioxide is freed from HCC3- ions and
Cl- ions moves in to the plasma and reenters the RBC and binds with H+ to form
carbonic acid which dissociates in to CO2 and water. This CO2
diffuses along its partial gradient from the blood to the alveoli (Figure 6.8).
Related Topics