Chapter: Basic & Clinical Pharmacology : Thyroid &Antithyroid Drugs
Thyroid Hormones
BASIC PHARMACOLOGY OF THYROID & ANTITHYROID DRUGS
THYROID HORMONES
Chemistry
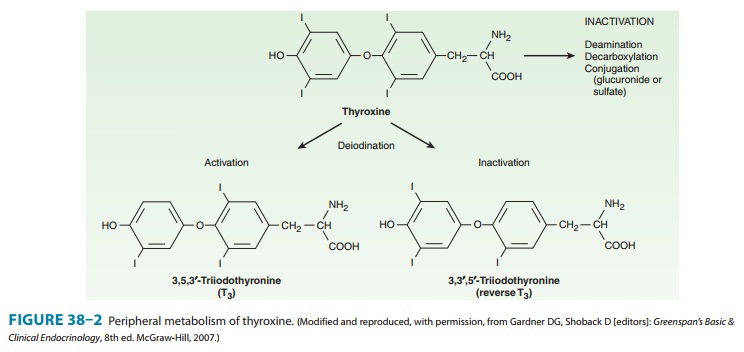
The structural
formulas of thyroxine and triiodothyronine as well as reverse triiodothyronine
(rT3) are shown in Figure
38–2. All of these naturally occurring molecules are levo (L) isomers. The synthetic dextro (D)
isomer of thyroxine, dextrothyroxine, has approximately 4% of the biologic
activity of the L-isomer as evidenced
by its lesser ability to suppress TSH secretion and correct hypothyroidism.
Pharmacokinetics
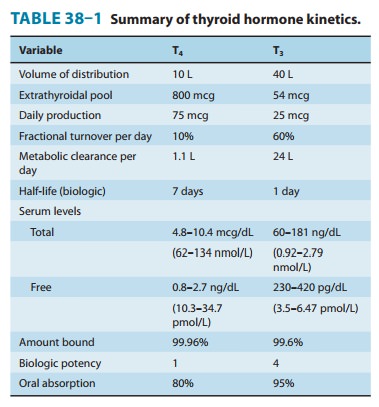
Thyroxine is absorbed
best in the duodenum and ileum; absorp-tion is modified by intraluminal factors
such as food, drugs, gas-tric acidity, and intestinal flora. Oral
bioavailability of current preparations of L-thyroxine averages 80% (Table 38–1). In con-trast, T3 is almost completely
absorbed (95%). T4 and T3 absorp-tion appears not to be affected by mild hypothyroidism
but may be impaired in severe myxedema with ileus. These factors are important
in switching from oral to parenteral therapy. For paren-teral use, the
intravenous route is preferred for both hormones.
In patients with
hyperthyroidism, the metabolic clearances of T4 and T3 are increased and the half-lives decreased;
the opposite is true in patients with hypothyroidism. Drugs that induce hepatic
microsomal enzymes (eg, rifampin, phenobarbital, carbamazepine, phenytoin,
tyrosine kinase inhibitors, HIV protease inhibitors) increase the metabolism of
both T4 and T3 (Table 38–3). Despite
this change in clearance, the normal hormone concentration is maintained in the
majority of euthyroid patients as a result of compensatory hyperfunction of the
thyroid. However, patients dependent on T4 replacement medication may require increased
dosages to maintain clinical effectiveness. A similar compensation occurs if
binding sites are altered. If TBG sites are increased by pregnancy, estrogens,
or oral contraceptives, there is an initial shift of hormone from the free to
the bound state and a decrease in its rate of elimination until the normal free
hormone concentration is restored. Thus, the concentration of total and bound
hormone will increase, but the concentration of free hormone and the
steady-state elimination will remain normal. The reverse occurs when thyroid
binding sites are decreased.
Mechanism of Action
A model of thyroid hormone
action is depicted in Figure 38–4, which shows the free forms of thyroid
hormones, T4 and T3,
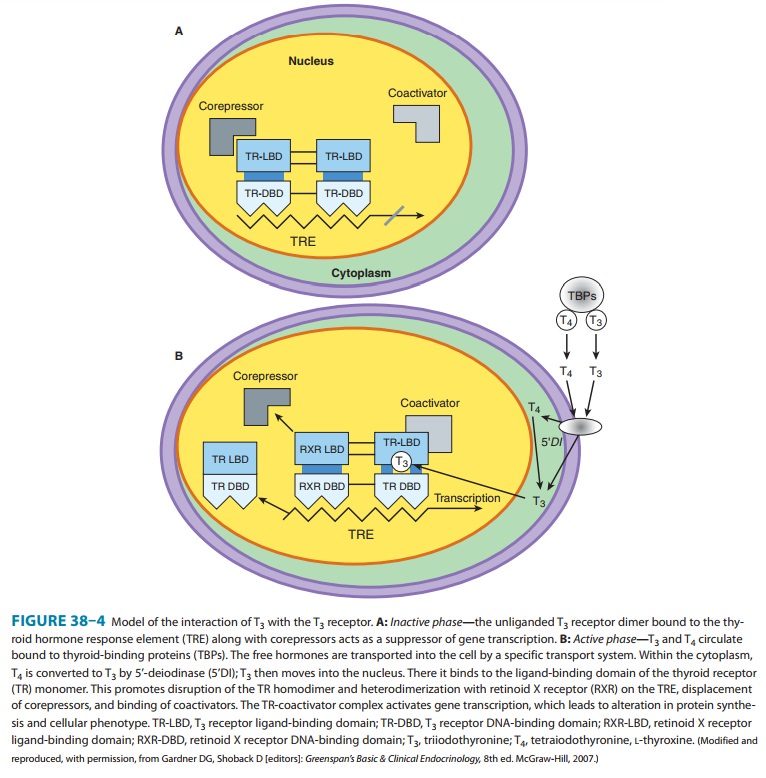
Within the
cell, T4 is converted to T3 by 5’-deiodinase, and the T3
enters the nucleus, where T3 binds to a specific T3
receptor protein, a member of the c-erb
oncogene family. (This family also includes the steroid hormone receptors and
receptors for vitamins A and D.) The T3 receptor exists in two
forms, α and β. Differing concentrations of receptor forms in different
tissues may account for variations in T3 effect on different
tissues.
Most
of the effects of thyroid on metabolic processes appear to be mediated by
activation of nuclear receptors that lead to increased formation of RNA and
subsequent protein synthesis, eg, increased formation of Na+/K+-ATPase.
This is consistent with the observation that the action of thyroid is
manifested in vivo with a time lag of hours or days after its administration.
Large
numbers of thyroid hormone receptors are found in the most hormone-responsive
tissues (pituitary, liver, kidney, heart, skeletal muscle, lung, and
intestine), while few receptor sites occur in hormone-unresponsive tissues
(spleen, testes). The brain, which lacks an anabolic response to T3,
contains an intermediate number of receptors. In congruence with their biologic
potencies, the affinity of the receptor site for T4
is about ten times lower than that for T3.
Under some conditions, the number of nuclear recep-tors may be altered to
preserve body homeostasis. For example, starvation lowers both circulating T3
hormone and cellular T3 receptors.
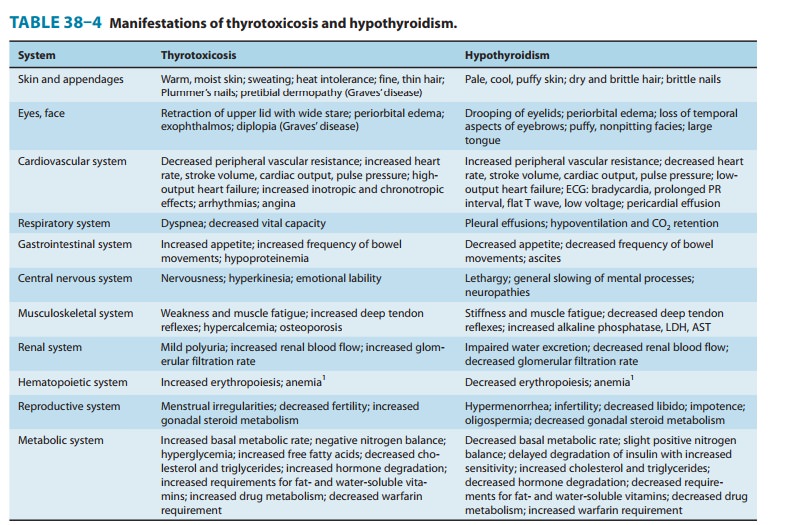
Effects of Thyroid Hormones
The
thyroid hormones are responsible for optimal growth, devel-opment, function, and
maintenance of all body tissues. Excess or inadequate amounts result in the
signs and symptoms of hyper-thyroidism or hypothyroidism, respectively (Table
38–4). Since T3 and T4
are qualitatively similar, they may be considered as one hormone in the discussion
that follows.
Thyroid
hormone is critical for the development and function-ing of nervous, skeletal,
and reproductive tissues.Its effects depend on protein synthesis as well as
potentiation of the secretion and action of growth hormone. Thyroid deprivation
in early life results in irreversible mental retardation and dwarfism—typical
of congenital cretinism.
Effects
on growth and calorigenesis are accompanied by a per-vasive influence on metabolism
of drugs as well as carbohydrates, fats, proteins, and vitamins. Many of these
changes are dependent upon or modified by activity of other hormones.
Conversely, the secretion and degradation rates of virtually all other
hormones, including catecholamines, cortisol, estrogens, testosterone, and
insulin, are affected by thyroid status.
Many of the manifestations of thyroid hyperactivity resemble sympathetic nervous system overactivity (especially in the cardio-vascular system), although catecholamine levels are not increased. Changes in catecholamine-stimulated adenylyl cyclase activity as measured by cAMP are found with changes in thyroid activity.
Possible
explanations include increased numbers of β receptors or enhanced
amplification of the β-receptor signal. Other clinical
symptoms reminiscent of excessive epinephrine activity (and par-tially
alleviated by adrenoceptor antagonists) include lid lag and retraction, tremor,
excessive sweating, anxiety, and nervousness. The opposite constellation of
effects is seen in hypothyroidism (Table 38–4).
Thyroid Preparations
See the Preparations
Available section at the end for a list of available preparations. These
preparations may be synthetic (levothyroxine, liothyronine, liotrix) or of
animal origin (desiccated thyroid).
Thyroid
hormones are not effective and can be detrimental in the management of obesity,
abnormal vaginal bleeding, or depres-sion if thyroid hormone levels are normal.
Anecdotal reports of a beneficial effect of T3
administered with antidepressants were not confirmed in a controlled study.
Synthetic
levothyroxine is the preparation of choice for thyroid replacement and
suppression therapy because of its stability, content uniformity, low cost,
lack of allergenic foreign protein, easy laboratory measurement of serum
levels, and long half-life (7 days), which permits once-daily administration.
In addition, T4 is converted to T3
intracellularly; thus, administration of T4
pro-duces both hormones. Generic levothyroxine preparations provide comparable
efficacy and are more cost-effective than branded preparations.
Although liothyronine
(T3) is three to four
times more potent than levothyroxine, it is not recommended for routine
replace-ment therapy because of its shorter half-life (24 hours), which
requires multiple daily doses; its higher cost; and the greater dif-ficulty of
monitoring its adequacy of replacement by conventional laboratory tests.
Furthermore, because of its greater hormone activity and consequent greater
risk of cardiotoxicity, T3 should be avoided in patients with cardiac disease. It is best
used for short-term suppression of TSH. Because oral administration of T3 is unnecessary, use
of the more expensive mixture of thyroxine and liothyronine (liotrix) instead
of levothyroxine is never required.
The use of desiccated
thyroid rather than synthetic prepara-tions is never justified, since the
disadvantages of protein antige-nicity, product instability, variable hormone
concentrations, and difficulty in laboratory monitoring far outweigh the
advantage of lower cost. Significant amounts of T3 found in some thyroid extracts and liotrix
may produce significant elevations in T3 levels and toxicity. Equi-effective doses are
100 mg of desiccated thyroid, 100 mcg of levothyroxine, and 37.5 mcg of
liothyronine.
The
shelf life of synthetic hormone preparations is about 2 years, particularly if
they are stored in dark bottles to minimize spontane-ous deiodination. The
shelf life of desiccated thyroid is not known with certainty, but its potency
is better preserved if it is kept dry.
Related Topics