Atomic Structure | Term 1 Unit 4 | 7th Science - The subatomic particles | 7th Science : Term 1 Unit 4 : Atomic Structure
Chapter: 7th Science : Term 1 Unit 4 : Atomic Structure
The subatomic particles
The subatomic particles
The discoveries made during the
twentieth century proved that atoms of all elements are made up of smaller
components -electron, proton and neutron. An electron from hydrogen atom is no
different from electron of a carbon atom. In the same manner, protons and
neutrons of all elements also have same characteristics. These particles that
make up the atom are called Subatomic Particles.
Proton (p)
The proton is the positively charged
particle and its located in the nucleus. Its positive charge is of the same
magnitude as that of the electron’s negative charge.
Neutron (n)
Neutron is inside the nucleus. The
neutron does not have any charge. Excepting hydrogen (protium), the nuclei of
all atoms contain neutrons.
Electron (e)
This is a negatively charged
particle. Electrons revolve around the nucleus of the atom in specific orbits.
The mass of an electron is negligible as compared to that of a proton or
neutron. Hence, the mass of an atom depends on the number of protons and
neutrons in the nucleus.
Protons and Neutrons are the two
types of particles in the nucleus of an atom. They are called nucleons.The
total negative charge of all an electrons outside the nucleus is equal to the
total positive charge in the nucleus. That makes the atom electrically neutral.
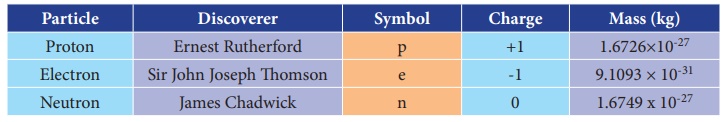
Charge and mass of the sub atomic particles:
ACTIVITY 2
Let us learn the
characteristics of the subatomic particles through the following activity.
Label the parts in the given diagram and answer the following.
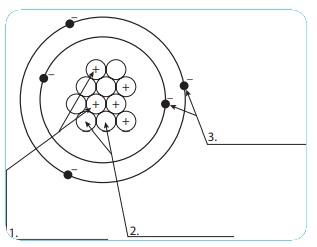
1. The positively charged
particleis proton.
2. The negatively charged
particleis electron.
3. Electron is
neutral.
Related Topics