Chapter: 11th Zoology : Chapter 9 : Locomotion and Movement
Structure of contractile proteins
Structure of contractile proteins
Contraction of the muscle depends on the presence of contractile proteins (Figure 9.3) such as actin and myosin in the myofilaments. The thick filaments are composed of the protein myosin which are bundled together whose heads produce at opposite ends of the filament. Each myosin molecule is made up of a monomer called meromyosin.
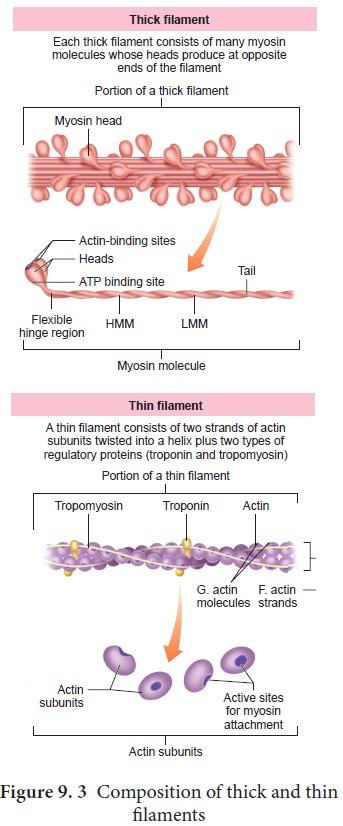
The meromyosin has two regions, a globular head with a short arm
and a tail. The short arm constitutes the heavy meromyosin (HMM). The tail
portion forms the light meromyosin (LMM). The head bears an actin- binding site
and an ATP- binding site. It also contains ATPase enzyme that split ATP to
generate energy for the contraction of muscle. The thin filaments are composed
of two interwined actin molecules. Actin has polypeptide subunits called
globular actin or G–actin and filamentous form or F-actin. Each thin filament
is made of two F-actins helically wound to each other. Each F-actin is a
polymer of monomeric G-actins. It also contains a binding site for myosin. The
thin filaments also contain several regulatory proteins like tropomyosin and troponin which help in regulating the contraction of muscles along
with actin and myosin.
The study of muscle is called myology.
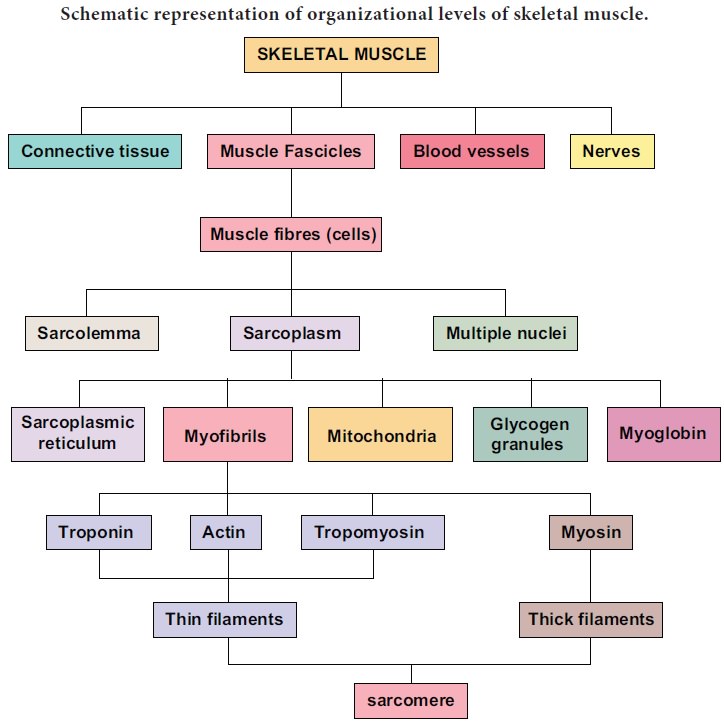
Schematic representation of organizational levels of skeletal muscle.
Related Topics