Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Fishes as social animals: reproduction
Sexual selection, dimorphism, and mate choice - Fishes as social animals: Courtship and spawning
Courtship and spawning
Sexual selection, dimorphism, and mate choice
Some traits of an animal function primarily to attract mates or to aid in battles between members of one sex for access to the other sex. Such sexually selected traits confer a mating advantage on an individual; they are a subset of natural selection, which usually involves traits that confer a survival advantage. Sexually selected traits may serve no other purpose than mating and may even handicap the possessor with respect to other, fitness-infl uencing activities. However, sexually selected traits can also confer a positive survival advantage, such as large size in males, which provides a physical defense from predators and is also favored by females during mating. Sexually selected traits are often referred to as secondary sexual characteristics. Primary characteristics include ovipositors, genitalia, and other copulatory structures such as claspers in elasmobranchs, gonopodia or priapia in livebearers and phallostethids, or brood patches or pouches and other structures used in parental care. In some instances, a character may be both secondary and primary, serving both in mate attraction and in copulation or parental care. In sticklebacks, male sticklebacks are attracted to females with swollen bellies, but the swelling results from the female’s ripe ovaries (Wootton 1976).
Secondary sexual characteristics have four general attributes: they are restricted to or are expressed differentially in one sex (usually the male), they do not appear until maturation, they often develop during a breeding season and then regress, and they generally do not enhance survival. Secondary characteristics take the form of sexual dimorphisms (differences in body parts between sexes), such as differences in body size, head shape, fin shape, dentition, and body ornamentation, or as dichromatisms (differences in coloration). We know comparatively little about electrical, chemical, and acoustic differences between the sexes, although differences in anatomy and physiology associated with these sensory modes are common (e.g., elephantfishes, salmons, minnows, gymnotid knifefishes, toadfishes, croakers, damselfishes, gobies). For example,males of the Plainfin Midshipman, Porichthys notatus(Batrachoididae), attract females by “humming”, a sound produced by contracting large muscles attached to the gas bladder walls. The sound-producing ability and its importance in courtship are reflected in numerous differences between the sexes. Males differ from females in having larger body size, different color, larger sonic muscles, and differing neural circuitry – involving larger cell bodies, dendrites, and axons in the brain – than are found in the females (Bass 1996).
Whereas the vast majority of fish species show no obvious sexual dimorphisms, many species are distinctly dimorphic. Males are often the larger sex in salmon, sauries, wrasses, and clingfishes, whereas females are larger in Mackerel and Whale sharks, sturgeon, true eels, ceratioid anglers, sticklebacks, halfbeaks, silversides, livebearers, blennies, and billfishes. Male Dolphinfish (Coryphaena hippurus, Coryphaenidae) are larger and have a distinctly blunter head than females. During spawning migrations, male anguillid eels develop larger eyes than females, and male salmon develop distinctly concave upper and lower jaws, called kype, that preclude feeding. Males may have trailing filaments at the ends of their dorsal, anal, or caudal fins (characins, African rivulines, rainbowfishes, anthiine seabasses, cichlids, wrasses); enlarged median or paired fins (lampreys, bichirs, freshwater flyingfishes, minnows, characins, killifishes, livebearers, dragonets, gobies, climbing gouramis); elongate tail fins (livebearers); or have elongate, lobule-tipped gill covers or pelvic fins that are displayed in front of females during courtship (Corynopoma, Characidae; Ophthalmochromis, Cichlidae). In elasmobranchs, males may have longer or sharper teeth than females, which serve to grasp the female during courtship and copulation; females in turn may have thicker skin than males.
Differences in body ornamentation include small bumps on the body, scales, and fins of mostly male fishes in 25 different families of primarily soft-rayed teleosts. These bumps are called nuptial or breeding tubercles when of epidermal origin, or contact organswhen of dermal origin (Collette 1977) (Figs 21.2, 21.3). Scale differences also include scale type: male cyprinodontids have ctenoid scales, whereas females have cycloid scales (Berra 2001). Other dimorphic ornaments include small hooks on the anal fins and even tail of some male characins (a kind of contact organ), and rostral papillae in male blind cavefishes, photophore patterns in lanternfishes, and pigmented egg dummies on the anal fins of cichlids. Some male minnows, cichlids, wrasses, and parrotfishes develop bony or fatty humps on the front (nuchal region) of their head.
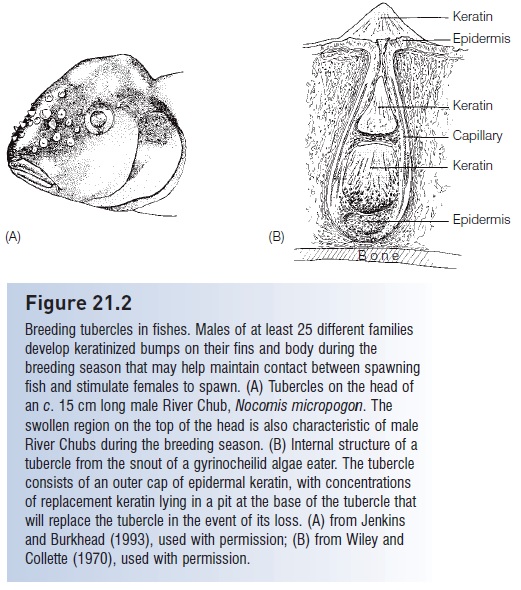
Figure 21.2
Breeding tubercles in fishes. Males of at least 25 different families develop keratinized bumps on their fins and body during the breeding season that may help maintain contact between spawning fish and stimulate females to spawn. (A) Tubercles on the head of an c. 15 cm long male River Chub, Nocomis micropogon. The swollen region on the top of the head is also characteristic of male River Chubs during the breeding season. (B) Internal structure of a tubercle from the snout of a gyrinocheilid algae eater. The tubercle consists of an outer cap of epidermal keratin, with concentrations of replacement keratin lying in a pit at the base of the tubercle that will replace the tubercle in the event of its loss. (A) from Jenkins and Burkhead (1993), used with permission; (B) from Wiley and Collette (1970), used with permission.

Figure 21.3
Extreme sexual dimorphism in a male Bluehead Chub, Nocomis leptocephalus. This c. 20 cm minnow builds pebble nests on stream bottoms and attracts females with its swollen head and distinct breeding tubercles. Photo by P. Vecsei, used with permission
Coloration differences are widespread and are usually expressed as more brightly colored males, either permanently (Bowfin, livebearers, killifishes, rainbowfishes, cichlids, wrasses, anabantids) or seasonally (minnows, sticklebacks, darters, sunfishes, cichlids). Male color change often involves development of bright or dark patches where they are most conspicuous because they break the rules of crypticity. Hence dark and light patches exist adjacent to one another, color transitions become sudden rather than gradual, or reverse counter shading develops (e.g., sticklebacks, sunfishes, temperate wrasses). Conspicuousness in the breeding season reinforces the premise that animals are willing to risk an increased predatory threat for a chance to reproduce (Breder & Rosen 1966; Fryer & Iles 1972; Meisner 2005).
The importance of sexually selected, dimorphic traits as species-isolating mechanisms has become increasingly obvious in recent years as human-caused alterations of the landscape interfere with mating patterns. In Lake Victoria, where “probably more vertebrate species are at imminent risk of extinction . . . than anywhere else in the world” (Ribbink 1987, p. 22), cichlids are separated taxonomically and apparently behaviorally by color variation in males. Females discriminate among closely related male suitors at least in part on color differences. Increased algal growth, resulting from heavy runoff of nutrients from the surrounding deforested hillsides (see Introduced predators), has increased turbidity and severely limited light transmission, especially at the long (red) and short (blue) ends of the visible light spectrum (Seehausen et al. 1997a) (Fig. 21.4A). Mate selection is apparently impaired by this absorption of long and short wavelengths because distinct red and blue colors in males are no longer obvious, leading to altered perception of mating colors and increased hybridization (Fig. 21.4B). “Where eutrophication turns the lights off, ecological and species diversity erode rapidly” (Seehausen et al. 1997a, p. 1810).
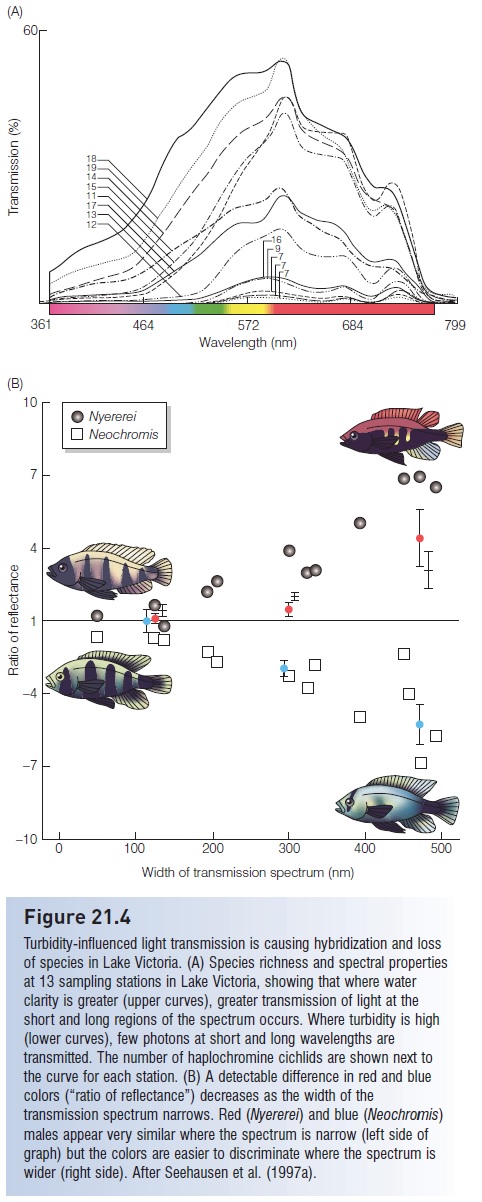
Figure 21.4
Turbidity-influenced light transmission is causing hybridization and loss of species in Lake Victoria. (A) Species richness and spectral properties at 13 sampling stations in Lake Victoria, showing that where water clarity is greater (upper curves), greater transmission of light at the short and long regions of the spectrum occurs. Where turbidity is high (lower curves), few photons at short and long wavelengths are transmitted. The number of haplochromine cichlids are shown next to the curve for each station. (B) A detectable difference in red and blue colors (“ratio of reflectance”) decreases as the width of the transmission spectrum narrows. Red (Nyererei) and blue (Neochromis) males appear very similar where the spectrum is narrow (left side of graph) but the colors are easier to discriminate where the spectrum is wider (right side). After Seehausen et al. (1997a).
Other researchers have linked alterations and reductions in spawning behavior to increased turbidity. Sexual selection was relaxed under conditions of experimentally induced turbidity in the Sand Goby, Pomatoschistus minutus. In clear water, females showed a strong preference for larger males, but this preference weakened as water clarity was reduced (Jarvenpaa & Lindstrom 2004). Hence, increased turbidity can alter mating systems by altering the intensity of sexual selection. Other, related findings include a reduction in the intensity of red coloration in male three-spined sticklebacks in the Baltic Sea, linked to eutrophication-caused algal growth and reduced visibility (Candolin et al. 2007). Decreased spawning frequency, disrupted timing of spawning, and a 93% reduction in number of viable eggs produced by the Tricolor Shiner, Cyprinella trichroistia, were interpreted as a result of disrupted visual cues associated with increased turbidity and resulting changes in light transmission (Burkhead & Jelks 2001).
Visual communication is not the only mode affected by anthropogenic impacts. Species recognition in the swordtail, Xiphophorus birchmanni, is chemically mediated, with females preferring conspecific males based on chemical cues. Fish tested in clean water maintain that species preference. Females tested in water subjected to sewage effl uent and agricultural runoff did not discriminate between conspecificc males and males of X. malinche, with which they can hybridize. The chemical most likely to interfere with communication was identified as humic acid, “a ubiquitous, natural product elevated to high levels by anthropogenic processes” (Fisher et al. 2006, p. 1187). It seems reasonable to assume that other sensory modalities involved in courtship and species recognition, such as acoustic and electrical
Related Topics