Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Skeleton, skin, and scales
Scales - Integumentary skeleton of Fishes
Scales
Scales are the characteristic external covering of fishes. There are four basic types of scales.
1 Placoid scales are characteristic of the Chondrichthyes, although they have a more restricted distribution inrays and chimaeras than in sharks. This type of scale has been called a “dermal denticle”, but this is notaccurate terminology because there are both epidermal and dermal portions, as in mammalian teeth. Eachplacoid scale consists of a flattened rectangular basal plate in the upper part of the dermis, from which a protruding spine projects posteriorly on the surface. The outer layer of the placoid scale is hard, enamel likevitrodentine, derived from ectoderm. Vitrodentineis no cellular and has a very low organic content. The scale has a cup or cone of dentine with a pulp cavity richly supplied with blood capillaries, just as in mammalian teeth. Placoid scales do not increase in sizewith growth; instead, new scales are added betweenolder scales. The teeth of elasmobranchs are evolutionary derivatives of placoid scales, and in factplacoid scales are homologous with teeth in allvertebrates.
2 Cosmoid scales were present in fossil coelacanths andfossil lungfishes. The scales of Recent lungfishes arehighly modified by loss of the dentine layer. Cosmoidscales are similar to placoid scales and probably arose from the fusion of placoid scales. Cosmoid scales arecomposed of two basal layers of bone: isopedine,which is the basal layer of dense lamellar bone, andcancellous (or spongy) bone, which is supplied withcanals for blood vessels. Over the bone layers is alayer of cosmine, a noncellular dentinelike substance.Over the cosmoid layer is a thin superficial layer ofvitrodentine. Growth is by addition of new lamellar bone underneath, not over the upper surface.
3 Ganoid scales were present in primitive fossilactinopterygians and are found in Chondrostei. Theyare modified cosmoid scales, with the cosminereplaced by dentine and the surface vitrodentinereplaced by ganoine, an inorganic bone salt secretedby the dermis. Ganoine is a calcified noncellularmaterial without canals. Ganoid scales are usuallyrhomboidal in shape and have articulating peg andsocket joints between them. The fossil palaeoniscoidscale is least modified in the bichirs, Polypteridae(three layers: ganoine, dentine, and isopedine). Ganoid scales are more modified in sturgeons (Acipenseridae)and paddlefishes (Polyodontidae), in which lamellae ofganoine lie above a layer of isopedine. Sturgeon scalesare modified into large plates, with most of the rest ofthe body naked.Scales of gars (Lepisosteidae) are similar to Polypteridae in external appearance but are moresimilar to those of the Acipenseridae andPolyodontidae in structure. In the Bowfin (Amia) thescale is greatly reduced in thickness to merely a collagenous plate with bony particles, very similar to the cycloid scales of Teleostei.
4 Cycloid and ctenoid scales are almost completely dermal. There is no enamel-like layer except perhaps thectenii (teeth on posterior border) and the most posterior and superficial ridges of the scale. Thesetypes of scales evolved from ganoid scales by loss ofthe ganoine and thinning of the bony dermal plate.Two major portions make up these scales: (i) a surface “bony” layer, which is an organic frameworkimpregnated with salts, mainly calcium phosphate (as hydroxyapatite) and calcium carbonate; and (ii) a deeper fibrous layer, or fi brillary plate, composedlargely of collagen.
Cycloid or ctenoid scales are present in the Teleostei,the vast majority of bony fishes. They have the advantage of being imbricate, overlapping like shingles on a roof ,which gives great flexibility compared with cosmoid andganoid scales. Small muscles pull unequally on the dermis,causing the anterior portion of the scale to becomedepressed in the dermis and covered over by the posteriormargin of the preceding scale. Cycloid scales lack ctenii.Breeding tubercles and contact organs (see Fig. 21.2) arepresent in many groups of fishes that lack ctenoid scales.
Including all scales with spines on their posterior margins under the term ctenoid is an oversimplification ofthe situation (Johnson 1984; Roberts 1993). Three different,general types of spined scales exist: (i) crenate, withsimple marginal indentations and projections; (ii) spinoid,with spines continuous with the main body of the scales;and (iii) ctenoid, with ctenii formed as separate ossifications distinct from the main body of the scale (Roberts1993). Crenate scales occur widely in the Elopomorphaand Clupeomorpha; spinoid scales occur widely in theEuteleostei; peripheral ctenoid scales (whole ctenii in onerow) occur, probably independently, in the Ostariophysi,Paracanthopterygii, and Percomorpha; and transformingctenoid scales (ctenii arising in two or three rows andtransforming into truncated spines) are a synapomorphy of the Percomorpha.
As with fish skin, the chemical composition of scales ispoorly known. About 41–84% is organic protein, mostly albuminoids such as collagen (24%) and ichthylepidin(76%). Up to 59% is bone, mostly Ca3(PO4)2 and CaCO3.
Phylogenetic significance of scale types
Scales have been used as a taxonomic tool since the beginningsof systematic ichthyology (Roberts 1993). Forexample, Louis Agassiz divided fishes into four groupsbased on their scale type. More recent classifications arebased on more characters but are similar to the system usedby Agassiz.
Whereas most groups of advanced acanthopterygian teleost`shave ctenoid scales, some “ctenoid” groups may also have cycloid scales, and many species will have ctenoidscales on some parts of the body and cycloid scales onothers. In the flatfishes, Pleuronectiformes, some specieshave ctenoid scales on the eyed side and cycloid scales on the blind side that is in contact with the bottom. Someflatfishes are sexually dimorphic, males having ctenoidscales and females having cycloid scales.
Scale size varies greatly in fishes. Scales may be microscopicand embedded as in freshwater eels (Anguillidae),which led to their being classified as non-kosher because ofthe supposed absence of scales. Scales are small in mackerels (Scomber), “normal” in perches (Perca), large enough toe used for junk jewelry in Tarpon (Megalops), and huge(the size of the palm of a human hand) in the IndianMahseer (Tor tor, a cyprinid gamefish reaching 43 kg inweight).
Development pattern of scales
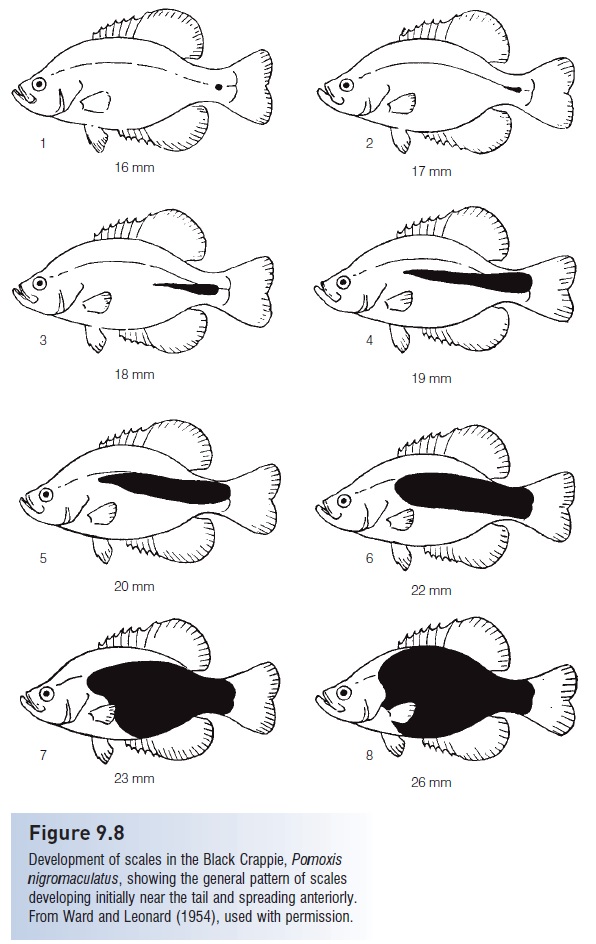
In actinopterygian (ray-finned) fishes, scales usually develop first along the lateral line on the caudal peduncle, then in rows dorsal and ventral to the lateral line, and then spread anteriorly (see Fig. 9.8). The last regions to develop scalesin ontogeny are the first to lose scales in phylogeny. Oncethe full complement of scales is attained in ontogeny, thenumber remains fixed. Therefore, the number of scales is auseful taxonomic character. Most scales remain in place forthe life of the fish, which makes scales valuable in recordingevents in the life history of an individual fish, such asreduced growth that generally occurs during the winter orduring the breeding season. Scales become deeply buried inthe skin with age in the Swordfish, Xiphias (Govoni et al.2004), leading some orthodox Jews to question if Swordfish are kosher, because kosher dietary laws require that a fish have both fins and scales.
Geographic variation can occur in the relative developmentof ctenoid scales in some species. For example, in the Swamp Darter (Etheostoma fusiforme) of the AtlanticCoastal Plain of the United States, the number of scales inthe interorbital area increases from north to south (Collette1962). In northern parts of the range, the few scales presentare embedded and cycloid. Further south, there is anincrease in number and in relative “ctenoidy” of the scales;more scales have the posterior surface of the scale projectingthrough the epidermis, and these scales have morectenii on them.
Lateral line scales form pores on scales from head to tail.Most fishes have complete lateral lines, that is, pored scales extend from behind the opercular region all the way to thebase of the caudal fin. Some species, such as the Swamp Darter, have incomplete lateral lines, with the pores extendingonly part way to the caudal base. Other patterns includedisjunct where there is an interruption between the upperand lower portions of the lateral line, as in most membersof the large family Cichlidae, multiplewith several lateral lines, and absent, where the lateral line is missing on thebody (Webb 1989).
Modifications of scales
Some fishes have scales that are deciduous, that is, easily shed. This is true of many species of herrings (Clupeidae)and anchovies (Engraulidae). It may be true of onespecies in a genus but not of another. For example, oftwo species of common Australian halfbeaks or garfishes(Hemiramphidae), scales remain in the River Garfish, Hyporhamphus regularis, but are easily lost in the Sea Garfish, H. australis.
Male darters of the genus Percina have caducous scales, a single row of enlarged scales along the ventral surface between the pelvic fins and the anus. Several structures inchondrichthyans may have arisen from fusions of modified placoid scales. These include the “spines” at the beginningof the first and second dorsal fins of the Spiny Dogfish, theprominent dorsal “spine” in some chimaeras (Holocephali),the caudal fin spine in stingrays (Dasyatidae), and the teeth on the rostrum of sawfishes (Pristis).
As mentioned earlier, the structure of placoid scales in Chondrichthyes is the same as the structure of teeth in vertebrates, leading to the question: Which came first? Did some primitive chondrichthyan ancestor develop teeth that then spread over the body? Or did the ancestor first developscales that then spread into the mouth and became modified into teeth? Apparently, the dermal armor of the earliest known vertebrates, the ostracoderms, broke up into smaller units, and some of these scales in the mouth evolved into teeth (Walker & Liem 1994).
In many teleost`s, there is an external dermal skeleton in addition to the internal supporting skeleton. This is composedof segmented bony plates in pipefishes (Syngnathidae)and poachers (Agonidae) and bony shields similar to placoid scales with vitrodentine in several South Americanarmored catfish families such as the Loricariidae. The body is enclosed in a bony cuirass (armor) in the shrimpfishes(Centriscidae) and is completely enclosed in a rigid bonybox in the trunkfishes (Ostraciidae).
Many fishes have protective scutes or spines. Theventral row of scales is modified into scutes with sharp,posteriorly directed points in herrings, such as the river herrings (Alosa) and the threadfins (Harengula). Somejacks (Carangidae) have lateral scutes along the posterior part of the lateral line. Sticklebacks (Gasterosteidae) have bony lateral plates. These plates vary in number andsize in Gasterosteus aculeatus, roughly correlated with the salinity of the habitat and the presence or absence of predators. Sharp erectable spines derived from scalesare present in porcupine fishes (Diodontidae). Largebony “warts” characterize lumpfishes (Cyclopterus). Surgeonfishes (Acanthuridae) are so named because of the pair of sharp, anteriorly directed spines on the caudal peduncle.
Three other modifications of scales are discussed elsewhere. Lateral line scales bear sensory structures. Lepidotrichia, fin rays supporting the fins, probably originated from scales (see above). The superficial bones of the skull originated as scales and have become modified into dermal bones (see above).
Related Topics