Chapter: Medical Surgical Nursing: Pain Management
Routes of Administration - Pain Management Strategies
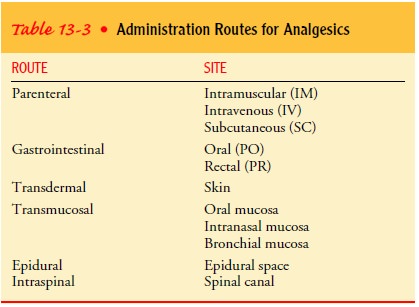
ROUTES
OF ADMINISTRATION
The route selected for
administering an analgesic agent (Table 13-3) depends on the patient’s
condition and the desired effect of the medication. Analgesic agents can be
administered by par-enteral, oral, rectal, transdermal, transmucosal,
intraspinal, or epidural routes. Each method of administration has advantages
and disadvantages. The route chosen should be based on the pa-tient’s needs.
Parenteral
Parenteral administration (intramuscular, intravenous, or subcu-taneous) of the analgesic medication produces effects more rapidly than oral administration, but these effects are of shorter duration.
Parenteral
administration may be indicated if the pa-tient is not permitted oral intake or
is vomiting. Medication ad-ministered by the intramuscular route enters the
bloodstream more slowly than medication given intravenously and is metabo-lized
slowly. The rate of absorption may be erratic; it depends on the site selected
and the amount of body fat.
The intravenous route is an alternative to intramuscular injec-tion for
many but not all analgesic medications. The intravenous route is the preferred
parenteral route in most acute care situations because it is much more
comfortable for the patient. In addition, peak serum levels and pain relief
occur more rapidly and reliably. Because it peaks rapidly (usually within
minutes) and is metabo-lized quickly, an appropriate intravenous dose will be
smaller and prescribed at shorter intervals than an intramuscular dose.
Intravenous opioids may
be administered by IV push or slow push (eg, over a 5- to 10-minute period) or
by continuous infu-sion with a pump. Continuous infusion provides a steady
level of analgesia and is indicated when pain occurs over a 24-hour pe-riod
(eg, after surgery for the first day or so, or in a patient with prolonged
cancer pain who cannot take medication by other routes). The dose of analgesic
agent is calculated carefully to re-lieve pain without producing respiratory
depression and other side effects.
The subcutaneous route for infusion of opioid analgesic agents is used
for patients with severe pain such as cancer pain; it is par-ticularly useful
for patients with limited intravenous access who cannot take oral medications,
and patients who are managing their pain at home. The dose of opioid that can
be infused through this route is limited because of the small volume that can
be administered at one time into the subcutaneous tissue. How-ever, this route
is often an effective and convenient way to man-age pain.
Oral Route
If the patient can take medication by mouth, oral administration is
preferred over parenteral administration because it is easy, non-invasive, and
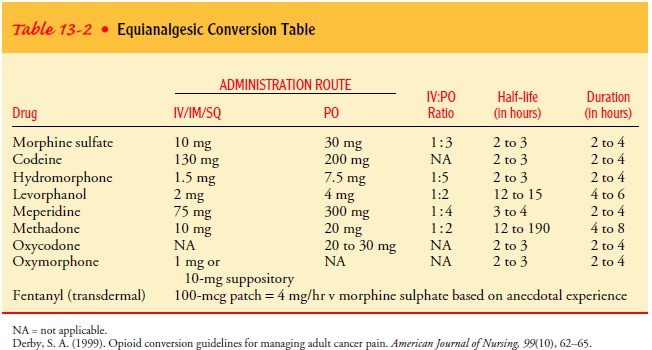
not painful. Severe pain can be relieved with oral opioids if the doses are
high enough (see Table 13-2).
In terminally ill patients with prolonged pain, doses may gradually be
increased as the disease progresses and causes more pain or as the person
builds up a tolerance to the medication. If these higher doses are increased
gradually, they usually provide additional pain relief without producing
respiratory depressionor sedation. If the route of administration is
changed from a par-enteral route to the oral route at a dose that is not
equivalent in strength (equianalgesic), the smaller oral dose may result in a
withdrawal reaction and recurrence of pain.
Rectal Route
The rectal route of administration may be indicated in patients who
cannot take medications by any other route. The rectal route may also be
indicated for patients with bleeding problems, such as hemophilia. The onset of
action of opioids administered rec-tally is unclear but is delayed compared
with other routes of administration. Similarly, the duration of action is
prolonged.
Transdermal Route
The transdermal route
has been used to achieve a consistent opi-oid serum level through absorption of
the medication via the skin. This route is most often used for cancer patients
who are at home or in hospice care and who have been receiving oral
sustained-release morphine. Fentanyl (Duragesic) is the only commercially available
transdermal medication. The preparation is a patch consisting of a reservoir
containing the medication and a membrane.
When the transdermal system is first applied to the skin, the fentanyl,
which is fat-soluble, binds to the skin and fat layers. Then it is slowly and
systemically absorbed. Therefore, there is a delay in effect while the dermal
layer is being saturated. A drug reservoir actually forms in the upper layer of
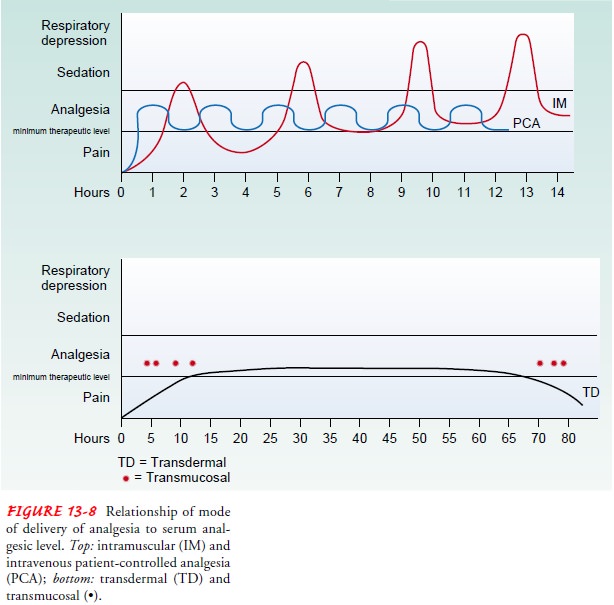
skin. This results in a slowly rising serum level and a slow tapering of the serum
level once the patch is removed (see Fig. 13-8). Because it takes 12 to 24
hours for the fentanyl levels to gradually increase from the first patch, the
last dose of sustained-release morphine should be given at the same time the
first patch is applied (Donner et al., 1996). Transdermal fentanyl is
associated with slightly less constipation than oral opioids. Absorption is
increased in the febrile patient. A heating pad should never be applied to the
area where the patch is applied. Transdermal fentanyl is much more expensive
than sustained-release morphine but less costly than methods that de-liver
parenteral opioids.
Once it is determined that switching from other routes of morphine
administration to the patch is appropriate, the correct dosage for the patch
must be calculated. If the patient uses an opioid other than morphine,
conversion to milligrams of oral morphine is the first step. After determining
how many mil-ligrams of morphine (or morphine equivalents) the patient has been
using over 24 hours, an initial dose of transdermal fentanyl can be calculated.
Pasaro (1997) suggests one method of calculating the initial dose of
fentanyl: the patient’s daily dose of morphine is divided by two. Thus, the
equivalent of 400 mg morphine used per day would be equivalent to 200 g
fentanyl per hour. Patients switched from morphine to fentanyl need to be
assessed not only for pain and potential side effects but also for dependence,
reflected by withdrawal symptoms, which may consist of shivering, a feel-ing of
coldness, sweating, headache, and paresthesia (Puntillo, Casella et al., 1997).
Patients may require short-acting opioids for breakthrough pain before the
systemic fentanyl level reaches a therapeutic level.
These conversions and the conversion-type table in the trans-dermal
fentanyl packet insert should be used only to establish the initial dose of
fentanyl when the patient switches from oral mor-phine to fentanyl (and not
vice versa). These tables and equationsare not meant to be used to determine
the dosages of oral mor-phine for a patient who has been receiving transdermal
fentanyl. Many patients will not achieve satisfactory analgesia from the
initial dose of transdermal fentanyl and will require an increase in their
fentanyl dose to treat breakthrough pain. If the table or equation is used
incorrectly to calculate a morphine dose, there is a risk of overdose. If the
patient requires a change from trans-dermal fentanyl back to oral or
intravenous morphine (as in the case of surgery), the patch should be removed
and intravenous morphine supplied on an assessed need basis.
Before applying a new
patch, the patient should be carefully checked for any older, forgotten
patches. These should be dis-carded. Patches should be replaced every 72 hours.
Transmucosal Route
The person with cancer pain who is being cared for at home may be
receiving continuous opioids using sustained-release morphine, hydromorphone,
oxycodone, transdermal fentanyl, or other med-ications. These patients often
experience short episodes of severe pain (eg, after coughing or moving), or
they may experience sud-den increases in their baseline pain resulting from a
change in their condition. These periods, called breakthrough pain, can be well
managed with an oral dose of a short-acting transmucosal opioid that has a
rapid onset of action. Currently the only transmucosal opioid available is
fentanyl, a lozenge on an applicator stick (often referred to as a lollipop by
patients).
Currently the only
approved and commercially available transmucosal opioid analgesic agents in a
nasal spray form are bu-torphanol (Stadol) and fentanyl. Butorphanol is a
complex med-ication that simultaneously acts to induce or promote (agonist) and inhibit or reverse (antagonist) opioid effects. It works
like an opioid agonist and an opioid antagonist at the same time. Butor-phanol
in any form cannot be combined with other opioids (eg, for cancer breakthrough
pain) because the antagonist com-ponent will block the action of the opioids
the patient is already receiving. The principal use of this agent is for brief,
moderate to severe pain, such as migraine headaches.
Intranasal fentanyl is useful in cancer-related breakthrough pain. Given
in this form, analgesia is achieved within 5 to 10 minutes and was rated as
achieving analgesia superior to oral morphine by 50% of patients in one study
(Zeppetella, 2000).
Intraspinal and Epidural Routes
Infusion of opioids or local anesthetic agents into the subarach-noid
space (intrathecal space or spinal canal) or epidural space has been used for
effective control of pain in postoperative patients and those with chronic pain
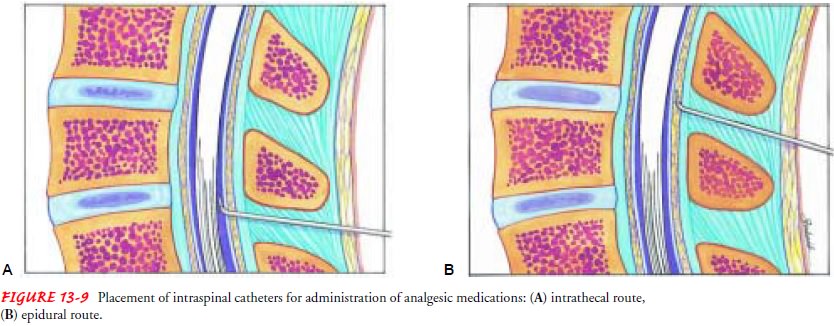
unrelieved by other methods. A catheter is inserted into the subarachnoid or
the epidural space at the thoracic or lumbar level for administration of opioid
or anes-thetic agents (Fig. 13-9). With intrathecal administration, the
medication infuses directly into the subarachnoid space and cerebrospinal
fluid, which surrounds the spinal cord. With epidural administration,
medication is deposited in the dura of the spinal canal and diffuses into the
subarachnoid space. It is be-lieved that pain relief from intraspinal
administration of opioids is based on the existence of opioid receptors in the
spinal cord.
Infusion of opioids and local anesthetic agents through an in-trathecal
or epidural catheter results in pain relief with fewer side effects, including
sedation, than with systemic analgesia. Adverse effects associated with
intraspinal administration include spinal headache resulting from loss of
spinal fluid when the dura is punc-tured. This is more likely to occur in
younger (less than 40 years of age) patients. The dura must be punctured with
the intrathecal route, and dural puncture may occur inadvertently with the
epidural route. When dural puncture inadvertently occurs, spinal fluid seeps
out of the spinal canal. The resultant headache is likely to be more severe
with an epidural needle because it is larger than a spinal needle, and
therefore more spinal fluid escapes.
Although respiratory depression generally peaks 6 to 12 hours after
epidural opioids are administered, it can occur earlier or up to 24 hours after
the first injection. Depending on the lipophilic-ity (affinity for body fat) of
the opioid injected, the time frame for respiratory depression can be short or
long. Morphine is hydro-philic, and the time for peak effect is longer compared
to fentanyl, which is a lipophilic opioid. All patients should be monitored
closely for at least the first 24 hours after the first injection, longer if
changes in respiratory status or level of consciousness occur. Opioid
antagonist agents such as naloxone must be available for intravenous use if
respiratory depression occurs.
The patient is also observed for urinary retention, pruritus, nausea, vomiting, and dizziness. Precautions must be taken to avoid infection at the catheter site and catheter displacement. Only medications without preservatives should be administered into the subarachnoid or epidural space because of the potential neurotoxic effects of preservatives.
During surgery, intrathecal opioids are used almost exclusively after a
spinal anesthetic agent is administered. For patients under-going large
abdominal surgical procedures, especially those at risk for postoperative complications,
a combination of a general in-haled anesthetic agent for the surgery and a
local epidural anes-thetic agent and epidural opioids administered after
surgery results in excellent pain control with fewer postoperative
complications.
Patients who have persistent, severe pain that fails to respond to other
treatments, or those who obtain pain relief only with the risk of serious side
effects, may benefit from medication administered by a long-term intrathecal or
epidural catheter. After the physician tunnels the catheter through the
subcutaneous tissue and places the inlet (or port) under the skin, the
medication is injected through the skin into the inlet and catheter, which
delivers the medication di-rectly into the epidural space. The medication may
need to be in-jected several times a day to maintain an adequate level of pain
relief.
In patients who require
more frequent doses or continuous in-fusions of opioid analgesic agents to
relieve pain, an implantable infusion device or pump may be used to administer
the medica-tion continuously. The medication is administered at a small,
con-stant dose at a preset rate into the epidural or subarachnoid space. The
reservoir of the infusion device stores the medication for slow release and
needs to be refilled every 1 or 2 months, depending on the patient’s needs.
This eliminates the need for repeated injec-tions through the skin.
NURSING MANAGEMENT OF SIDE EFFECTS
Headache resulting from spinal fluid loss may be delayed. There-fore,
the nurse needs to assess regularly for headache after either type of catheter
is placed. Should headache occur, the patient should remain flat in bed and
should be given large amounts of fluids (provided the medical condition
allows), and the physician should be notified. An epidural blood patch may be
carried out to reduce leakage of spinal fluid.
Cardiovascular effects
(hypotension and decreased heart rate) may result from relaxation of the
vasculature in the lower ex-tremities. Therefore, the nurse assesses frequently
for decreases in blood pressure, pulse rate, and urine output.
For patients experiencing urinary retention and pruritus, the physician
may prescribe small doses of naloxone. The nurse ad-ministers these doses in a
continuous intravenous infusion that is small enough to reverse the side
effects of the opioids without re-versing the analgesic effects.
Diphenhydramine (Benadryl) may also be used to relieve opioid-related pruritus.
PROMOTING HOME AND COMMUNITY-BASED CARE
The patient who receives epidural analgesic agents at home and the
family must be taught how to administer the prescribed med-ication using
sterile technique and how to assess for infection. The patient and family also
need to learn how to recognize side effects and what to do about them. Although
respiratory depres-sion is uncommon, urinary retention may be a problem, and
pa-tients and families must be prepared to deal with it if it occurs. Implanted
analgesic delivery systems can be safely and confi-dently used at home only if
health care personnel are available for consultation and, possibly,
intervention on short notice.
Related Topics