Chapter: Medical Immunology: Diagnostic Immunology
Radioimmunoassay
Radioimmunoassay
The introduction of radiolabeled components into immunoassays was a very significant development, because it made it possible to develop assays combining high specificity and high sensitivity.
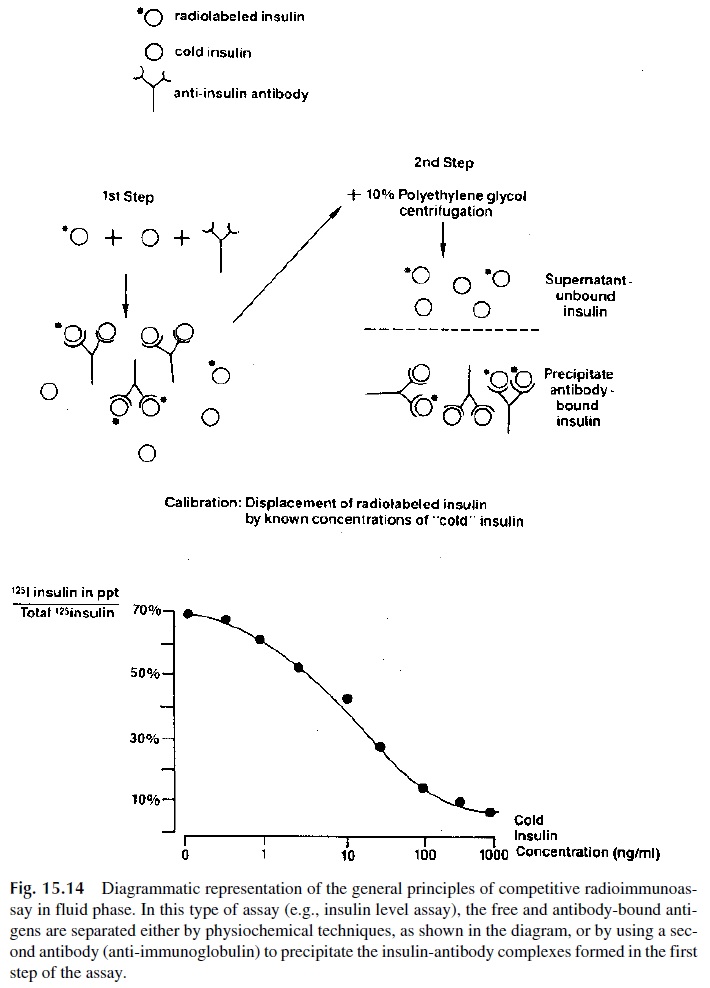
The first radioimmunoassays (RIAs) were based on the principle of competitive binding: unlabeled antigen competes with radiolabeled antigen for binding to antibody with the appropriate specificity. Thus, when mixtures of constant amounts of radiolabeled and variable amounts of unlabeled antigen are incubated with fixed concentrations of the cor-responding antibody, the amount of radiolabeled antigen that remains unbound is directly proportional to the quantity of unlabeled antigen in the mixture. One of the problems with the early assays was the separation of free antigen from antigen-antibody complexes. Usu-ally that was achieved by either cross-linking the antigen-antibody complexes with a sec-ond antibody or by means of the addition of reagents that promoted the precipitation of anti-gen-antibody complexes, such as ammonium sulfate or polyethylene glycol. Counting radioactivity in the precipitates allowed the determination of the amount of radiolabeled antigen co-precipitated with the antibody and a calibration curve was constructed by plot-ting the percentage of antibody-bound radiolabeled antigen against known concentrations of a standardized unlabeled antigen (Fig. 15.14). The concentrations of antigen in patient samples were extrapolated from that curve.
The introduction of solid-phase RIA simplified considerably the separation of free and antibody-bound radiolabeled antigen (Fig. 15.15). Depending on the objective of the assay, antigen or antibodies are immobilized by coupling or absorption to a solid surface (test tube wall, polystyrene beads, etc.). Competitive solid phase RIA for antigens are car-ried out by adding mixtures of labeled and unlabeled antigen to the solid phase containing immobilized antibody. Unbound reagents are washed off from the solid phase and ra-dioactivity is counted to determine the amount of labeled antibody retained in each one of the different mixtures tested. A calibration curve is constructed based on the binding of la-beled antigen in mixtures with a known amount of unlabeled antigen used as calibrator.
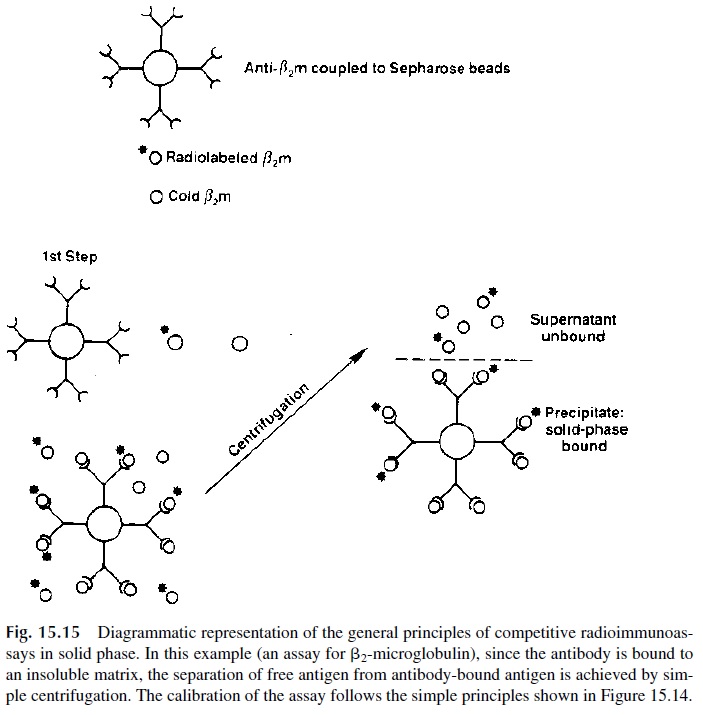
From that curve the concentrations of the same antigen in biological samples can be deter-mined, based on how much radiolabeled antigen from the mixtures with unknown samples became associated with the immobilized antibody.
Noncompetitive solid phase RIA for the detection of specific antibodies has also been described. The antigen is bound to the solid phase, the antigen-coated solid phase is exposed to a sample containing antibody, and a radiolabeled anti-human immunoglobulin is used to assay the antibody that becomes bound to the immobilized antigen.
Radioimmunoassays have been used with extremely good results in the assay of many different hormones (insulin, aldosterone, human follicle-stimulating hormone, pro-gesterone, testosterone, thyroxin, vasopressin, etc.), proteins ( α-fetoprotein, carcinoem-bryonic antigen, IgE, hepatitis B surface antigen), nucleic acids [DNA, vitamins (vitamin B12), drugs (digoxin, LSD, barbiturate derivatives], enzymes (pepsin, trypsin), etc.
The extremely high sensitivity of RIA, which can easily determine concentrations in ng/mL range or even lower, is the reason why so many applications were developed. The main drawbacks of RIA are the cost of equipment and reagents, the short shelf life of radi-olabeled compounds, and the problems associated with the disposal of radiolabeled sub-stances. In recent years, RIA have been virtually replaced by quantitative fluorescence as-says, chemiluminescence assays, and enzyme immunoassays.
Related Topics