Chapter: Plant Biochemistry: Phloem transport distributes photoassimilates to the various sites of consumption and storage
Proteinases mobilize the amino acids deposited in storage proteins
Proteinases mobilize the amino acids deposited in storage proteins
Our knowledge about the mobilization of the amino acids from storage proteins derives primarily from investigations of processes during seed germination. In most cases, germination is induced by the uptake of water, and as a result of this protein bodies fuse to form a vacuole. The hydrolysis of the storage proteins is catalyzed by proteinases, which are in part depos-ited as inactive pro-forms together with the storage proteins in the protein bodies. Other proteinases are newly synthesized and transferred via the lumen of the ER and the Golgi apparatus to the vacuoles (Fig. 14.2). These enzymes are initially synthesized as inactive pro-forms. Activation of these pro-proteinases proceeds by limited proteolysis, in which a section of the sequence is removed by a specific peptidase. The remainder of the polypep-tide represents the active proteinase.
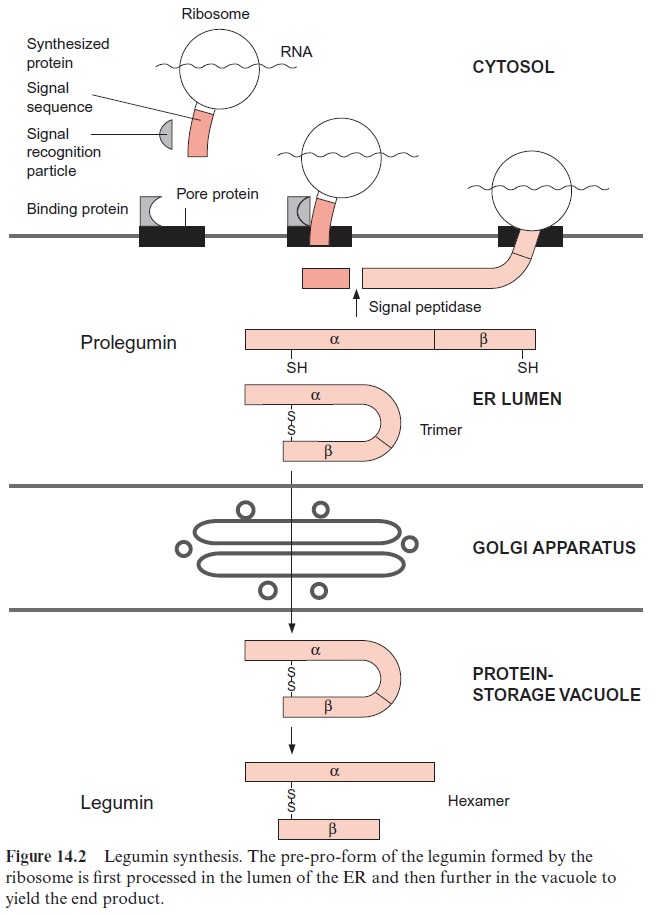
The degradation of the storage proteins is also initiated by limited pro-teolysis. A specific proteinase first removes small sections of the protein sequence, resulting in a conformational change of the storage protein. In cereal grains, S-S bridges of storage proteins are cleaved by reduced thiore-doxin . The unfolded protein is then susceptible to hydroly-sis by various proteinases, for example exopeptidases, which split off amino acids one after the other from the end of the protein molecule, and endopeptidases, which cleave within the molecule. In this way storage pro-teins are completely degraded in the vacuole and the liberated amino acids are provided as building material to the germinating plant.
Related Topics