Chapter: Essentials of Anatomy and Physiology: Development, Heredity, and aging
Prenatal Development
PRENATAL DEVELOPMENT
The human lifespan is usually considered the period between birth and death; however, the 9 months before birth are a critical part of existence. What happens in these 9 months profoundly affects the rest of a person’s life. Although most people develop normally and are born without defects, approximately 3 of every 100 people are born with a birth defect so severe that it requires medical attention during the first year of life. Later in life, many more people discover previously unknown problems, such as the tendency to develop asthma, certain brain disorders, or cancer.
The prenatal (pr̄e -nā ′ t̆a l; before birth) period, the period from conception to birth, can be divided into three parts: (1) During approximately the first 2 weeks of development, the primitive germ layers are formed; (2) from about the second to the eighth week of development, the major organ systems come into existence; and (3) during the last 7 months of the prenatal period, the organ systems grow and become more mature. Between the time of fertilization and 8 weeks of development, the developing human is called an embryo (em′ br̄e -ō ). From 8 weeks to birth, the develop-ing human is called a fetus (f̄e ′ tus; offspring).
To calculate the clinical age of an unborn child, the medi-cal community uses the mother’s last menstrual (men′ stroo-̆a l) period (LMP). An embryo or a fetus is therefore considered to bea certain number of days post-LMP. Most embryologists, on the other hand, use developmental age, which begins with fertilization,to describe the timing of developmental events. Because fertiliza-tion is assumed to occur approximately 14 days after LMP, the developmental age is 14 days less than the clinical age.
Fertilization
Fertilization is the union of a sperm cell and an oocyte, alongwith their genetic material (chromosomes), to produce a new individual. After sperm cells are ejaculated into the vagina, they are transported through the cervix and the body of the uterus to the uterine tubes, where fertilization occurs. The swimming abil-ity of the sperm cells and the muscular contractions of the uterus and uterine tubes are responsible for the movement of sperm cells through the female reproductive tract. Both oxytocin released by the female posterior pituitary and prostaglandins within the semen stimulate contractions in the uterus and uterine tubes.
While passing through the uterus and the uterine tubes, the sperm cells undergo capacitation. Capacitation (kă-pas′ i-tā′ shŭn) makes the sperm cells capable of releasing the concentrated enzymes contained in the acrosome, a region of the sperm cell head. The enzymes digest a pathway through the cumulus cells and the zona pellucida of the secondary oocyte (ō′ ō-sı̄t; egg cell). One sperm cell attaches to the oocyte cell membrane and enters the oocyte (figure 20.1, step 1).
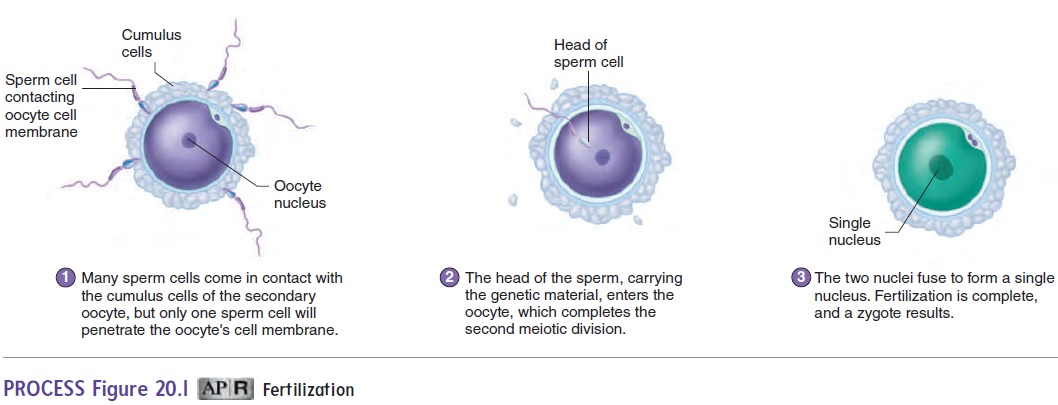
Hundreds of sperm cells reach the secondary oocyte, but nor-mally a change in the oocyte cell membrane prevents more than one sperm cell from entering the secondary oocyte. The secondary oocyte undergoes the second meiotic division only after a sperm cell enters it (figure 20.1, step 2). After the second meiotic division, the oocyte nucleus moves to the center of the cell, where it meets the nucleus of the sperm cell. Each of these nuclei has 23 chromo-somes. Their fusion, which completes the process of fertilization, produces a zygote (zı̄′ gōt; having a yoke) that has 46 chromosomes (figure 20.1, step 3). The zygote develops into the embryo.
Early Cell Division
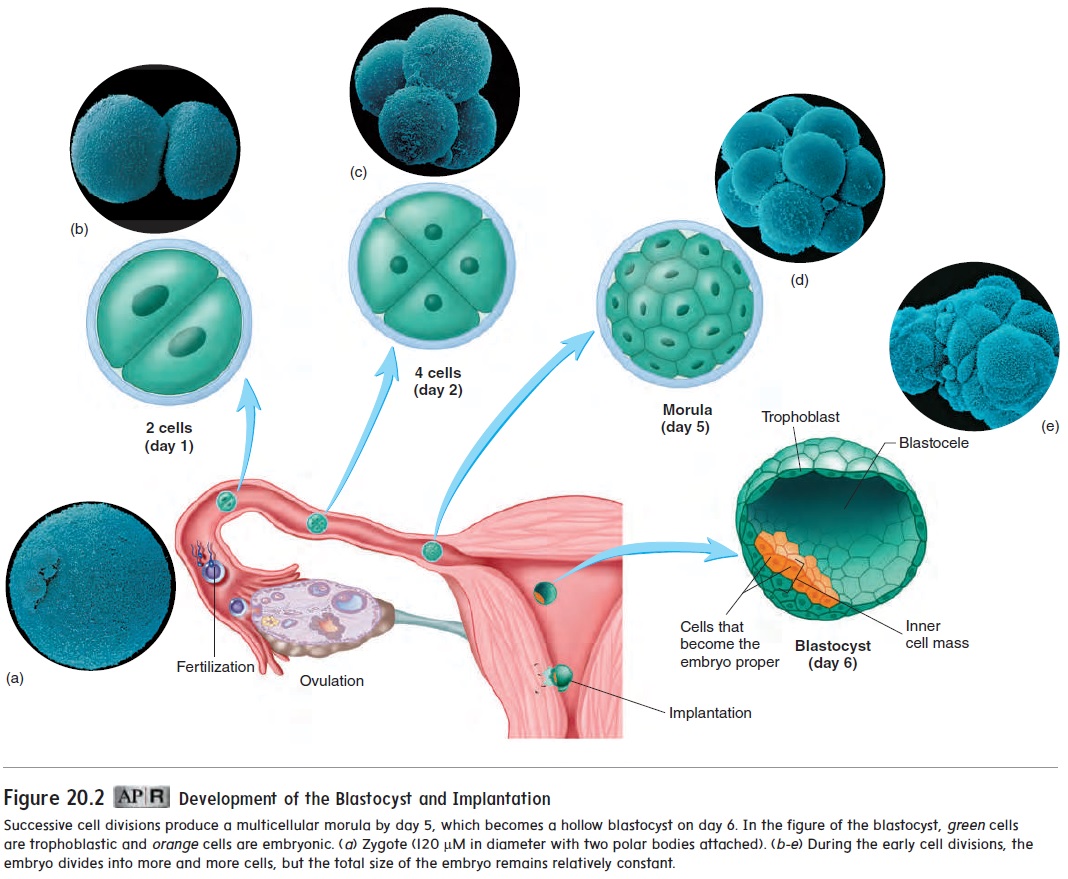
About 18–36 hours after fertilization, the zygote divides to form two cells (figure 20.2). Those two cells divide to form four cells, which divide to form eight, and so on. Even though the number of cells increases, the size of each cell decreases, so that the total mass of cells remains about the same size as the zygote. These cells have the ability to develop into a wide range of tissues. As a result, the total number of cells can be decreased, increased, or reorganized during this period without affecting normal development.
Blastocyst
After fertilization, multiple cell divisions have produced an embry-onic mass of about 12–16 cells, called a morula (mōr′ ū-lă; mul-berry) (figure 20.2). Most of the cells of the morula will not form the embryo proper but will form support structures, such as the placenta.
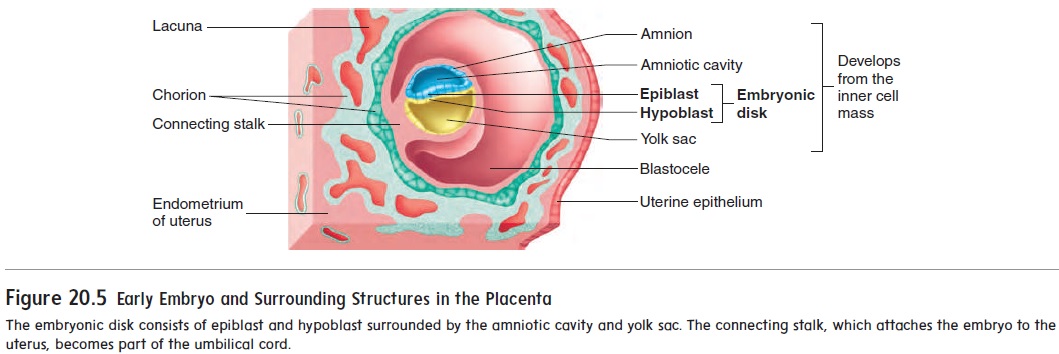
When a cavity begins to appear within the mass of cells, the whole structure is called a blastocyst (blas′ tō-sist) (figure 20.2). The fluid-filled cavity is called the blastocele(blas′ tō-sēl). Most of the blastocele is surrounded by a single layer of cells, but at one end of the blastocyst, the cells are several layers thick. This thick-ened area is called the inner cell mass. Not all cells of the blasto-cyst give rise to the new individual. The embryo proper, which will become the new individual, will develop from only a few cells of the inner cell mass. These cells are commonly referred to as stemcells because they give rise to all the cell types within the body.The remaining cells of the blastocyst are called the trophoblast (trof′ ō-blast, trō′ fō-blast), which forms the embryonic part of the placenta and the membranes (chorion and amnion) surrounding the embryo.
Implantation of the Blastocyst and Development of the Placenta
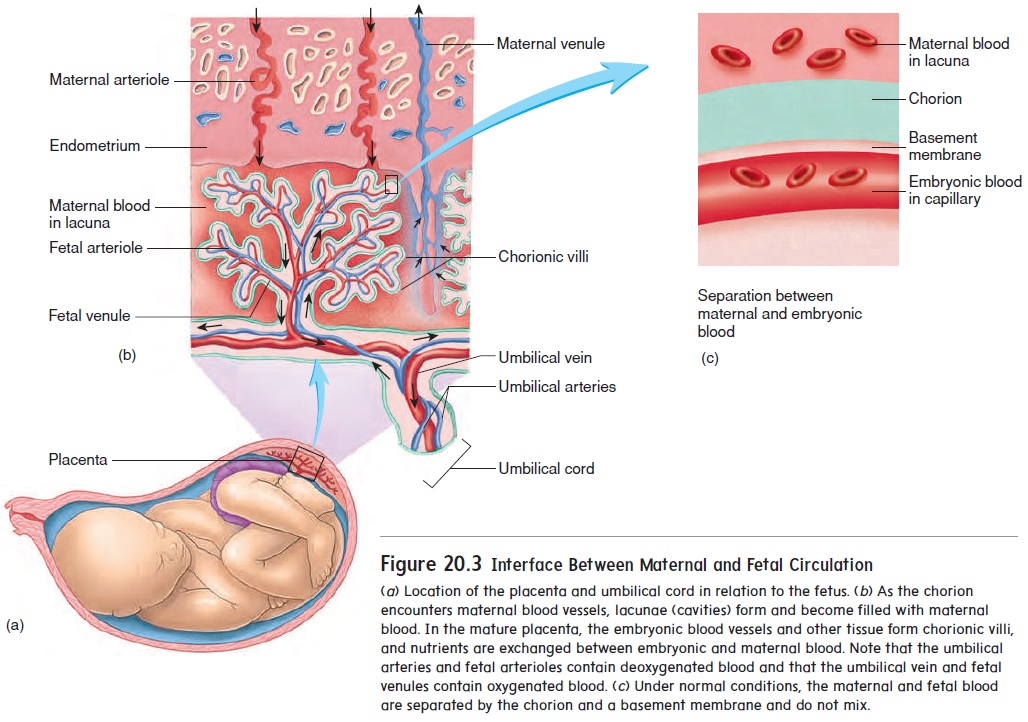
As the blastocyst burrows into the uterine wall, trophoblast cells, called the chorion (kō′ rē-on), form the embryonic portion of the placenta (plă-sen′ tă), the organ of nutrient and wasteproduct exchange between the embryo and the mother. Fingerlike projections, called chorionic villi, protrude into cavities formed within the maternal endometrium. Those cavities, called lacunae (lă-koo′ nē), are filled with maternal blood (figure 20.3). In the mature placenta, the embryonic blood supply is separated from the maternal blood supply by the embryonic capillary wall, a basement membrane, and a thin layer of chorion. As a result, the embryonic blood and maternal blood do not mix. Nutrients and waste products must cross this semipermeable barrier between the two circulations.
Initially, the embryo is attached to the placenta by a connecting stalk. As the embryo matures, the connecting stalk elongates and becomes the umbilical (ŭm-bil′ i-kăl; navel) cord (figure 20.3b). Within the umbilical cord, blood vessels carry blood from the embryo to the placenta and from the placenta to the embryo.
Maternal Hormonal
The chorion secretes human chorionic gonadotropin (gō′ nad-ō-trō′ pin) (hCG), which travels in the blood to the maternal ovary and causes the corpus luteum to remain functional. The secretion of hCG begins shortly after implantation, increases rapidly, and reaches a peak about 8 or 9 weeks after fertilization. Subsequently, hCG levels decline to a lower level, where they are maintained throughout the remainder of the pregnancy (figure 20.4). Most pregnancy tests are designed to detect hCG in either urine or blood.
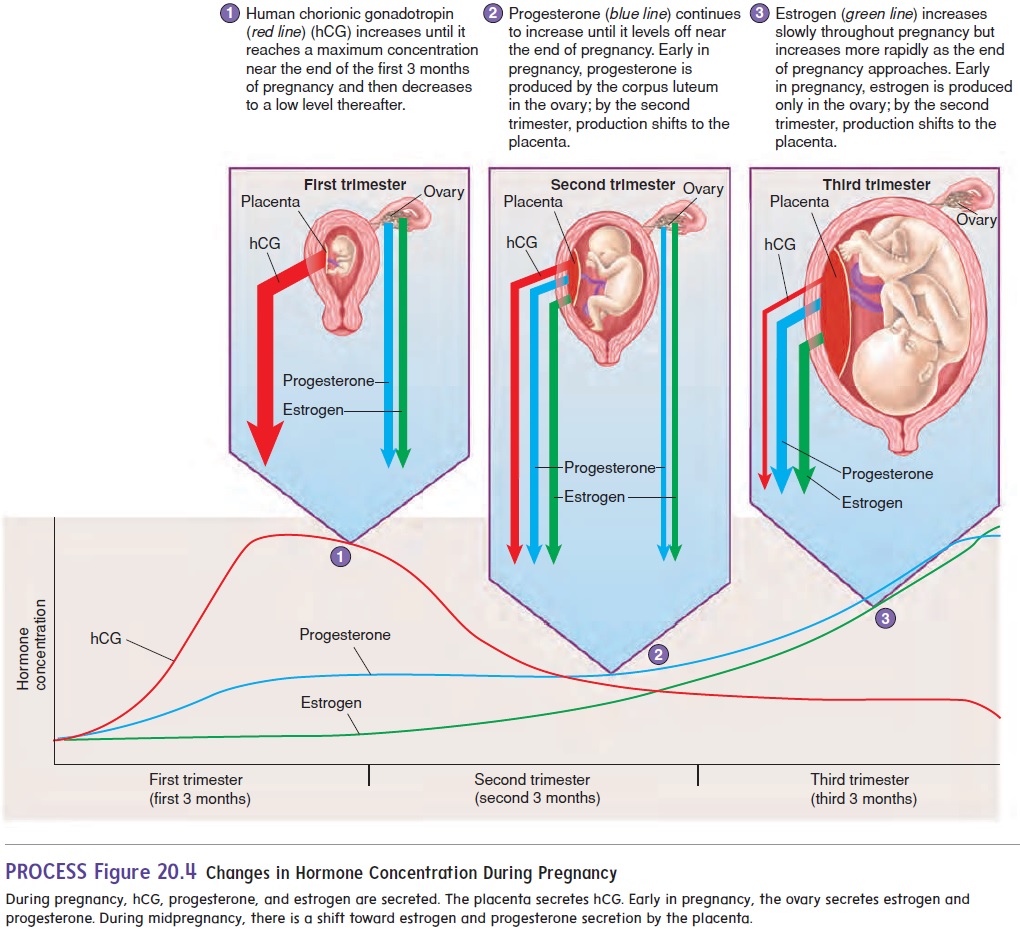
The estrogen and progesterone secreted by the corpus luteum are essential for maintaining the endometrium for the first 3 months of pregnancy. After the placenta forms, it also begins to secrete estrogen and progesterone. By the third month of pregnancy, the placenta has become an endocrine gland that secretes sufficient quantities of estrogen and progesterone to maintain pregnancy, and the corpus luteum is no longer needed. Estrogen and progesterone levels increase in the mother’s blood throughout pregnancy.
Formation of the Germ
After implantation, a new cavity, called the amniotic (am-nē-ot′ ik) cavity, forms inside the inner cell mass and causes the part of theinner cell mass nearest the blastocele to separate as a flat disk of tissue called the embryonic disk (figure 20.5). The amniotic cavity is bounded by a membrane called the amnion and is filled with amniotic fluid. The embryo will grow in the amniotic cavity,where the amniotic fluid forms a protective cushion. The embryonic disk at first is composed of two layers of cells: anepiblast adja-cent to the amniotic cavity and a hypoblast on the side of the disk opposite the amniotic cavity. A third cavity, the yolk sac, forms inside the blastocele from the hypoblast.
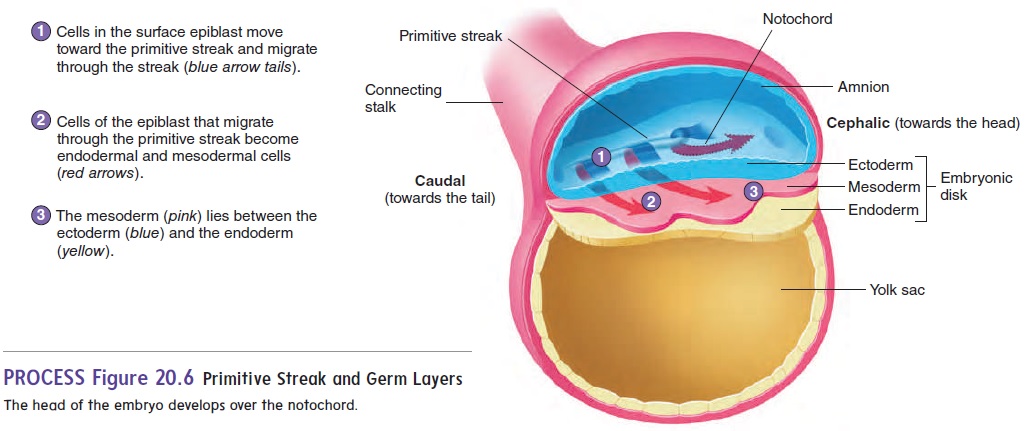
At about 14 days after fertilization, the embryonic disk has become a slightly elongated, oval structure. Some of the epiblast cells migrate toward the center of the disk, forming a thickened
The formation of the primitive streak establishes the central axis of the embryo. Some of the epiblast cells migrate through the primitive streak. Of these migrating cells, some displace the hypoblast to form the endoderm (en′ dō-derm; inside layer) while others emerge between the epi-blast and endoderm as a new germ layer, called the mesoderm (mez′ ō-derm; middle layer). Those epiblast cells that do not migrate form the ectoderm (ek′ tō-derm; outside layer). This pro-cess of cell migration and the formation of three distinct germ layersis called gastrulation. The embryo is now three-layered, having ectoderm, mesoderm, and endoderm (figure 20.6). All the tissues of an adult can be traced to these three germ layers (table 20.1).
From about day 14 until about day 35, the embryo is at maxi-mum risk from environmental toxins and drugs that can cause birth defects. The causes of birth defects are a major unsolved issue in biology at present. However, it appears that oxidative damage to key molecules in certain developing cells and/or changes in the DNA function within those cells may be involved.

A specialized group of cells at the cephalic (towards the head) end of the primitive streak moves from one end of the primitive streak to the other and, in some yet unknown way, organizes the embryo. A cordlike structure called the notochord (n̄o ′ t ̄o -k̄o rd) is formed by these cells as they move down the primitive streak. The notochord marks the central axis of the developing embryo (figure 20.6).
Neural tube and neural Crest Formation
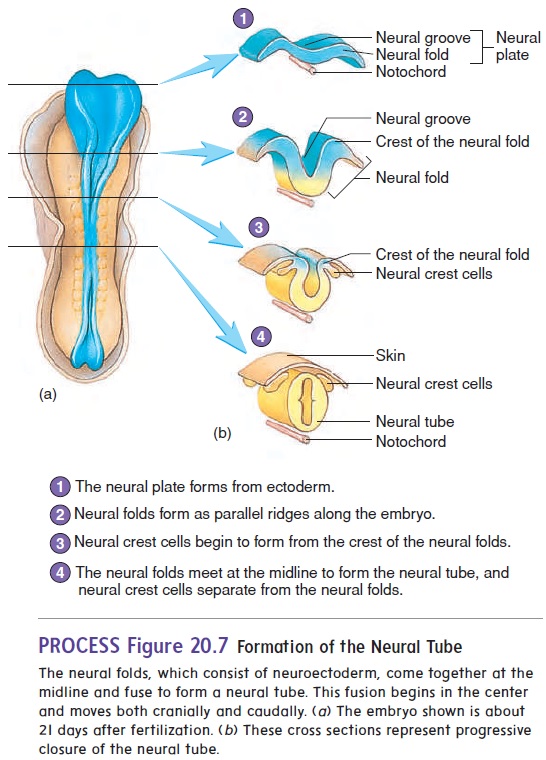
At about 18 days after fertilization, the ectoderm overlying the notochord thickens to form the neural plate. The lateral edges of the plate begin to rise like two ocean waves coming together. These edges are called the neural folds, and between them lies a neural groove. The neural folds begin to meet in the midline and fuse into aneural tube (figure 20.7). The cells of the neural tube are called neuroectoderm (noor-ō -ek′ tō -derm) (table 20.1). Neuroectoderm becomes the brain, the spinal cord, and parts of the peripheral nervous system. The neural tube becomes com-pletely closed by day 26. If the neural tube fails to close, major defects of the central nervous system can result.
Anencephaly (an′en-sef′̆a-l̄e; no brain) is a birth defectwherein much of the brain fails to form because the neural tube did not close in the region of the head. A baby born with anen-cephaly cannot survive. Spina bifida (spı̄ ′ n̆a bif ′ i -d̆a ; split spine) is a general term describing defects of the spinal cord or vertebral column. Spina bifida can range from a simple defect with one or more vertebral spinous processes split or missing but no clinical manifestation to a more severe defect that can result in paralysis of the limbs or the bowels and bladder, depending on where the defect occurs. More severe forms of spina bifida result from failure of the neural tube in the area of the spinal cord to close. It has been demonstrated that adequate amounts of the B vitamin folate, more commonly referred to as folic acid, in the diet during pregnancy can reduce the risk of such defects.
As the neural folds come together and fuse, a population of cells breaks away from the neuroectoderm all along the crests of the folds. Most of these neural crest cellsbecome part of the peripheral nervous system or become melanocytes in the skin. In the head, neural crest cells also contribute to the skull, the dentin of teeth, blood vessels, and general connective tissue.
Formation of the General Body structure
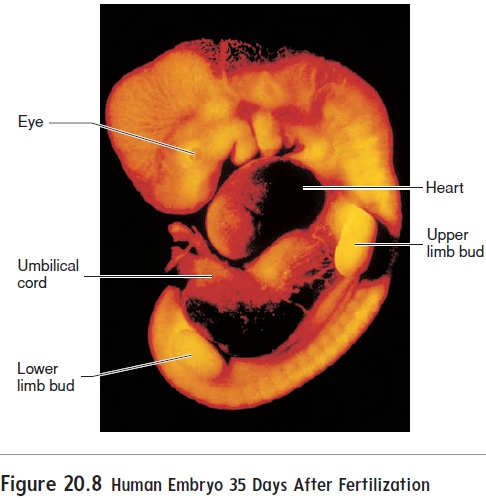
Arms and legs first appear at about 28 days after fertilization as limb buds (figure 20.8) and quickly begin to elongate. At about 35days, expansions called hand and foot plates form at the ends of the limb buds. Zones of cell death between the future fingers and toes of the hand and foot plates help sculpt the fingers and toes.
The face develops by fusion of five growing masses of tis-sue, called processes (figure 20.9). One, the frontonasal pro-cess, forms the forehead, nose, and center of the upper jaw andlip. Two maxillary processes form the maxillae (upper lip and jaw), and two mandibular processes form the mandible (lower lip and jaw).
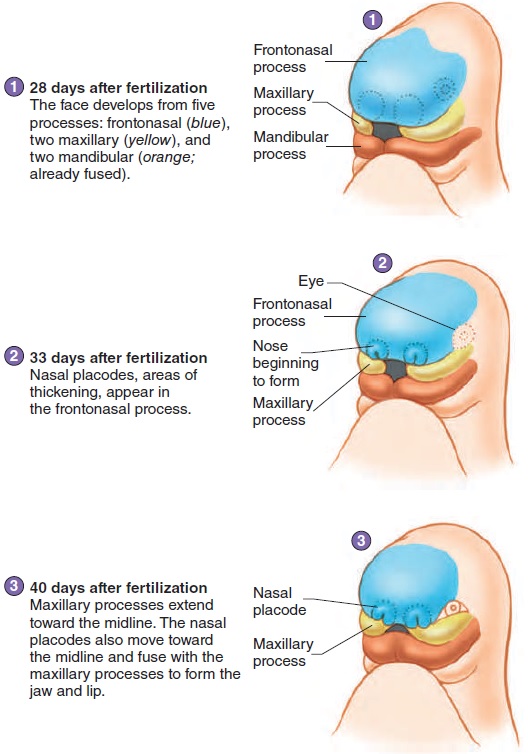
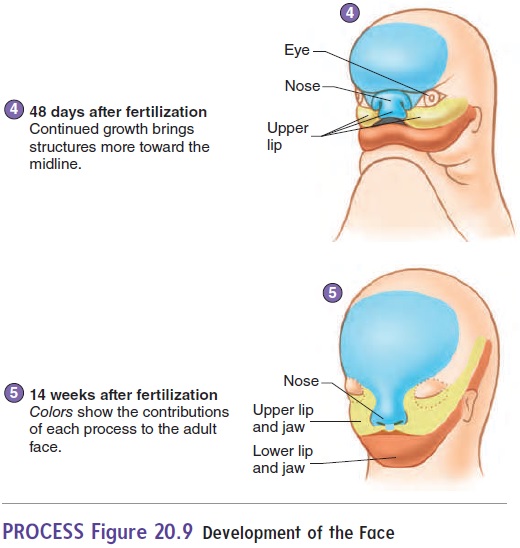
The nose begins as two structures, one on each side of the forehead mass. As the brain enlarges and the face matures, the two parts of the nose approach each other in the midline and fuse (figure 20.9). The two masses forming the upper jaw expand toward the midline and fuse with part of the nose to form the upper jaw and lip. A cleft lipresults from failure of these struc-tures to fuse. Cleft lips usually do not occur in the midline, but
The roof of the mouth, or palate, begins to form as vertical shelves of tissue that grow on the inside of the maxillary masses. These shelves swing to a horizontal position and begin to fuse with each other at about 56 days of development. If the palate does not fuse, a midline cleft in the roof of the mouth called a cleft palate results. A cleft palate can range in severity from aslight cleft of the uvula to a fissure extending the entire length of the palate. A cleft lip and cleft palate can occur together, forming a continuous fissure.
Development of the Organ Systems
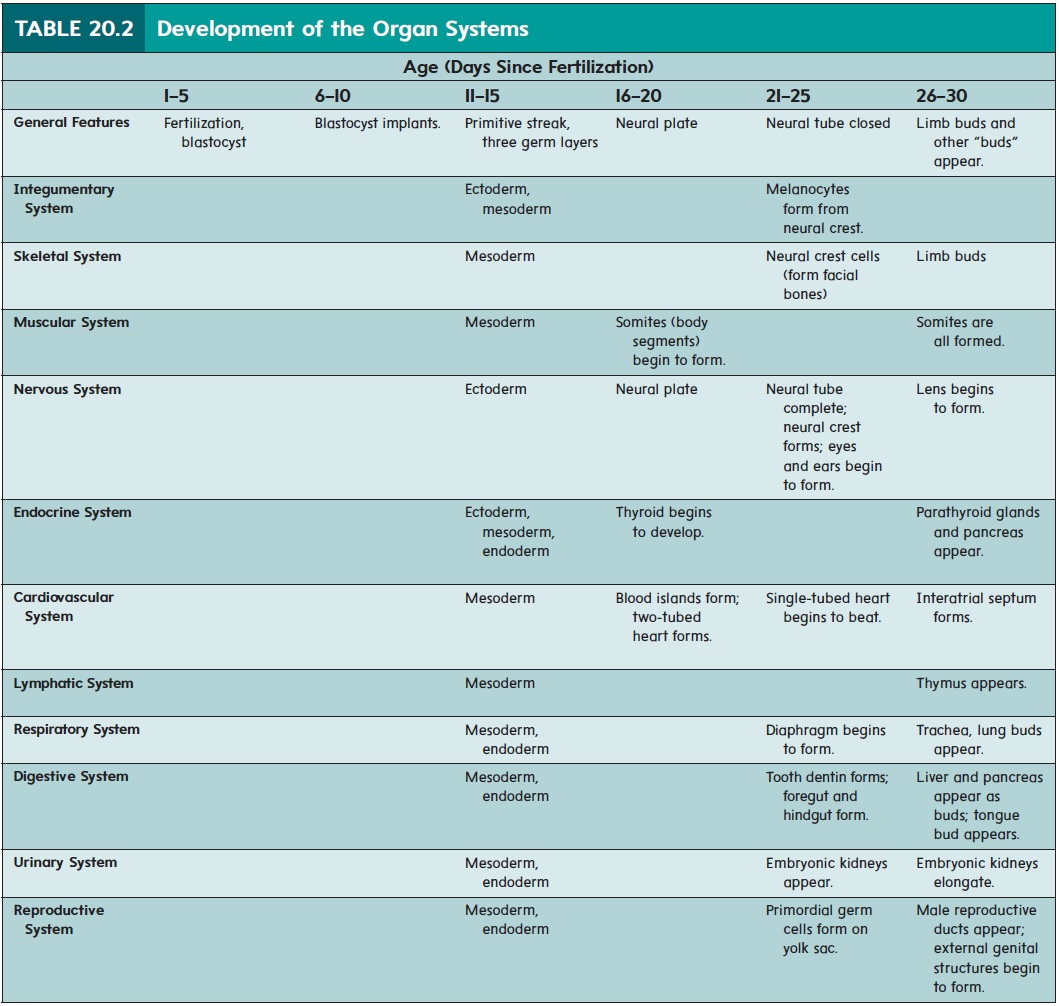
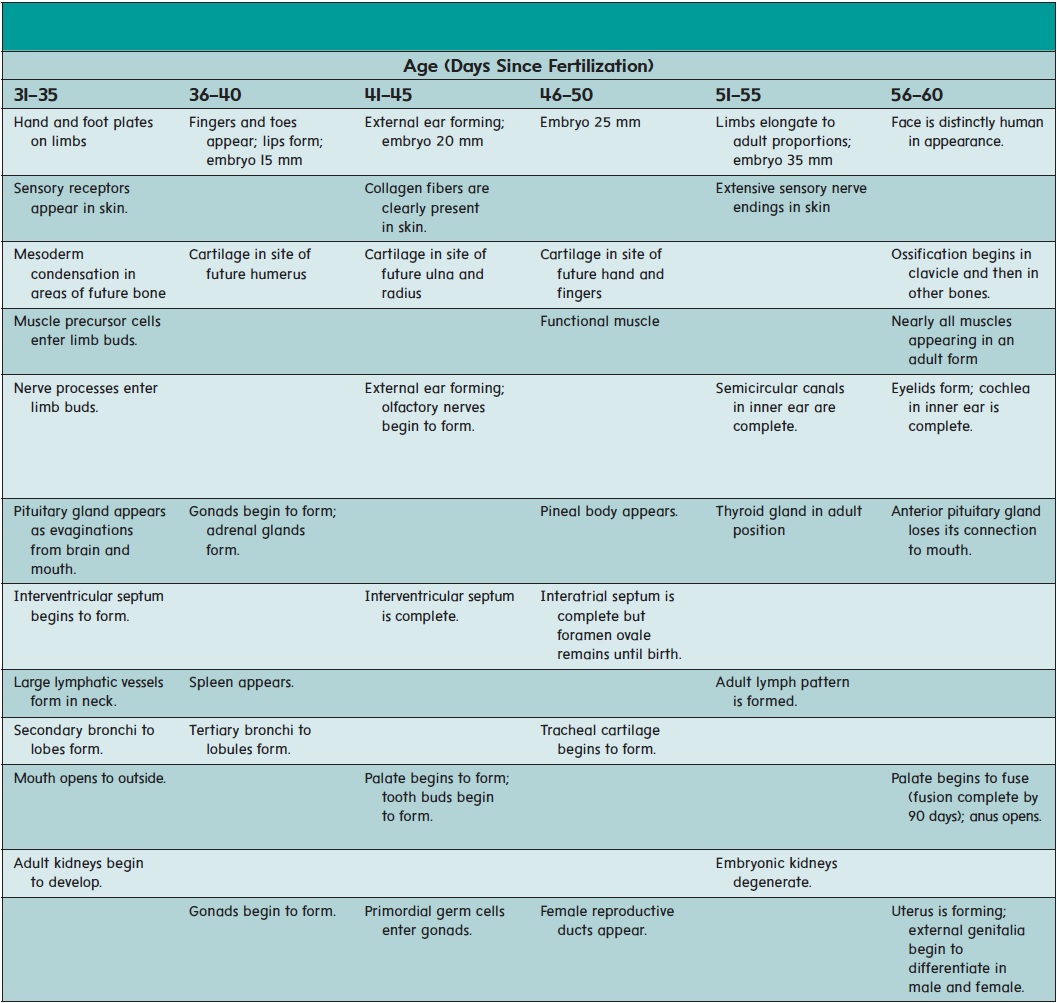
The major organ systems appear and begin to develop during the embryonic period (second to eighth week of development). This period is therefore also called the period of organogenesis (ōr′ gă-nō-jen′ ĕ-sis). The individual organ systems are listed in table 20.2; only general comments about a few select systems are presented in the text.
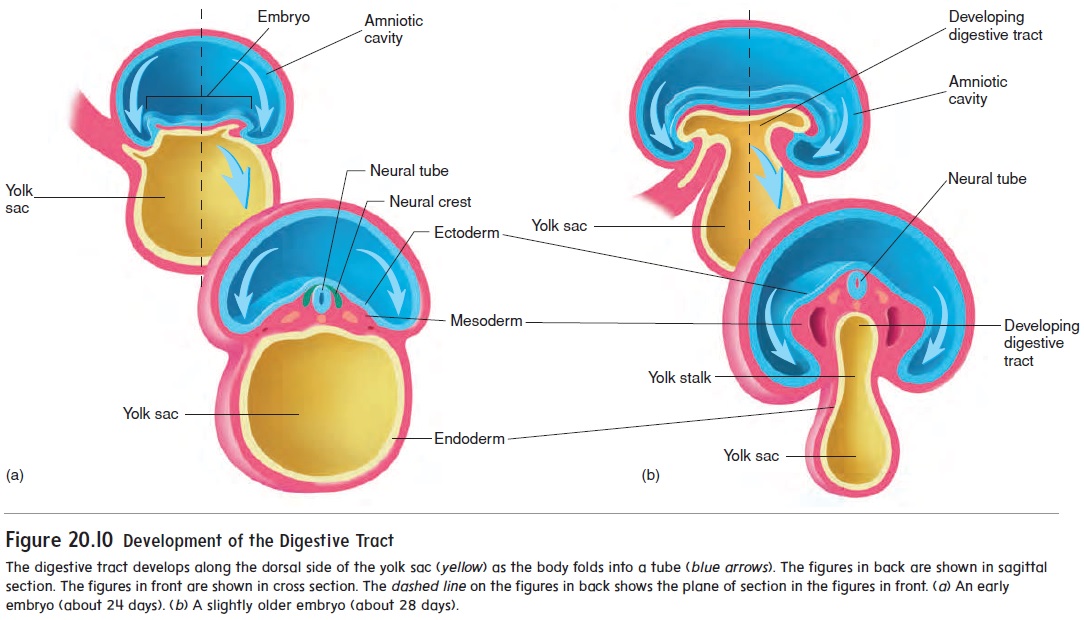
While the neural tube is forming (18–26 days), the remainder of the embryo is folding to form a tube along the upper part of the yolk sac (figure 20.10). The developing digestive tract pinches off from the yolk sac as a tube but remains attached in the center to the yolk sac by a yolk stalk.
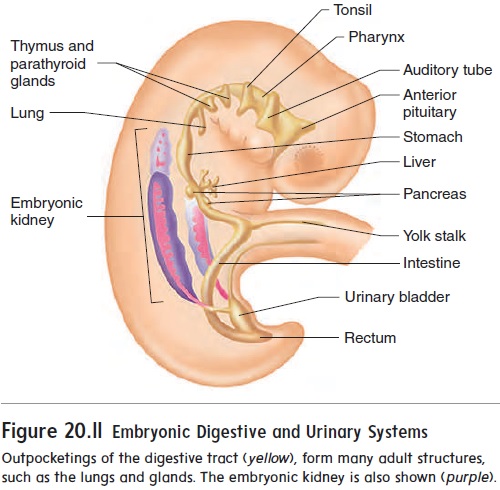
A considerable number of outpocketings appear at about 28 days after fertilization along the entire length of the digestive tract (figure 20.11). A surprisingly large number of important internal organs develop from those outpocketings, including the auditory tubes, tonsils, thymus, anterior pituitary gland, thyroid gland, parathyroid glands, lungs, liver, pancreas, and urinary bladder.
The heart develops from two blood vessels, which lie side by side in the early embryo and fuse about 21 days after fertilization into a single, midline heart (figure 20.12, steps 1 and 2). At about this time, the primitive heart begins to beat. Blood vessels form from “blood islands” on the surface of the yolk sac and inside the embryo. These islands expand and fuse to form the circulatory system.
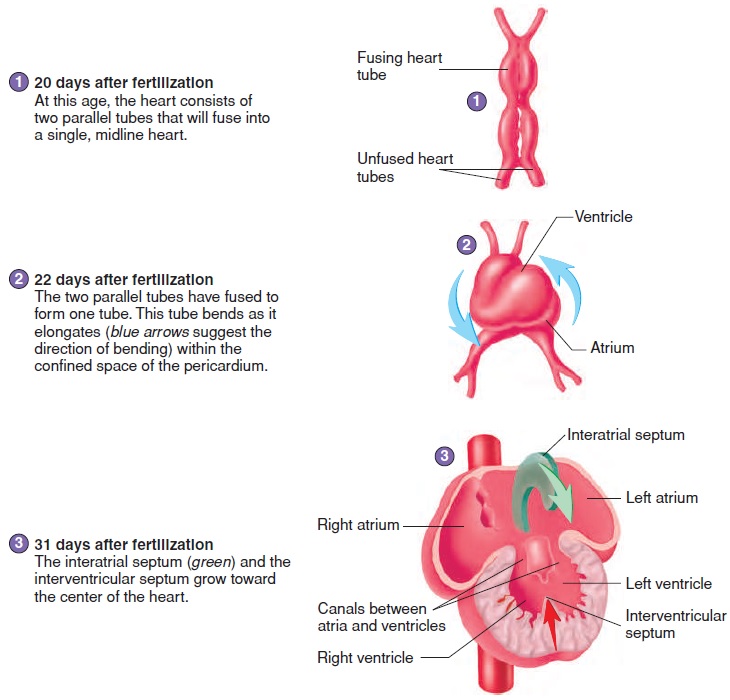
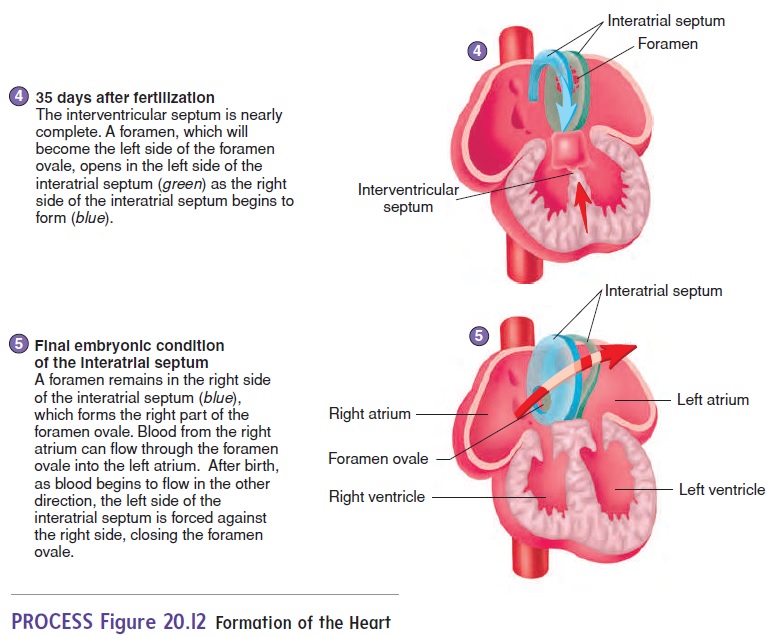
The major chambers of the heart, the atrium and ventricle, expand rapidly. The single ventricle is subdivided into two chambers by the development of an interventricular(in-ter-ven-trik′ ŭ-lăr) septum (figure 20.12, steps 3 and 4). If the interventricular sep-tum does not grow enough to completely separate the ventricles, a ventricular septal defect results.
An interatrial (in-ter-ā′ trē-ăl) septum forms to separate the two atria (figure 20.12, steps 3–5). An opening in the interatrial septum called the foramen ovale (ō-val′ ē) connects the two atria and allows blood to flow from the right to the left atrium in the fetus. Because of the foramen ovale, some blood in the fetus passes from the right atrium to the left atrium and bypasses the right ventricle and the lungs. The foramen ovale normally closes off at the time of birth, and blood then circulates through the right ventricle and the lungs. If this does not occur, an interatrial septal defect occurs. An interatrial septal defect or a ventricular septal defect usually results in a heart murmur.
The kidneys develop from mesoderm located along the lateral wall of the body cavity (see figure 20.11). The embryonic kidneys are much more extensive than the adult kidneys, extending the entire length of the body cavity. They are closely associated with internal reproductive organs, such as the ovaries or testes, and reproductive ducts, such as the uterine tubes or ductus deferens. Most of the embryonic kidneys degenerate, with only a very small part forming the adult kidney.
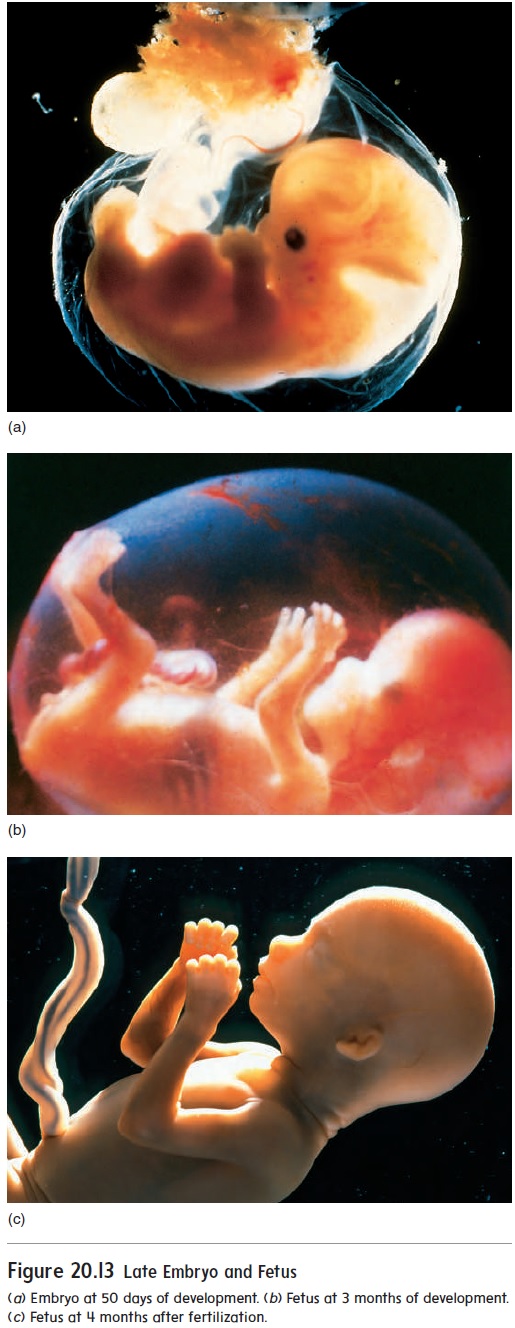
Growth of the Fetus
The embryo becomes a fetus about 8 weeks after fertilization (figure 20.13). The beginning of the fetal period is marked by the beginning of bone ossification. In the embryo, most of the organ systems are developing, whereas in the fetus the organs are present. During the fetal period, the organ systems enlarge and mature. The fetus grows from about 3 cm and 2.5 g (0.09 oz) at 8 weeks to 50 cm and 3300 g (7 lb, 4 oz) at the end of preg-nancy. The growth during the fetal period represents more than a 15-fold increase in length and a 1400-fold increase in weight. The amniotic fluid contains toxic waste products from thefetus’s digestive tract and kidneys. Fine, soft hair called lanugo (lă-noo′ gō) covers the fetus, and a waxy coat of loose epithelial cells called vernix caseosa (ver′ niks kā′ sē-ō′ să) forms a protec-tive layer between the fetus and the amniotic fluid.
Subcutaneous adipose tissue that accumulates in the fetus provides a nutrient reserve, helps insulate, and aids the newborn in sucking by strengthening and supporting the cheeks, so that a small vacuum can be developed in the oral cavity.
Peak body growth occurs late in gestation, but as the placenta reaches its maximum size, the oxygen and nutrient supply to the fetus becomes limited. Growth of the placenta essentially stops at about 35 weeks, limiting fetal growth.
At approximately 38 weeks of development, the fetus is ready to be delivered. The average weight at this point is 3250 g (7 lb, 2 oz) for a female fetus and 3300 g (7 lb, 4 oz) for a male fetus.
Related Topics