Chapter: Essentials of Anatomy and Physiology: Endocrine System
Pituitary and hypothalamus - Endocrine Glands and Their Hormones
Pituitary and hypothalamus
The pituitary (pi-too′ i-tār-rē; pituita, phlegm or thick mucous secretion) gland is also called the hypophysis (hı̄-pof′ i-sis; hypo, under + physis, growth). It is a small gland about the size of a pea (figure 10.12,top). It rests in a depression of the sphenoid bone inferior to the hypothalamus of the brain. The hypothalamus (hı̄′ pō-thal′ ă-mŭs; hypo, under + thalamos) is an important auto-nomic nervous system and endocrine control center of the brain located inferior to the thalamus. The pituitary gland lies posterior to the optic chiasm and is connected to the hypothalamus by a stalk called the infundibulum (in-fŭn-dib′ŭ-lŭm; a funnel). The pituitary gland is divided into two parts: The anterior pituitary is made up of epithelial cells derived from the embryonic oral cavity; the posterior pituitary is an extension of the brain and is composed of nerve cells. The hormones secreted from each lobe of the pituitary gland are listed in table 10.2.
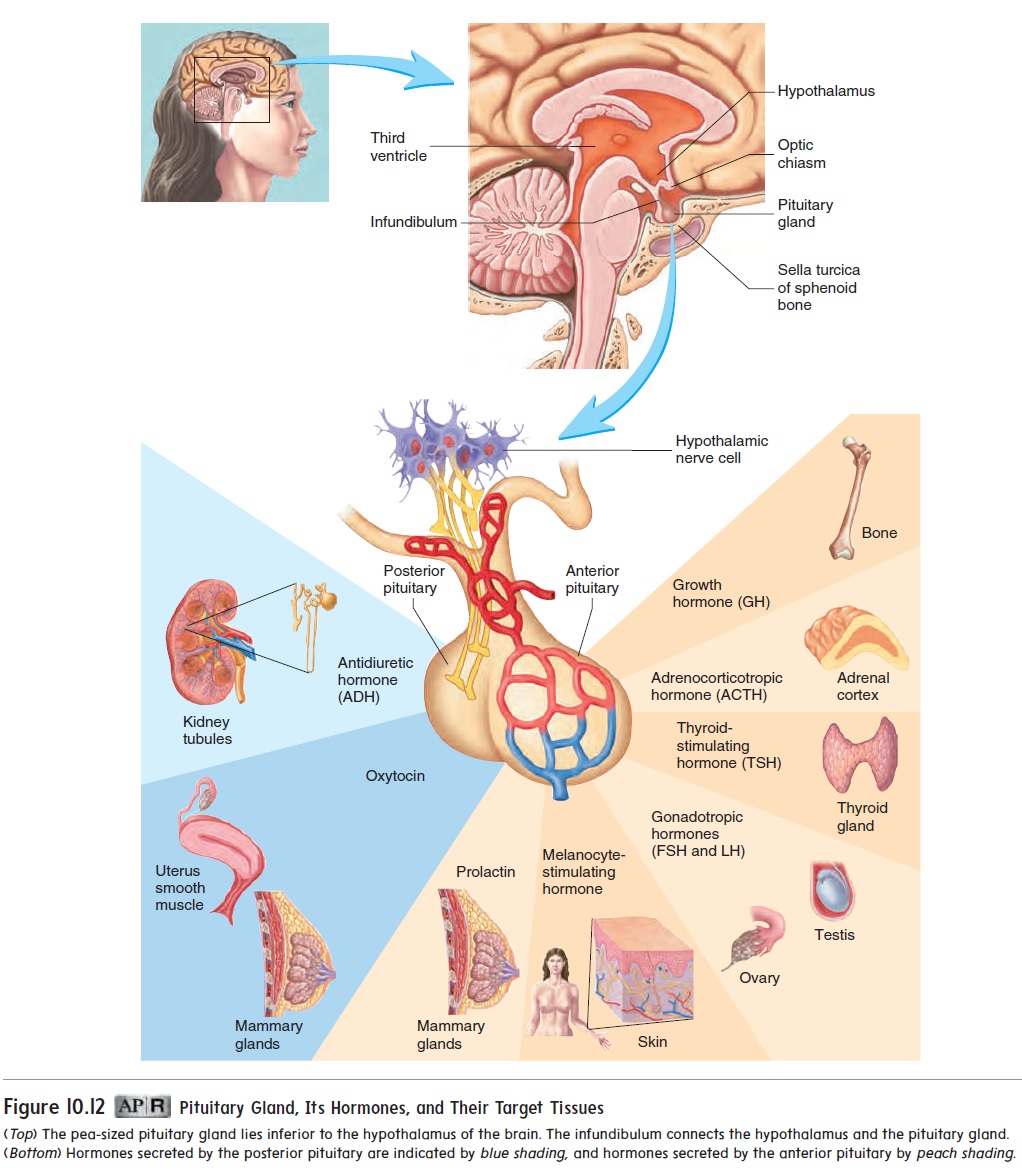
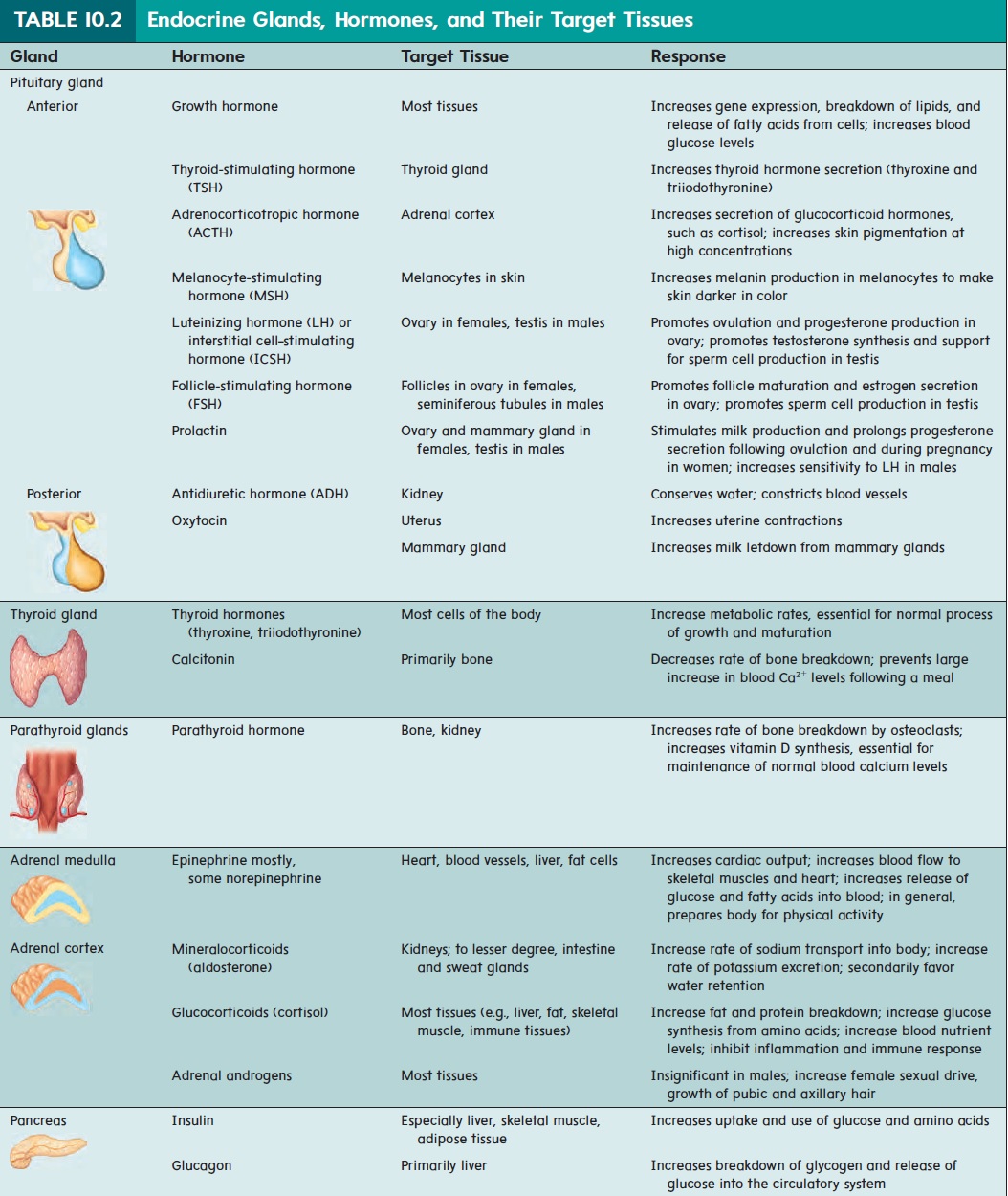
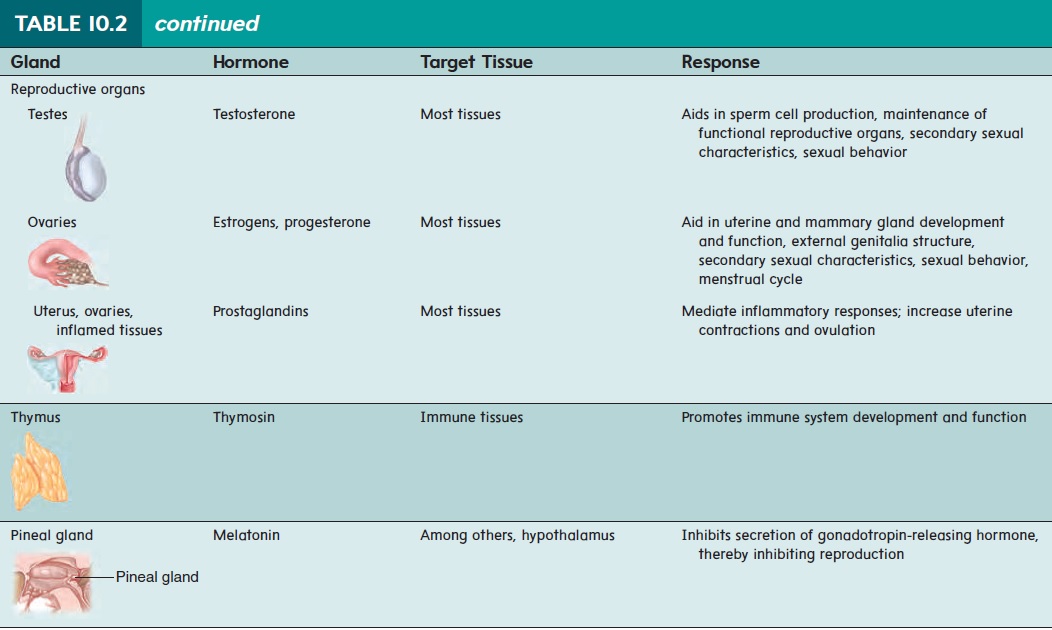
Hormones from the pituitary gland control the functions of many other glands in the body, such as the ovaries, the testes, the thyroid gland, and the adrenal cortex (figure 10.12, bottom). The pituitary gland also secretes hormones that influence growth, kid-ney function, birth, and milk production by the mammary glands. Historically, the pituitary gland was known as the body’s mastergland because it controls the function of so many other glands.However, we now know that the hypothalamus controls the pitu-itary gland in two ways: hormonal control and direct innervation.
Hormonal Control of the Anterior Pituitary
Neurons of the hypothalamus produce and secrete neuropeptides that act on cells of the anterior pituitary gland (figure 10.13). They act as either releasing hormones or inhibiting hormones. Each releasing hormone stimulates the production and secretion of a specific hormone by the anterior pituitary, whereas each inhibiting hormone decreases the secretion of a specific anterior pituitary hormone. Releasing and inhibiting hormones enter a capillary bed
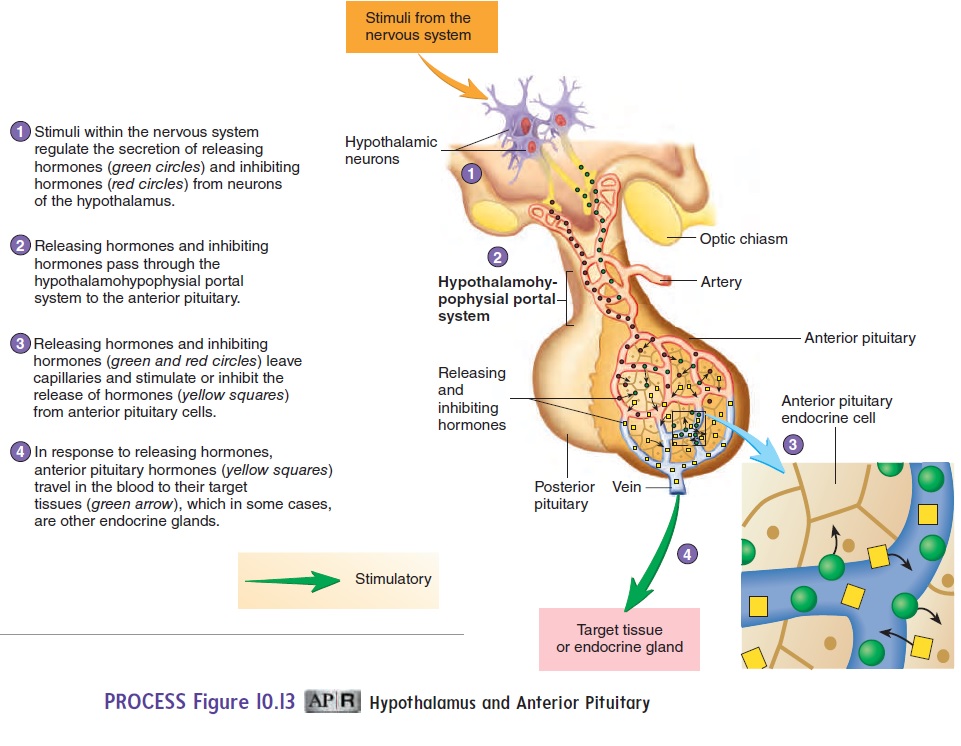
in the hypothalamus and are transported through veins to a second capillary bed in the anterior pituitary. There they leave the blood and bind to membrane-bound receptors involved with regulating anterior pituitary hormone secretion. The capillary beds and veins that transport the releasing and inhibiting hormones are called the hypothalamic-pituitary portal system.
Direct Innervation of the Posterior Pituitary
Stimulation of neurons within the hypothalamus controls the secre-tion of hormones from the posterior pituitary (figure 10.14). The cell bodies of these neurons are in the hypothalamus, and their axons extend through the infundibulum to the posterior pituitary. Hormones are produced in the nerve cell bodies and transported through the
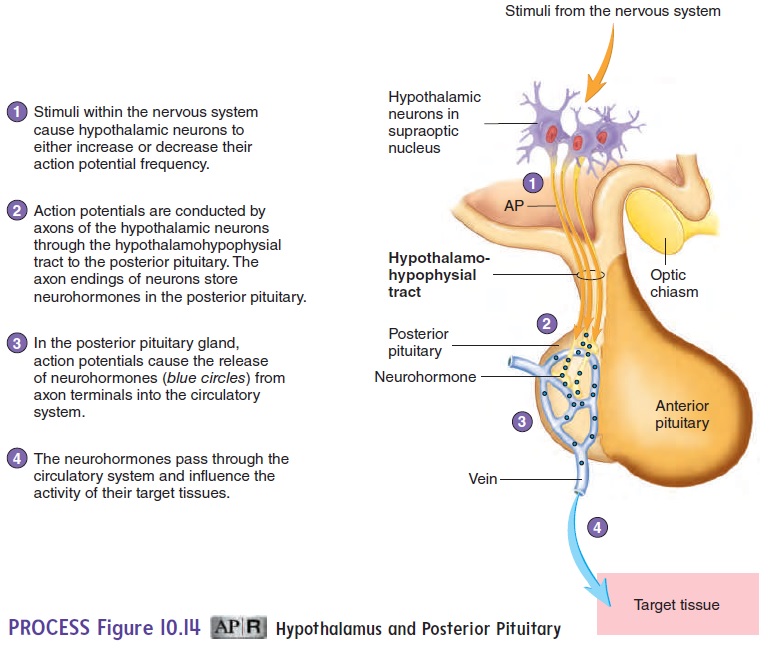
When these nerve cells are stimulated, action potentials from the hypothalamus travel along the axons to the posterior pituitary and cause the release of hormones from the axon endings.
Within the hypothalamus and pituitary, the nervous and endo-crine systems are closely interrelated. Emotions such as joy and anger, as well as chronic stress, influence the endocrine system through the hypothalamus. Conversely, hormones of the endocrine system can influence the functions of the hypothalamus and other parts of the brain.
Hormones of the Anterior Pituitary
Growth hormone (GH) stimulates the growth of bones, muscles,and other organs by increasing gene expression. It also resists protein breakdown during periods of food deprivation and favors lipid breakdown. Too little growth hormone secretion can result from abnormal development of the pituitary gland. A young per-son suffering from a deficiency of growth hormone remains small, although normally proportioned, and is called a pituitary dwarf (dw̄o rf). This condition can be treated by administering growth hor-mone. Because GH is a protein, it is difficult to produce artificially using conventional techniques. However, human genes for GH have been successfully introduced into bacteria using genetic engineering techniques. The gene in the bacteria causes GH synthesis, and the GH can be extracted from the medium in which the bacteria are grown. Thus, modern genetic engineering has provided a source of human GH for people who produce inadequate quantities.
Excess growth hormone secretion can result from hormone-secreting tumors of the pituitary gland. If excess growth hormone is present before bones finish growing in length, exaggerated bone growth occurs. The person becomes abnormally tall, a condition called giantism (j̄ı ′ an-tizm). If excess hormone is secreted after growth in bone length is complete, growth continues in bone diam-eter only. As a result, the facial features and hands become abnor-mally large, a condition called acromegaly (ak-r̄o -meg′ ̆a -l̄e ).
The secretion of growth hormone is controlled by two hor-mones from the hypothalamus. A releasing hormone stimulates growth hormone secretion, and an inhibiting hormone inhibits its secretion. Most people have a rhythm of growth hormone secre-tion, with daily peak levels occurring during deep sleep. Growth hormone secretion also increases during periods of fasting and exercise. Blood growth hormone levels do not become greatly elevated during periods of rapid growth, although children tend to have somewhat higher blood levels of growth hormone than do adults. In addition to growth hormone, genetics, nutrition, and sex hormones influence growth.
Part of the effect of growth hormone is influenced by a group of protein hormones called insulin-like growth factors (IGFs), or somatomedins (sō ′ mă -tō -mē ′ dinz). Growth hormone increases IGF secretion from tissues such as the liver, and the IGF molecules bind to receptors on the cells of tissues such as bone and cartilage, where they stimulate growth. The IGFs are similar in structure to insulin and can bind, to some degree, to insulin receptors. Also, insulin, at high concentrations, can bind to IGF receptors.
Thyroid-stimulating hormone (TSH) binds to membrane-bound receptors on cells of the thyroid gland and causes the cells to secrete thyroid hormone. When too much TSH is secreted, the thyroid gland enlarges and secretes too much thyroid hormone. When too little TSH is secreted, the thyroid gland decreases in size and secretes too little thyroid hormone. The rate of TSH secretion is increased by a releasing hormone from the hypothalamus.
Adrenocorticotropic (a-drē′nō-kōr′ti-kō-trō′pik) hormone (ACTH) binds to membrane-bound receptors on cells in the cortex ofthe adrenal glands. ACTH increases the secretion of a hormone fromthe adrenal cortex called cortisol (k̄o r′ ti-sol), also called hydrocorti-sone. ACTH is required to keep the adrenal cortex from degenerating. ACTH molecules also bind to melanocytes in the skin and increase skin pigmentation . One symptom of too much ACTH secretion is darkening of the skin. The rate of ACTH secre-tion is increased by a releasing hormone from the hypothalamus.
Gonadotropins (gō′nad-ō-trō′pinz) are hormones that bindto membrane-bound receptors on the cells of the gonads (ovaries and testes). They regulate the growth, development, and functions of the gonads. In females, luteinizing (loo′ t̄e -̆ı -n̄ı z-ing) hormone(LH) causes the ovulation of oocytes and the secretion of the sexhormones estrogen and progesterone from the ovaries. In males, LH stimulates interstitial cells of the testes to secrete the sex hormone testosterone and thus is sometimes referred to as interstitial (in-ter-stish′ăl) cell–stimulating hormone (ICSH). Follicle-stimulatinghormone (FSH) stimulates the development of follicles in the ova-ries and sperm cells in the testes. Without LH and FSH, the ovaries and testes decrease in size, no longer produce oocytes or sperm cells, and no longer secrete hormones. A single releasing hormone from the hypothalamus increases the secretion of both LH and FSH.
Prolactin (prō-lak′tin;pro,precursor+lact,milk) binds tomembrane-bound receptors in cells of the breast, where it helps pro-mote development of the breast during pregnancy and stimulates the production of milk following pregnancy. The regulation of prolactin secretion is complex and may involve several substances released from the hypothalamus. There are two main regulatory hormones; one increases prolactin secretion and one decreases it.
Melanocyte-stimulating (mel′ă-nō-sı̄t) hormone (MSH) binds to membrane-bound receptors on melanocytes and causes them to synthesize melanin. The structure of MSH is similar to that of ACTH, and oversecretion of either hormone causes the skin to darken. Regulation of MSH is not well understood, but there appear to be two regulatory hormones from the hypothalamus— one that increases MSH secretion and one that decreases it.
Hormones of the Posterior Pituitary
Antidiuretic (an′tē-dı̄-ŭ-ret′ik;anti,against+uresis,urine vol-ume) hormone (ADH) binds to membrane-bound receptors and increases water reabsorption by kidney tubules. This results in less water lost as urine. ADH can also cause blood vessels to constrict when released in large amounts. Consequently, it is sometimes also called vasopressin (vā-sō-pres′in). Reduced ADH release from the posterior pituitary results in large amounts of dilute urine.
A lack of ADH secretion causes diabetes insipidus, which is the production of a large amount of dilute urine. The consequences of diabetes insipidus are not obvious until the condition becomes severe, producing many liters of urine each day. The large urine volume causes an increase in the concentration of the body fluids and the loss of important electrolytes, such as Ca2+, Na+, and K+. The lack of ADH secretion may be familiar to some who have ever had alcohol to drink. The diuretic actions of alcohol are due to its inhibition of ADH secretion.
Oxytocin (ok′sı̆-tō′sin; swift birth) binds to membrane-boundreceptors, and causes contraction of the smooth muscle cells of the uterus as well as milk letdown from the breasts in lactating women. Commercial preparations of oxytocin, known as Pitocin, are given under certain conditions to assist in childbirth and to constrict uterine blood vessels following childbirth.
Related Topics