Chapter: Basic & Clinical Pharmacology : Skeletal Muscle Relaxants
Neuromuscular Blocking Drugs
NEUROMUSCULAR BLOCKING DRUGS
History
During
the 16th century, European explorers found that natives in the Amazon Basin of
South America were using curare, an arrow poison that produced skeletal muscle
paralysis, to kill animals. The active compound, d-tubocurarine, and its modern synthetic derivatives have had a
major influence on the practice of anesthe-sia and surgery and have proved
useful in understanding the basic mechanisms involved in neuromuscular
transmission.
Normal Neuromuscular Function
The mechanism of neuromuscular transmission at the motor end
plate is similar to that described for preganglionic cholinergic nerves. The
arrival of an action potential at the motor nerve terminal causes an influx of
calcium and release of the neurotrans-mitter acetylcholine. Acetylcholine then
diffuses across the synaptic cleft to activate the nicotinic receptors located
on the motor end plate. As noted, the adult NM receptor is composed
of five peptides: two alpha peptides, one beta, one gamma, and one delta
peptide (Figure 27–1). The binding of two acetylcholine
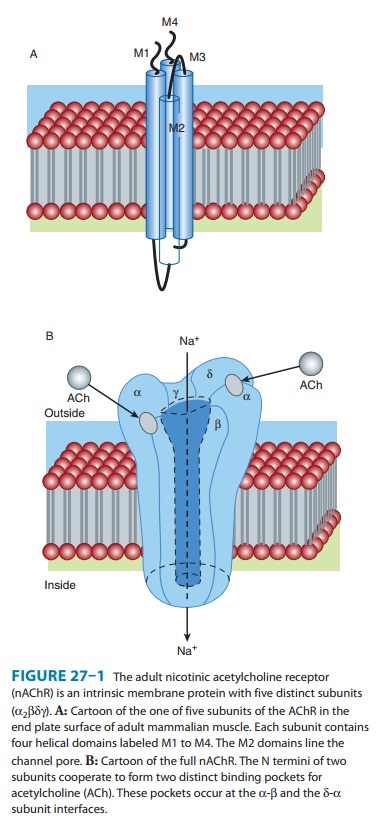
molecules to receptors on the α-β and δ-α subunits causes opening of the channel. The
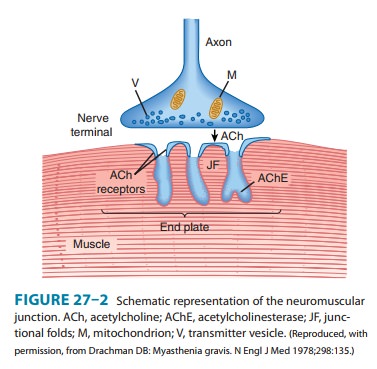
subsequent movement of sodium and potas-sium through the channel is associated
with a graded depolarization of the end plate membrane (Figure 27–2). This
change in voltage is termed the motor end plate potential. The magnitude of the
end plate potential is directly related to the amount of acetylcholine
released. If the potential is small, the permeability and the end plate
potential return to normal without an impulse being propagated from the end
plate region to the rest of the muscle membrane.
However,
if the end plate potential is large, the adjacent muscle membrane is
depolarized, and an action potential will be propa-gated along the entire
muscle fiber. Muscle contraction is then initi-ated by excitation-contraction
coupling. The released acetylcholine is quickly removed from the end plate
region by both diffusion and enzymatic destruction by the local
acetylcholinesterase enzyme.
At
least two additional types of acetylcholine receptors are found within the
neuromuscular apparatus. One type is located on the presynaptic motor axon
terminal, and activation of these receptors mobilizes additional transmitter
for subsequent release by moving more acetylcholine vesicles toward the
synaptic mem-brane. The second type of receptor is found on perijunctional
cells and is not normally involved in neuromuscular transmission. However,
under certain conditions (eg, prolonged immobiliza-tion, thermal burns), these
receptors may proliferate sufficiently to affect subsequent neuromuscular
transmission.
Skeletal
muscle relaxation and paralysis can occur from inter-ruption of function at
several sites along the pathway from the central nervous system (CNS) to
myelinated somatic nerves, unmyelinated motor nerve terminals, nicotinic
acetylcholine receptors, the motor end plate, the muscle membrane, and the
intracellular muscular contractile apparatus itself.
Blockade of end plate function can be accomplished by two basic
mechanisms. Pharmacologic blockade of the physiologic agonist acetylcholine is
characteristic of the antagonist neuromus-cular blocking drugs (ie,
nondepolarizing neuromuscular blocking drugs). These drugs prevent access of
the transmitter to its recep-tor and thereby prevent depolarization. The
prototype of this nondepolarizing subgroup is d-tubocurarine. The
second type of blockade can be produced by an excess of a depolarizing agonist,
such as acetylcholine. This seemingly paradoxical effect of acetyl-choline also
occurs at the ganglionic nicotinic acetylcholine recep-tor. The prototypical
depolarizing blocking drug is succinylcholine.
A similar depolarizing block can be produced by acetylcholine itself when high
local concentrations are achieved in the synaptic cleft (eg, by cholinesterase
inhibitor intoxication) and by nicotine and other nicotinic agonists. However,
the neuromuscular block produced by depolarizing drugs other than
succinylcholine cannot be precisely controlled and is of no clinical value.
Related Topics