Chapter: Basic & Clinical Pharmacology : Adrenoceptor Agonists & Sympathomimetic Drugs
Mixed-Acting Sympathomimetics
Mixed-Acting Sympathomimetics
Ephedrine occurs in various plants and has been used in
Chinafor over 2000 years; it was introduced into Western medicine in 1924 as
the first orally active sympathomimetic drug. It is found in ma huang, a
popular herbal medication . Ma huang contains multiple ephedrine-like alkaloids
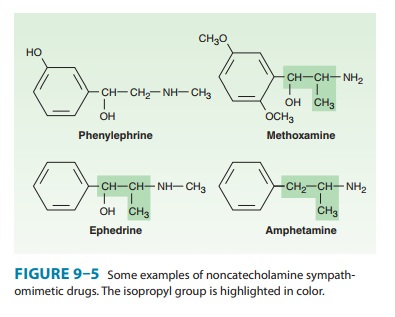
in addition to ephedrine. Because ephedrine is a noncatechol phenylisopro-pylamine
(Figure 9–5), it has high bioavailability and a relatively long duration of
action—hours rather than minutes. As with many other phenylisopropylamines, a
significant fraction of the drug is excreted unchanged in the urine. Since it
is a weak base, its excretion can be accelerated by acidification of the urine.
Ephedrine
has not been extensively studied in humans despite its long history of use. Its
ability to activate β
receptors probably accounted for its earlier use in asthma. Because it gains
access to the central nervous system, it is a mild stimulant. Ingestion of
ephedrine alkaloids contained in ma huang has raised important safety concerns.
Pseudoephedrine, one of four
ephedrine enantiomers, has been available over the counter as a component of
many decongestant mixtures. However, the use of pseu-doephedrine as a precursor
in the illicit manufacture of metham-phetamine has led to restrictions on its
sale.
Phenylpropanolamine was a common component
in over-the-counter appetite suppressants. It was removed from the mar-ket
because its use was associated with hemorrhagic strokes in young women. The
mechanism of this potential adverse effect is unknown, but the drug can
increase blood pressure in patients with impaired autonomic reflexes.
Related Topics