Chapter: Basic & Clinical Pharmacology : Drug Biotransformation
Microsomal Mixed Function Oxidase System & Phase I Reactions
MICROSOMAL MIXED FUNCTION OXIDASE
SYSTEM & PHASE I REACTIONS
Many drug-metabolizing enzymes are located in the lipophilic endoplasmic reticulum membranes of the liver and other tissues
When
these lamellar membranes are isolated by homogenization and fractionation of
the cell, they re-form into vesicles called microsomes. Microsomes retain most of the morphologic andfunctional
characteristics of the intact membranes, including the rough and smooth surface
features of the rough (ribosome-studded) and smooth (no ribosomes) endoplasmic
reticulum. Whereas the rough microsomes tend to be dedicated to protein
synthesis, the smooth microsomes are relatively rich in enzymes responsible for
oxidative drug metabolism. In particular, they contain the important class of
enzymes known as the mixed func-tion
oxidases (MFOs), or monooxygenases. The
activity of theseenzymes requires both a reducing agent (nicotinamide adenine
dinucleotide phosphate [NADPH]) and molecular oxygen; in a typical reaction,
one molecule of oxygen is consumed (reduced) per substrate molecule, with one
oxygen atom appearing in the product and the other in the form of water.
In
this oxidation-reduction process, two microsomal enzymes play a key role. The
first of these is a flavoprotein, NADPH-cytochrome
P450 oxidoreductase (POR).One
mole of thisenzyme contains 1 mol each of flavin mononucleotide (FMN) and
flavin adenine dinucleotide (FAD). The second microsomal enzyme is a
hemoprotein called cytochrome P450,
which serves as the terminal oxidase. In fact, the microsomal membrane har-bors
multiple forms of this hemoprotein, and this multiplicity is increased by
repeated administration of or exposure to exogenous chemicals (see text that
follows). The name cytochrome P450 (abbreviated as P450 or CYP) is derived
from the spectral proper-ties of this hemoprotein. In its reduced (ferrous)
form, it binds carbon monoxide to give a complex that absorbs light maximally
at 450 nm. The relative abundance of P450s, compared with that of the reductase
in the liver, contributes to making P450 heme reduction a rate-limiting step in
hepatic drug oxidations.
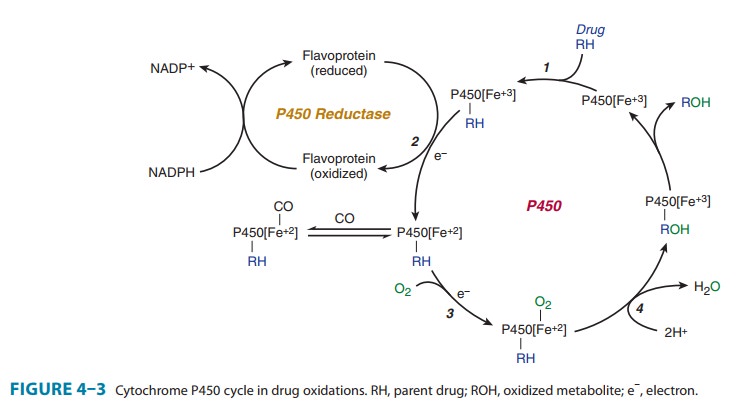
Microsomal
drug oxidations require P450, P450 reductase, NADPH, and molecular oxygen. A
simplified scheme of the oxida-tive cycle is presented in Figure 4–3. Briefly,
oxidized (Fe3+) P450 combines with a drug substrate to form a binary complex
(step 1). NADPH donates an electron to the flavoprotein P450 reductase, which
in turn reduces the oxidized P450-drug complex (step 2). A second electron is
introduced from NADPH via the same P450 reductase, which serves to reduce
molecular oxygen and to form an “activated oxygen”-P450-substrate complex (step
3). This complex in turn transfers activated oxygen to the drug substrate to
form the oxidized product (step 4).The potent oxidizing properties of this
activated oxygen permit oxidation of a large number of substrates. Substrate
specificity is very low for this enzyme complex. High lipid solubility is the
only common structural feature of the wide variety of structurally unre-lated
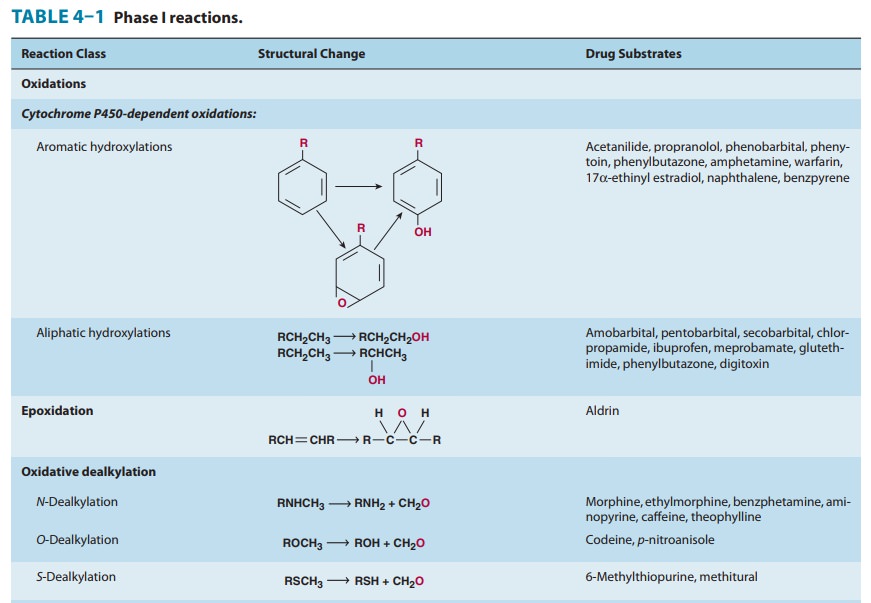
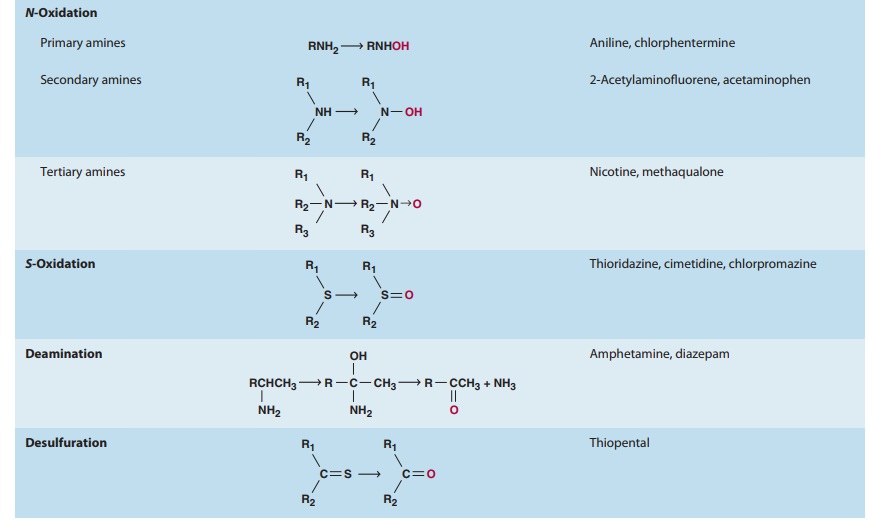
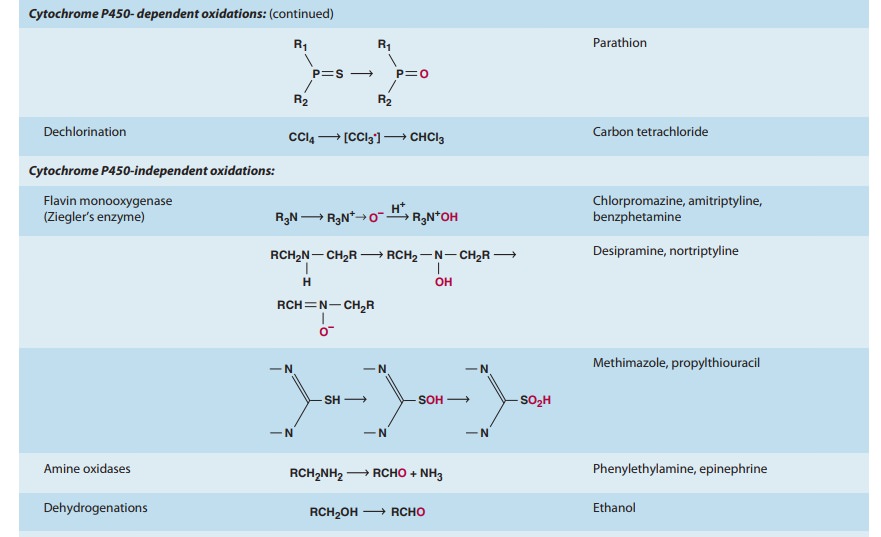
drugs and chemicals that serve as substrates in this system (Table 4–1).
However, compared with many other enzymes including phase II enzymes, P450s are
remarkably sluggish cata-lysts, and their drug biotransformation reactions are
slow.
Related Topics