Chapter: Civil : Construction Materials: Lime, Cement, Aggregates, Mortar
Manufacture Of Cement
Manufacture Of Cement
Calcareous and argillaceous raw
materials are used in the manufacture of Portland cement. The calcareous
materials used are cement rock, limestone, marl, chalk and marine shell. The
argillaceous materials consist of silicates of alumina in the form of clay, shale,
slate and blast furnace slag.
From the
above materials, others like lime, silica, alumina, iron oxide and small
quantities of other chemicals are obtained. Cement can be manufactured either
by dry process or wet process.
Dry
Process
The dry
process is adopted when the raw materials are quite hard. The process is slow
and the product is costly. Limestone and clay are ground to fine powder
separately and are mixed. Water is added to make a thick paste. The cakes of
this paste, which contain about 14 per cent of moisture, are dried and are
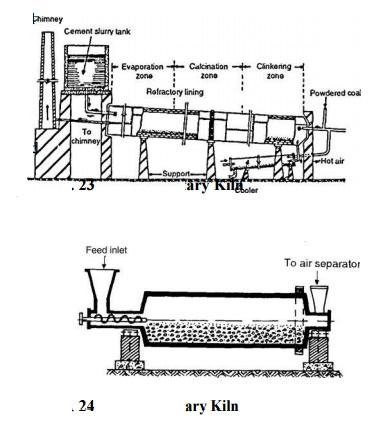
charged into rotary kiln (Fig. 23). The product obtained after calcination in
rotary kiln is called clinker. The clinker is obtained as a result of incipient
fusion and sintering at a temperature of about 1400 o - 1500 o C. Because ferric
oxide has lower melting point than the other oxides, it acts as a flux. Aeration
of cement clinker, which is commonly practised to slake free lime, also causes
an absorption of some moisture and carbon
dioxide. Absorption of moisture tends to decrease the setting
whereas that of carbon dioxide accelerates setting. The clinker is cooled
rapidly to preserve the metastable compounds and their solid solutions -
dispersion of one solid in another - which
are made as the clinker is heated. Clinker is then cooled and ground in tube
mills (Fig. 24),
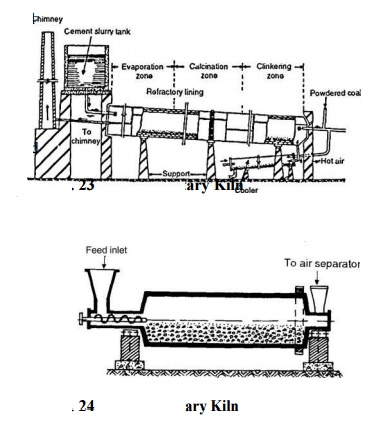
where
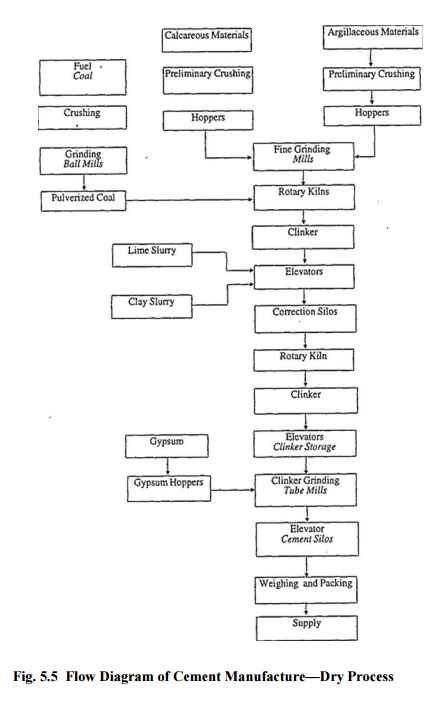
2-3% of gypsum is added. Generally, cement is stored in bags of 50 kg. A flow
diagram of dry process is shown in Fig. 25. The purpose of adding gypsum is to
coat the cement particles by interfering with the process of hydration of the
cement particles. This retards the setting of cement.
Wet
Process
operations in the wet process of cement manufacture
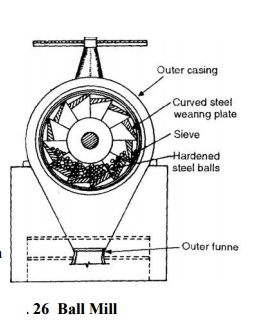
mixing, burning and grinding. The crushed raw erials are fed into ball mill
(Fig. 26) and a little water dded. On operating the ball mill, the steel balls
in it verize the raw materials which form a slurry with er. This slurry is
passed to silos (storage tanks), where proportioning of the compounds is
adjusted to ensure red chemical composition. The corrected slurry ing about 40
per cent moisture content, is then fed rotary kiln (Fig. 24) where it loses
moisture and ms into lumps or nodules. These are finally burned at 0-1600 o C.
The nodules change to clinker at this
perature. Clinker is cooled and then ground in tube s. While grinding the clinker,
about 3 per cent gypsum dded. The cement is then stored in silos from where
supplied. A flow diagram of manufacturing cement wet process is shown in Fig.
27.
Comparison
of Wet and Dry Process: The chief advantages of the wet process are
the low cost of excavating and grinding raw materials, the accurate
control of composition and homogeneity of the slurry, and the economical
utilization of fuel through the elimination of separated drying operations. On
the other hand the longer kilns, essential in the wet process, cost more and
are less responsive to a variable clinker demand than the short kilns which can
be used in the dry process.
Related Topics