Chapter: Mechanical : Heat and Mass Transfer : Conduction
Lumped Heat Analysis
LUMPED HEAT ANALYSIS:
Lumped
Capacity System-Necessary Physical Assumptions
We know that a
temperature gradient must exist in a material if heat energy is to be conducted
into or out of the body. When Bi < 0.1, it is assumed that the internal
thermal resistance of the body is very small in comparison with the external
resistance and the transfer of heat energy is primarily controlled by the
convective heat transfer at the surface. That is, the temperature within the
body is approximately uniform. This idealised assumption is possible, if
(a) the
physical size of the body is very small,
(b) the
thermal conductivity of the material is very large, and
(c) the
convective heat transfer coefficient at the surface is very small and there is
a large
temperature difference across the fluid layer at the
interface.
An Expression for Evaluating the
Temperature Variation in a Solid Using Lumped
Capacity Analysis
Let us consider a small
metallic object which has been suddenly immersed in a fluid during a heat
treatment operation. By applying the first law of
Heat
flowing out of the body = Decrease in the internal thermal energy of
during
a time dt the body during that time dt
or,
hAs( T -T¥ )dt = - pCVdT
where As is
the surface area of the body, V is the volume of the body and C is the specific
heat capacity.
or,
(hA/ rCV)dt = - dT /( T -T¥)
with
the initial condition being: at t = 0, T = Ts
The
solution is : ( T -T¥)/( Ts -T¥)
= exp(-hA / rCV)t
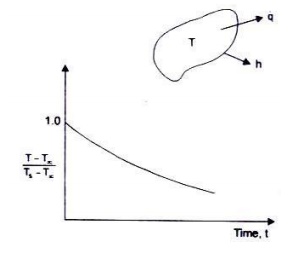
Fig. depicts the cooling of a body (temperature
distribution time) using lumped thermal capacity system. The temperature
history is seen to be an exponential decay.
We
can express
Bi
× Fo = (hL/k)×( at/L2)
= (hL/k)(k/ rC)(t/L2) =
(hA/ rCV)t,
where
V / A is the characteristic length L.
And,
the solution describing the temperature variation of the object with respect to
time is given
(
T -T¥)/( Ts -T¥)
= exp(-Bi· Fo)
Related Topics