Chapter: Medical Physiology: Renal Regulation of Potassium, Calcium, Phosphate, and Magnesium; Integration of Renal Mechanisms for Control of Blood Volume and Extracellular Fluid Volume
Importance of Pressure Natriuresis and Pressure Diuresis in Maintaining Body Sodium and Fluid Balance
Importance of Pressure Natriuresis and Pressure Diuresis in Maintaining Body Sodium and Fluid Balance
One of the most basic and powerful mechanisms for control of blood volume and extracellular fluid volume, as well as for the maintenance of sodium and fluid balance, is the effect of blood pressure on sodium and water excretion—called the pressure natriuresis and pressure diuresis mechanisms, respectively. As discussed in, this feedback between the kidneys and the circulatory system also plays a domi-nant role in long-term blood pressure regulation.
Pressure diuresis refers to the effect of increased blood pressure to raise urinary volume excretion, whereas pressure natriuresis refers to the rise in sodium excretion that occurs with elevated blood pres-sure. Because pressure diuresis and natriuresis usually occur in parallel, we refer to these mechanisms simply as “pressure natriuresis” in the following discussion.
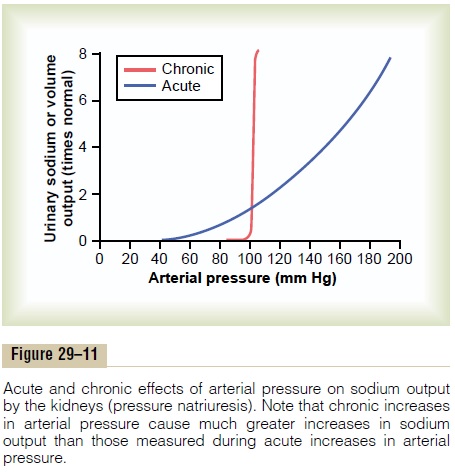
Figure 29–11 shows the effect of arterial pressure on urinary sodium output. Note that acute increases in blood pressure of 30 to 50 mm Hg cause a twofold to threefold increase in urinary sodium output. This effect is independent of changes in activity of the sym-pathetic nervous system or of various hormones, such as angiotensin II, ADH, or aldosterone, because pres-sure natriuresis can be demonstrated in an isolated kidney that has been removed from the influence of these factors. With chronic increases in blood pressure, the effectiveness of pressure natriuresis is greatly enhanced because the increased blood pressure also, after a short time delay, suppresses renin release and, therefore, decreases formation of angiotensin II and aldosterone. As discussed previously, decreased levels of angiotensin II and aldosterone inhibit renal tubular reabsorption of sodium, thereby amplifying the direct effects of increased blood pressure to raise sodium and water excretion.
Pressure Natriuresis and Diuresis Are Key Components of a Renal-Body Fluid Feedback for Regulating Body Fluid Volumes and Arterial Pressure
The effect of increased blood pressure to raise urine output is part of a powerful feedback system that operates to maintain balance between fluid intake and output, as shown in Figure 29–12. The extracellular fluid volume, blood volume, cardiac output, arterial pressure, and urine output are all controlled at the same time as separate parts of this basic feedback mechanism.
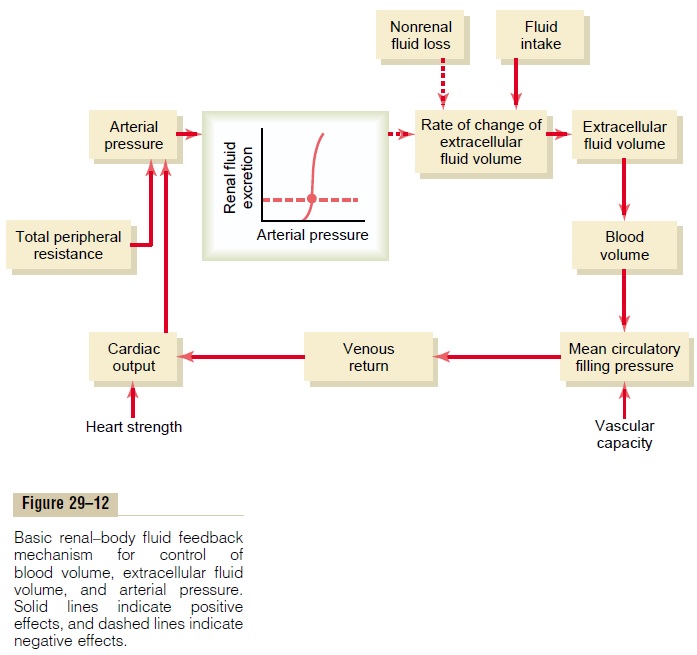
During changes in sodium and fluid intake, this feed-back mechanism helps to maintain fluid balance and to minimize changes in blood volume, extracellular fluid volume, and arterial pressure as follows:
1. An increase in fluid intake (assuming that sodium accompanies the fluid intake) above the level of urine output causes a temporary accumulation of fluid in the body.
2. As long as fluid intake exceeds urine output, fluid accumulates in the blood and interstitial spaces, causing parallel increases in blood volume and extracellular fluid volume. As discussed later, the actual increases in these variables are usually small because of the effectiveness of this feedback.
3. An increase in blood volume raises mean circulatory filling pressure.
4. An increase in mean circulatory filling pressure raises the pressure gradient for venous return.
5. An increased pressure gradient for venous return elevates cardiac output.
6. An increased cardiac output raises arterial pressure.
7. An increased arterial pressure increases urine output by way of pressure diuresis. The steepness of the normal pressure natriuresis relation indicates that only a slight increase in blood pressure is required to raise urinary excretion severalfold.
8. The increased fluid excretion balances the increased intake, and further accumulation of fluid is prevented.
7 Thus, the renal-body fluid feedback mechanism operates to prevent continuous accumulation of salt and water in the body during increased salt and water intake. As long as kidney function is normal and the pressure diuresis mechanism is operating effectively, large changes in salt and water intake can be accom-modated with only slight changes in blood volume, extracellular fluid volume, cardiac output, and arterial pressure.
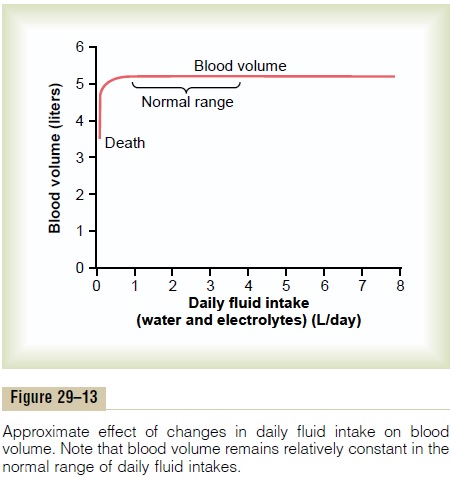
The opposite sequence of events occurs when fluid intake falls below normal. In this case, there is a ten-dency toward decreased blood volume and extracellu-lar fluid volume, as well as reduced arterial pressure. Even a small decrease in blood pressure causes a large decrease in urine output, thereby allowing fluid balance to be maintained with minimal changes in blood pressure, blood volume, or extracellular fluid volume. The effectiveness of this mechanism in pre-venting large changes in blood volume is demon-strated in Figure 29–13, which shows that changes in blood volume are almost imperceptible despite large variations in daily intake of water and electrolytes, except when intake becomes so low that it is not suf-ficient to make up for fluid losses caused by evapora-tion or other inescapable losses.
Precision of Blood Volume and Extracellular Fluid Volume Regulation
By studying Figure 29–12, one can see why the blood volume remains almost exactly constant despite extreme changes in daily fluid intake. The reason for this is the following: (1) a slight change in blood volume causes a marked change in cardiac output, (2) a slight change in cardiac output causes a large change in blood pressure, and (3) a slight change in blood pressure causes a large change in urine output. These factors work together to provide effective feedback control of blood volume.
The same control mechanisms operate whenever there is a loss of whole blood because of hemorrhage. In this case, fluid is retained by the kidneys, and other parallel processes occur to reconstitute the red blood cells and plasma proteins in the blood. If abnormali- ties of red blood cell volume remain, such as occurs when there is deficiency of erythropoietin or other factors needed to stimulate red blood cell production, the plasma volume will simply make up the difference, and the overall blood volume will return essentially to normal despite the low red blood cell mass.
Related Topics