Chapter: Ophthalmology: Glaucoma
Glaucoma: Examination Methods
Examination Methods
Oblique Illumination of the Anterior Chamber
The anterior chamber is illuminated by a beam
of light tangential to the plane of the iris. In eyes with an anterior chamber
of normal depth, the iris is
uni-formly illuminated. This is a sign of a deep anterior chamber with an open
angle (see Fig. 1.12).
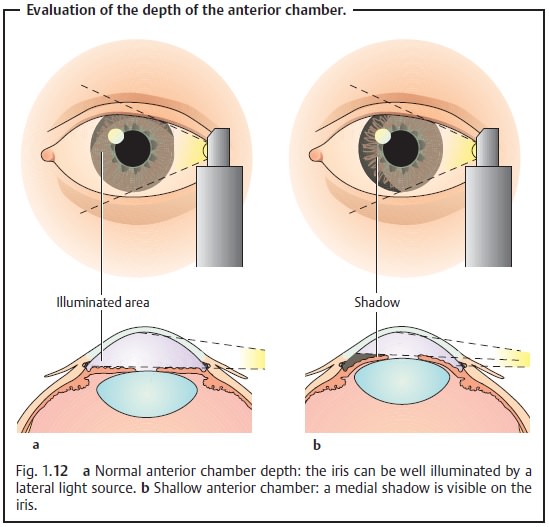
In eyes with a shallow anterior chamber and an angle that is partially or
completely closed, the iris protrudes anteriorly and is not uniformly
illumi-nated (see Fig. 1.12).
Slit-Lamp Examination
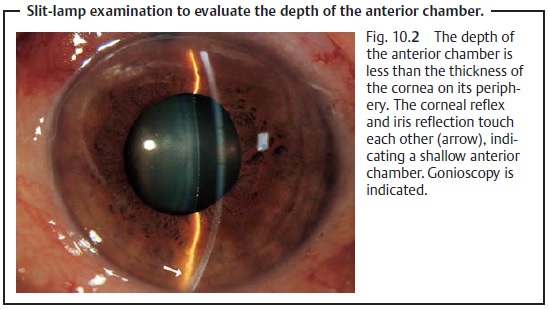
The central
and peripheral depth of the anterior chamber should be evaluated on the
basis of the thickness of the cornea. An anterior chamber that is less than
three times as deep as the thickness of the cornea in the center with a
peripheral depth less than the thickness of the cornea suggests a narrow angle
(Fig. 10.2). Gonioscopy is
essential for further evaluation.
To evaluate the depth of the anterior chamber
with a slit-lamp bio-microscope, select a narrow setting for the light beam.
The beam should strike the eye at a slight angle to the examiner’s line of sight.
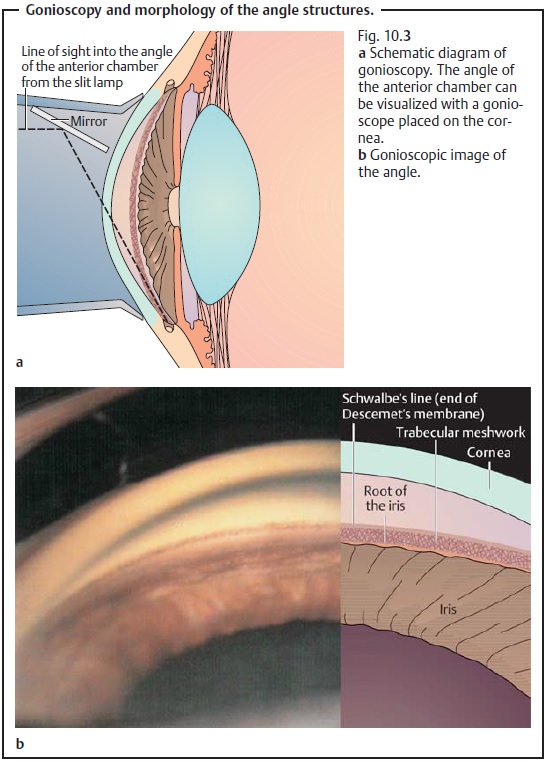
Gonioscopy
The angle of the anterior chamber is evaluated with a gonioscope placed directly on the cornea (Fig. 10.3a and b).
Gonioscopy can differentiate the following
conditions:
❖ Open angle: open angle glaucoma.
❖ Occluded angle: angle closure glaucoma.
❖ Angle access is narrowed: configuration with
imminent risk angle of an acute closure glaucoma.
❖ Angle is occluded: secondary angle closure
glaucoma, for example due to neovascularization in rubeosis iridis.
❖ Angle open but with inflammatory cellular
deposits, erythrocytes, or pig-ment in the trabecular meshwork: secondary open
angle glaucoma.
Gonioscopy is the examination of choice for
identifying the respective presenting form of glaucoma.
Measuring Intraocular Pressure
Palpation (Fig. 1.15):
Comparative palpation of both eyeballs is a pre-liminary examination that can
detect increased intraocular pressure.
❖If the examiner can indent the eyeball, which
fluctuates under palpation, pressure is less than 20 mm Hg.
❖An eyeball that is not resilient but rock hard
is a sign of about 60 – 70 mm Hg of pressure (acute angle closure glaucoma).
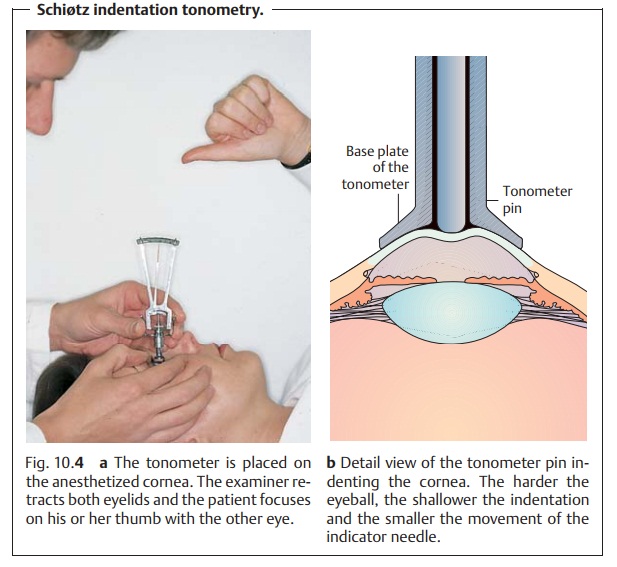
Schiøtz indentation tonometry (Figs. 10.4aandb):
This examinationmeas-ures the degree to which the cornea can
be indented in the supine patient. Thelower the intraocular pressure, the
deeper the tonometer pin sinks and the greater distance the needle moves.
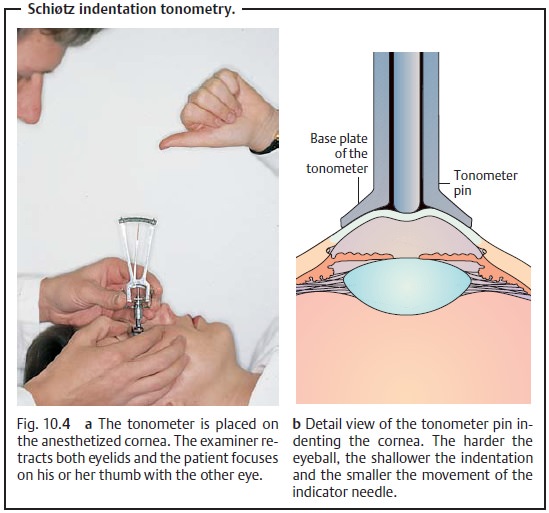
Indentation tonometry often provides inexact
results. For example the rigidity of the sclera is reduced in myopic eyes,
which will cause the tonome-ter pin to sink more deeply for that reason alone.
Because of this, indentation tonometry has been largely supplanted by
applanation tonometry.
Applanation tonometry:
This method is themost
common method of meas-uring intraocular pressure. It permits the examiner
to obtain a measurementon a sitting patient within a few seconds (Goldmann’s
method, see Fig. 10.5a – c) or on a
supine patient (Draeger’s method). A flat tonometer tip has adiameter of 3.06
mm for applanation of the cornea over a corresponding area (7.35 mm2). This method eliminates the rigidity of the sclera as a source
of error (see also tonometric self-examination).
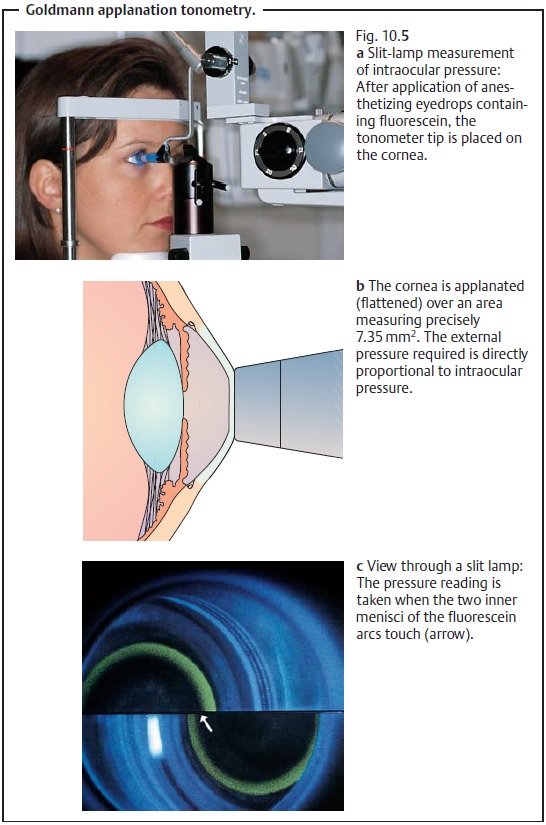
Intraocular pressure of 22 mm Hg is regarded
as suspicious. Caution:
Infection is possible in the presence of
conjunctivitis.
Pneumatic non-contact tonometry:
The electronic tonometer directs a3 ms blast of air against the cornea. The tonometer records the deflection of the cornea and calculates the intraocular pressure on the basis of this deformation.
Advantages:
❖Does not require the use of a topical
anesthetic.
❖ Non-contact measurement eliminates risk of
infection (may be used to measure intraocular pressure in the presence of
conjunctivitis).
Disadvantages:
❖ Calibration is difficult.
❖Precise measurements are possible only within
low to middle range pres-sures.
❖Cannot be used in the presence of corneal
scarring.
❖ Examination is unpleasant for the patient.
❖ Air flow is loud.
❖The instrument is more expensive to purchase
than an applanation tonometer.
Measuring the twenty-four-hour pressure curve (Fig. 10.6):
This examina-tion is performed to analyze fluctuations of the pressure level over a 24-hourperiod in patients with suspected glaucoma.
A single measurement may not be
representative. Only a 24-hour curve provides reliable information about the
pressure level.
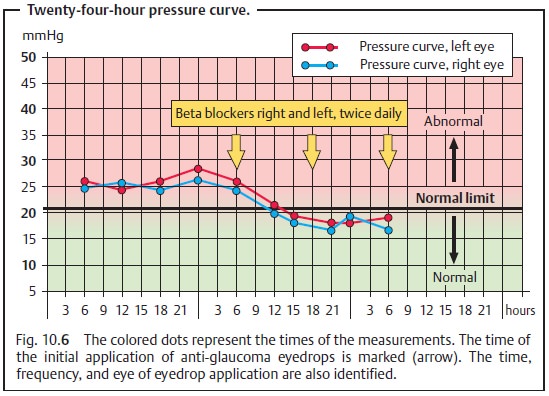
Intraocular pressure fluctuates in a rhythmic
pattern. The highest values frequently occur at night or in the early morning
hours. In normal patients, these fluctuations in intraocular pressure rarely
exceed 4 – 6 mm Hg.
Pressure is measured on the ward at 6:00 a.m.,
noon, 6:00 p.m.,
00 p.m., and midnight. Outpatient 24-hour pressure curves without night-time and early morning measurements are less reliable.
In glaucoma patients maintained on eyedrops,
special attention should be given to the time of application. Pressure is
measured immediately prior to applying the eyedrops. In this manner,
measurements are obtained when the effect of the eyedrops is weakest.
Tonometric self-examination:
Recent developments have made it possiblefor patients to measure intraocular pressure themselves at home in a manner similar to self-monitoring of blood pressure and blood glucose (Fig. 10.7). The patient tonometer makes it possible to obtain a 24-hour pressure curve from any number of measurements obtained under normal everyday conditions.
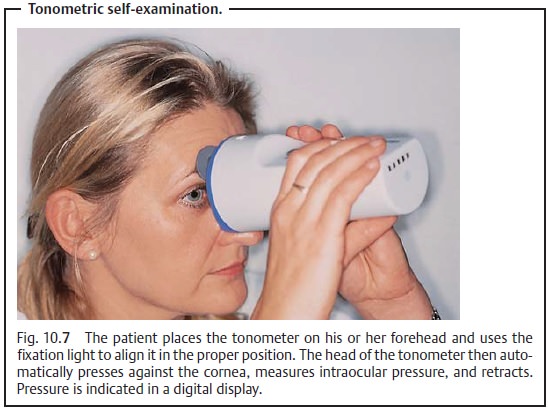
A patient tonometer may be prescribed in applicable
cases (such as increased risk of acute glaucoma). However, using the device
requires a certain degree of skill on the part of the patient. Patients who
have problems applying eye-drops are best advised not to attempt to use a
patient tonometer. Younger and well motivated patients are the best candidates
for tonometric self-examina-tion.
Optic Disk Ophthalmoscopy
The optic disk has a physiologic indentation
known as the optic cup. In the presence of persistently elevated intraocular
pressure, the optic cup becomes enlarged and can be evaluated by
ophthalmoscopy.
Stereoscopic examination of the optic disk
through a slit-lamp biomicroscope fitted with a contact lens provides a
three-dimensional image. The optic cup may be examined stereoscopically with
the pupil dilated.
The optic nerve is the eye’s “glaucoma memory.” Evaluating this
struc-ture will tell the examiner whether damage from glaucoma is present and
how far advanced it is.
Normal optic cup (Fig. 10.8): The
normal anatomy can vary widely. Largenormal optic cups are nearly always round and differ from the vertical elonga-tion of the optic cup
seen in eyes with glaucoma.
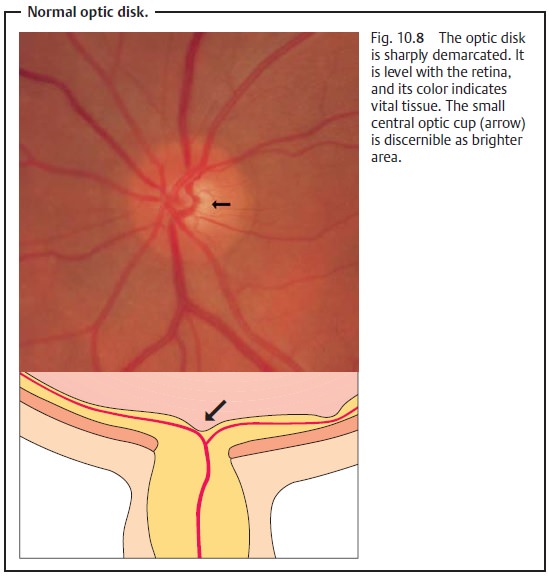
Documenting the optic disk: Recording findings in
sketchesis suitable
forroutine documentation and follow-up examination of the optic disk.Photo-graphing the optic disk with a fundus
camera permits longer-term follow-up. Stereoscopic
photography also provides a three-dimensional
image. Optic disk measurement and tomography can provide precise measurements of theoptic nerve.
Optic disk measurement.The area of the optic disk, optic cup, and neuroreti-nal rim (vital optic disk tissue) can be measured by planimetry on two-dimen-sional photographs of the optic nerve.
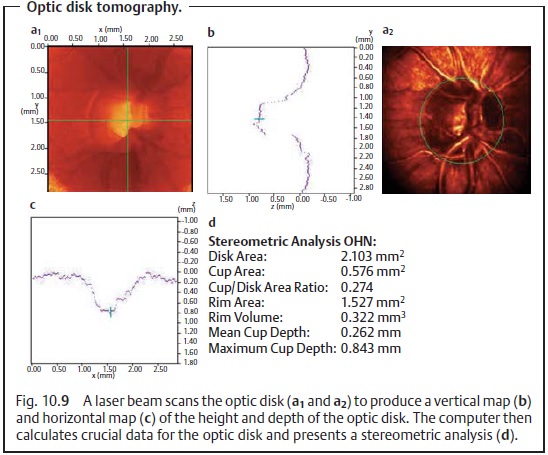
Optic disk tomography.Modern laser scanning ophthalmoscopes permitthree-dimensional documentation of the optic nerve (Fig. 10.9).
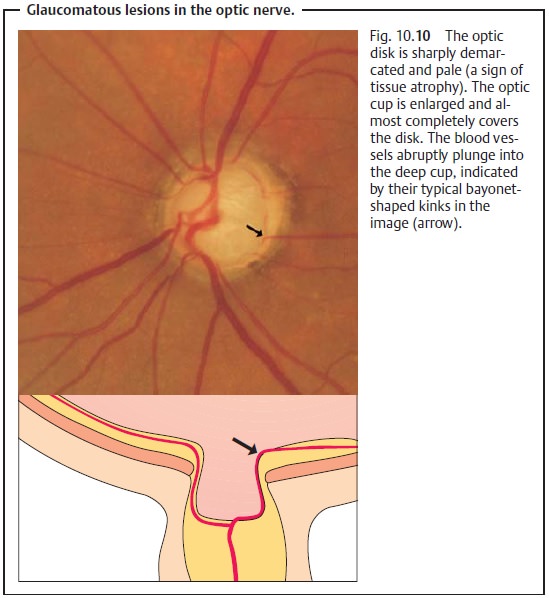
Glaucomatous changes in the optic nerve: Glaucoma produces typicalchanges in the shape of the optic cup. Progressive destruction of
nerve fibers,fibrous and vascular tissue, and glial tissue will be observable.
This tissue atrophy leads to an increase
in the size of the optic cup and to pale
discolorationof the optic disk (Fig. 10.10).
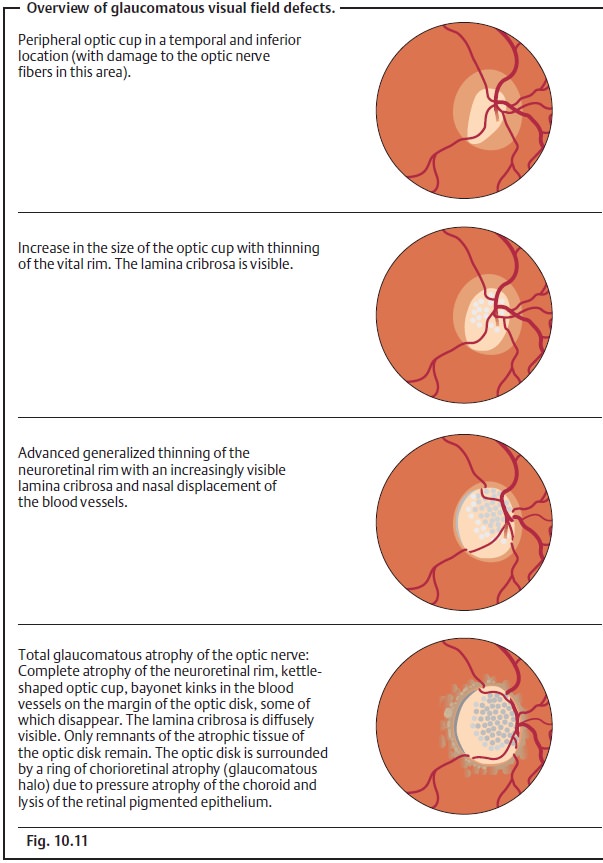
Progressive glaucomatous changes in the optic
disk are closely associated with increasing visual field defects (Figs. 10.11a – d).
Visual Field Testing
Detecting glaucoma as early as possible requires documenting glaucomatous visual field defects at the earliest possible stage. We know that glaucomatous visual field defects initially manifest themselves in the superior paracentral nasal visual field or, less frequently, in the inferior field, as relative scotomas that later progress to absolute scotomas (Fig. 10.11a – d).
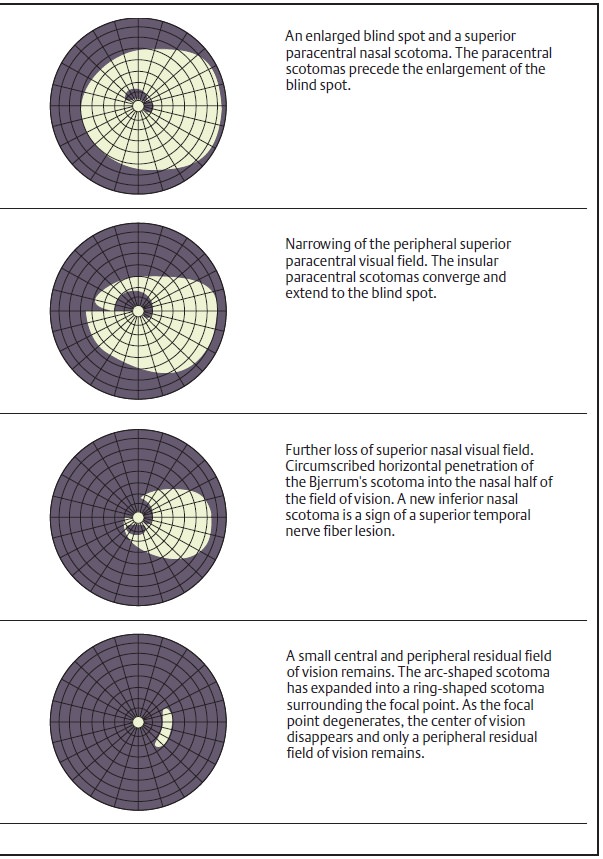
Computerized static perimetry (measurement of the sensitivity to differ-ences in light) is
superior to any kinetic method in detecting these early glaucomatous visual
field defects. Computer-controlled semiautomatic grid perimetry devices such as
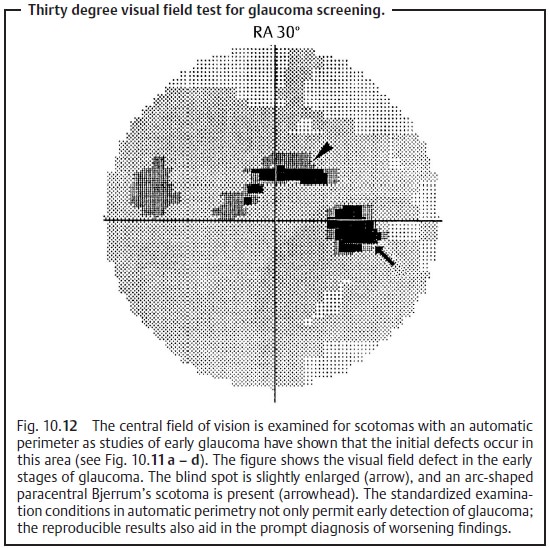
the Octopus or Humphrey field analyzer are used to examine the central 30
degree field of vision (modern campimetry; Fig. 10.12).
Reproducible visual field findings are important in follow-up to
exclude any enlargement of the defects.
Examination of the Retinal Nerve Fiber Layer
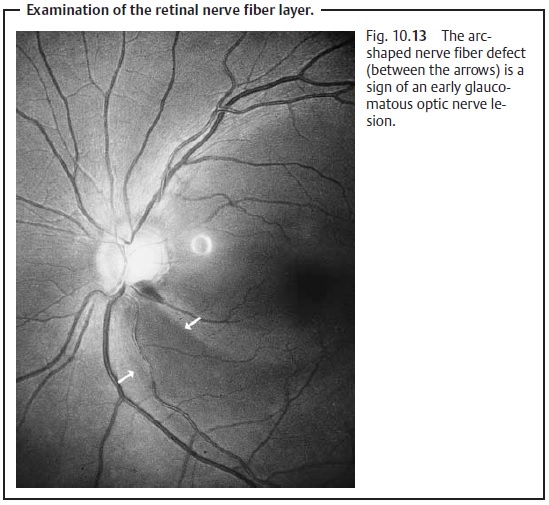
The retinal nerve fibers have a characteristic arrangement, which explains the typical visual field defects that occur in primary open angle glaucoma. In addition to the early progressive optic nerve and visual field defects, arc-shaped defects also occur in the nerve fiber layer. These defects may beobserved in light with red components (Fig. 10.13).
Related Topics