Chapter: Plant Biochemistry: A large diversity of isoprenoids has multiple functions in plant metabolism
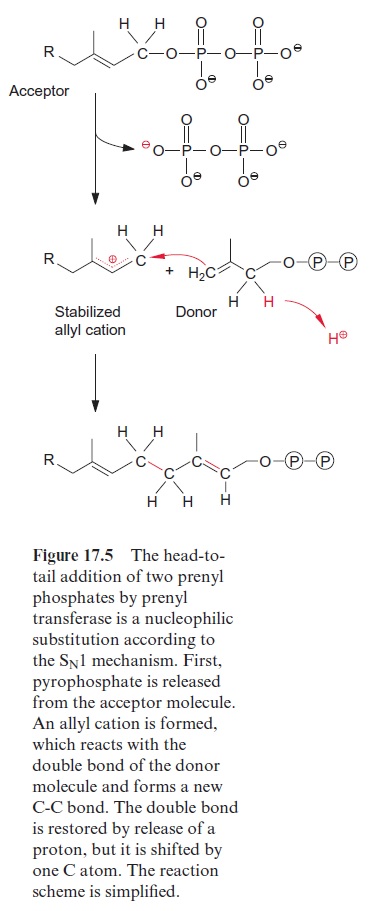
Geranylgeranyl pyrophosphate is the precursor for defense compounds, phytohormones, and carotenoids
Geranylgeranyl pyrophosphate is the precursor for defense compounds, phytohormones, and carotenoids
The cyclization of geranylgeranyl-PP leads to the formation of the diter-pene casbene (Fig. 17.13). Casbene is a phytoalexin of castor bean (Ricinus communis). The diterpene phorbol is an ester in the latex of plants of the spurge family (Euphorbiae). Phorbol acts as a toxin against herbivores; even skin contact causes severe inflammation. Since phorbol esters induce the formation of tumors, they are widely used in medical research. Geranylgeranyl-PP is also the precursor for the synthesis of gib-berellins, a group of phytohormones.
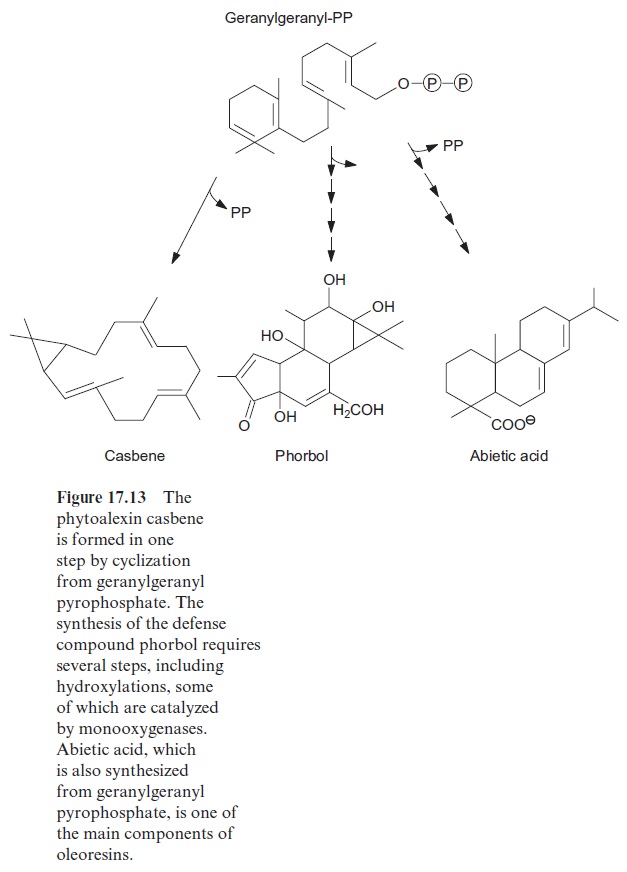
Oleoresins protect trees from parasites
In forests of the temperate zone, conifers are widely spread and often reach an old age, some species being far over 1,000 years old. This demonstrates that conifers have been very successful in protecting themselves from brows-ing enemies. One of the greatest threats is the bark beetle, which not only causes damage itself, but also opens the destroyed bark to fungal infections.
To protect themselves, the trees secrete oleoresins (tree resins), which seal the wound site and kill insects and fungi. The conifer oleoresins are a complex mixture of terpenoids, about half of which consist of a volatile turpentine fraction (many monoterpenes and some sesquiterpenes) and the other half of a non-volatile rosin fraction(diterpenes). The turpentine fraction contains a number of compounds that are toxic for insects and fungi (e.g., limonene (Fig. 17.5)). The rosin fraction is comprised of resin acids, the main compo-nent of which is abietic acid (Fig. 17.13). When the tree is wounded, stored oleoresin leaks through channels or is synthesized directly at the infected sites. It is presently being investigated how the toxic properties of the differ-ent components of the oleoresins affect different insects and fungi. Scientists are hopeful that such knowledge will make it possible to employ genetic engi-neering to enhance the parasite resistance of trees growing in large forests.
Carotene synthesis delivers pigments to plants and provides an important vitamin for humans
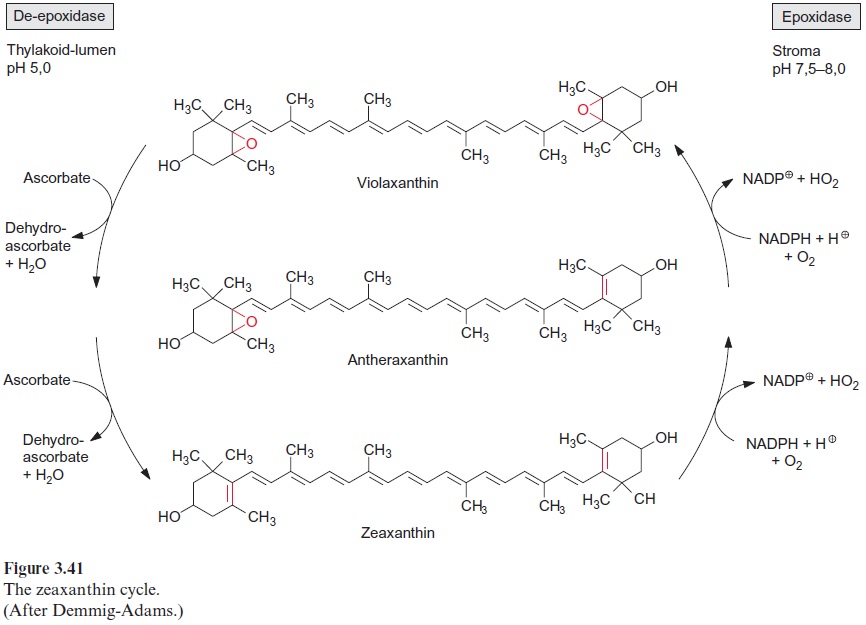
The synthesis of carotenoids requires two molecules of geranylgeranyl-PP, which, as in the synthesis of squalene, are linked by head-to-head condensation (Fig. 17.14). Upon release of the first pyrophosphate, the intermediate pre-phytoene pyrophosphate is formed, and the subsequent release of the second pyrophosphate results in the formation of phytoene, where the two prenyl residues are linked to each other by a carbon-carbon double bond. Catalyzed by two different desatu-rases, phytoene is converted to lycopene. According to recent results, these desaturations proceed via dehydrogenation reactions, in which hydrogen is transferred via FAD to O2. Cyclization of lycopene then results in the forma-tion of β-carotene. Another cyclase generates α-carotene. The hydroxylation of β-carotene leads to the xanthophyll zeaxanthin. The formation of the xan-thophyll violaxanthin from zeaxanthin is described in Figure 3.41.
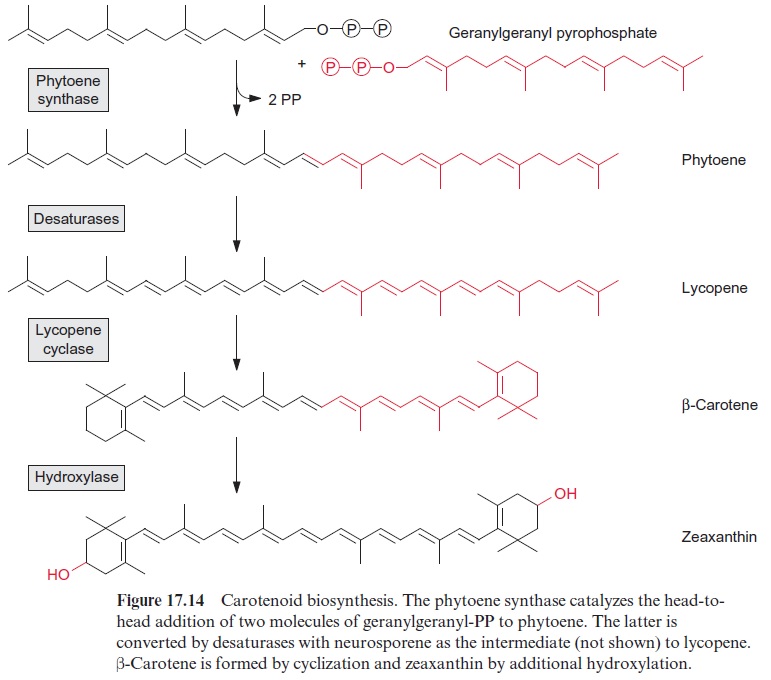
β-Carotene is the precursor for the synthesis of the visual pigment rhodopsin. Since β-carotene cannot be synthesized by humans, it is as pro vitaminA an essential part of the human diet. Hundreds of millions of peo-ple, especially in Asia, where rice dominates the diet and there is a lack of β-carotene in the food supply, suffer from severe provitaminA deficiency. Because of this, many children become blind. A recent success was the introduction of all the enzymes of the synthesis pathway from geranyl geranyl pyrophosphate to β-carotene into the endosperm of rice grains by genetic engineering. These transgenic rice lines produce β-carotene contai ning grains, with a yellowish color, and have therefore been called “golden rice.” Non-profit organizations have placed these transgenic rice lines at the disposal of many breeding stations in Asian countries, where they are at present crossed with local rice varieties. It is hoped that the serious pro vitaminA deficiency in wide parts of the world populations can be over-come through the cultivation of “golden rice.”
Related Topics